Flagship Pioneering’s Senda Bio secures $123M for new ‘programmable medicines’
Biotech startup Senda Biosciences is developing technology that yields a new type of medicine with two programming components: one directs the therapy to go to […]

Biotech startup Senda Biosciences is developing technology that yields a new type of medicine with two programming components: one directs the therapy to go to […]

Merck aims to bolster its genetic medicines pipeline through a new research alliance with Orna Therapeutics, a startup whose technology forms RNA into a circle […]

GSK is partnering with Mersana Therapeutics on the development of an antibody-drug conjugate that is approaching Phase 1 testing. Unlike other ADCs that deliver a […]

Alnylam Pharmaceuticals drug Onpattro, an FDA-approved treatment for nerve pain caused by hereditary transthyretin amyloidosis, now has Phase 3 data showing it can also help […]

Beam Therapeutics’ application to begin human testing of a cell therapy made with base-editing technology is now under an FDA clinical hold. In other clinical […]

The U.S. government has signed a supply agreement securing 66 million doses of Moderna’s Covid-19 vaccine booster shots, which are designed to address the omicron […]

Novartis is partnering with algorithms startup Anumana to develop a new artificial intelligence-based solution for cardiovascular diseases. The goal is to find ways to detect […]

BY KIM BELLARD I saw a great quote by Alfred North Whitehead the other day: “It is the business of the future to be dangerous.” […]

Kernal Biologics has technology that enables the delivery of messenger RNA therapies to a wider range of destinations in the body. The Series A financing […]

Sarepta Therapeutics has more clinical data showing the safety and efficacy of its experimental gene therapy for Duchenne muscular dystrophy. A pivotal Phase 3 test […]

An Intellia Therapeutics therapy that uses CRISPR to perform in vivo genomic edits has clinical data showing that the therapeutic effects continue for a year […]

Pfizer and Merck have each won FDA approvals in the past year for new pneumococcal vaccines, but GSK aims to top both of them with […]

ProFound Therapeutics emerged from stealth with technology that it claims has discovered tens of thousands of previously undiscovered proteins—many of them promising new drug targets. […]

Kelonia Therapeutics is developing cell therapies made by the in vivo engineering of a patient’s cells made possible by the targeted delivery of a genetic […]

The lead cancer drug candidate of Foghorn Therapeutics is now under a partial clinical hold following a patient death. According to the biotech, the patient […]

Sonata Therapeutics is developing drugs that reprogram cellular microenvironments to send signals intended to have curative effects. Cancer is the initial focus of the biotech, […]

Otsuka Pharmaceutical is ending its alliance with Akebia Therapeutics, a move that comes less than two months after the FDA rejected their partnered anemia drug, […]

PepGen’s technology improves the delivery of a therapy to more tissue types and the clinical-stage biotech plans to use its IPO cash to continue developing […]

Biogen struck a deal that could bring a digital therapeutic to its multiple sclerosis product portfolio. The drugmaker is partnering with startup MedRhythms in the […]

Biogen withdrew an application seeking approval in Europe for Alzheimer’s disease drug Aduhelm, a move that follows a negative opinion on the therapy issued by […]

Satellite Bio has emerged from stealth with technology for bioengineering tissue to restore organ function. The regenerative medicine startup, based on research from MIT and […]

Be Biopharma is developing cell therapies by engineering B cells to churn out therapeutic proteins with an initial focus on cancer and rare disease. The […]

Biogen’s Alzheimer’s disease drug Aduhelm will only be covered for Medicare beneficiaries participating in a clinical trial. The final coverage determination will have the effect […]

The FDA rejected Akebia Therapeutics drug vadadustat as a treatment for anemia caused by chronic kidney disease. According to the biotech, the agency cited safety […]

Single-cell analysis biotech Celsius Therapeutics has raised $83 million to advance its pipeline of drugs for autoimmune disorders and cancer. The startup’s lead autoimmune program, […]

LifeMine Therapeutics, a company that analyzes fungal genomes to find molecules that have potential as new medicines, has raised $175 million in financing. Among the […]

Alnylam Pharmaceuticals has proprietary lipid nanoparticle technology used in its FDA-approved RNA therapies. The biotech company claims that the messenger RNA Covid-19 vaccines from Moderna […]

Treatment aimed at people who cannot be vaccinated is boost to firm’s coronavirus portfolio AstraZeneca has received UK regulatory approval for its long-acting Covid-19 antibody […]

Novartis is paying Voyager Therapeutics $53 million up front to begin a partnership focused on the discovery of capsids for central nervous system disorder gene […]

Diagnostics developer Sherlock Biosciences has advanced its molecular diagnostic capabilities, which can now produce faster results on low-cost devices. The startup will use its Series […]

Artificial intelligence is a powerful tool for rare disease drug discovery. The founders of Vesalius Therapeutics say AI and machine learning can also be applied […]

New startup hC Bioscience is developing therapies based on transfer RNA, molecules that can be leveraged to address disease-causing proteins. The biotech joins several companies […]

Agios Pharmaceuticals has its third FDA-approved product, and the first under its new rare disease strategy. The regulatory decision also marks the first approved treatment […]

Several biotech companies are rising to the challenge of developing small molecules to drug RNA. Remix Therapeutics is joining the chase, but its different approach […]

Gene-editing medicines developer Intellia Therapeutics is paying $45 million up front to acquire Rewrite Therapeutics. The startup’s technology brings Intellia another tool for its genetic […]

The Center for Medicare and Medicaid Services proposes covering Biogen Alzheimer’s disease drug Aduhelm only for people enrolled in a clinical trial. In explaining this […]

Avrobio is stopping enrollment in a clinical trial for its Fabry disease program after new results showed variability in how the gene therapy was taken […]

The FDA has accepted Amylyx Pharmaceuticals’ submission for amyotrophic lateral sclerosis drug AMX0035 and it expects to issue a regulatory decision by late June. A […]

Six months after Biogen was awarded regulatory approval for its Alzheimer’s disease drug Aduhelm, the company the company is laying out a timeline for the […]

Eli Lilly is building its cancer drug pipeline via a research alliance with Foghorn Therapeutic, a biotech whose technology discovers drugs addressing targets in a […]

Radiopharmaceuticals deploy radiation to damage cancer DNA, but Curie Therapeutics sees these therapies opening the door to a wider range of ways to kill tumors. […]

Blueprint Medicines is paying $250 million up front to acquire Lengo Therapeutics, developer of targeted cancer therapies. The startup’s lead drug candidate is on track […]

Anglo-Swedish pharmaceutical company has come a long way since it fought off a takeover bid in 2014 Little expense has been spared at the giant […]

After years of delays and rising costs, AstraZeneca has formally unveiled its $1 billion R&D facility in Cambridge, said to be the biggest science lab […]

Generate Biomedicines applies artificial intelligence and machine learning to protein analysis, which it uses to program its protein therapies for particular applications. The startup now […]

Chroma Medicine is developing epigenetic therapies that turn genes on or off. The biotech startup has emerged from stealth mode backed by a $125 million […]

Arbor Biotechnologies raised new cash to keep up the pace in the chase for new genetic medicines. The MIT spinout applies artificial intelligence and machine […]

Alltrna, a biotech startup founded by Flagship Pioneering, is developing a new type of medicines based on a form of RNA called transfer RNA, or […]

Apple Tree Partners’ latest cancer biotech company, Marengo Therapeutics, is using antibodies to selectively activate T cells to fight cancer. Led by former Merck KGaA […]

Amylyx Pharmaceuticals has submitted its amyotrophic lateral sclerosis (ALS) drug for FDA review based on positive Phase 2 data after discussions with the agency over […]

Biogen amyotrophic lateral sclerosis drug, tofersen, failed a Phase 3 clinical trial, but the drug showed improvement according to secondary and exploratory goals of the […]

Biomanufacturing technologies company Resilience is partnering with Harvard in a deal that could see innovations from the university’s labs commercialized under new businesses. Resilience has […]

Rectify Pharmaceuticals is taking an approach to disease that led to a successful franchise of Vertex Pharmaceutical lung drugs and expanding it to more diseases […]

Pyxis Oncology’s IPO raised $168 million as the biotech steers toward the clinic with drug candidates and technology licensed from Pfizer. Theseus Pharma, IsoPlexis, and […]

The gene therapy pipeline of Voyager Therapeutics has hit several setbacks in the past year, but a strategy shift to its capsid technology is now […]

Vedanta Biosciences is developing its microbiome therapy with support from the Biomedical Advanced Research and Development Authority. Under terms of that contract, the positive Phase […]

Biotech startup Exo Therapeutics aims to overcome the challenges of drugging proteins by targeting exosites, locations that modulate enzyme activity. The company has four small […]

Cross-border biotech Anji Pharma closed $70 million in Series B funding to advance a drug pipeline that includes metabolic and cancer drugs. The biotech’s business […]

The FDA has approved the first biosimilar that references the blockbuster Roche drug Lucentis. The Biogen and Samsung BioLogics joint venture that developed the biologic […]

Luminopia, a startup with a digital therapeutic for amblyopia, or lazy eye, recently published the results of a phase 3 study showing its TV-based treatment […]

A Roche drug that failed as a treatment for neurological disorders is now the lead program for Disc Medicines. CEO John Quisel said Roche’s clinical […]

Atavistik Bio is one of several companies discovering and developing drugs that work by allostery, binding to less obvious sites of a target protein. Acting […]

Customers can order medicines for mild ailments, makeup, toiletries and more from 14 stores The pharmacy chain Boots will become the latest large retailer to […]

Though Gilead Sciences leads the group of companies developing immunotherapies that target the cancer protein CD47, Pfizer contends that molecules it is getting through the […]

For much of this summer, Sesen Bio said all of the FDA’s communications pointed toward regulatory approval for the biotech’s bladder cancer drug. Though Sesen […]

The artificial intelligence-based technology of Iterative Scopes brings computer vision analysis to endoscopic images. The startup’s technology was initially developed to assist gastroenterologists in finding […]

An AstraZeneca lupus drug has been awarded FDA approval for treating the most common form of the autoimmune disorder. The regulatory decision gives AstraZeneca a […]

Patients taking new Alzheimer’s disease drug Aduhelm will be evaluated in a real-world study lasting up to five years. Biogen announced plans for the observational […]

The engineered viruses used to deliver gene therapies can spark complications and they can’t be re-dosed. Ring Therapeutics says viruses that evolved with humans can […]

Seres Therapeutics’ ambitions to treat ulcerative colitis with a microbiome drug were dealt a setback as its experimental therapy failed in Phase 2 testing. The […]

An amyotrophic lateral sclerosis drug from Amylyx Pharmaceuticals is being prepared for Phase 3 clinical testing on track to begin later this quarter. To support […]

Startup PAQ Therapeutics is developing drugs work like Pac Man, gobbling up components of a cell associated with disease. The biotech has closed $30 million […]

By paying $125 million up front for rights to multiple sclerosis drug candidate orelabrutinib, Biogen secures a place among those companies trying to treat the […]

A hemophilia A patient in a Sigilon Therapeutics clinical trial developed a well-known complication to treatments for the blood disorder. The potential problem for Sigilon […]

The drug label for Biogen Alzheimer’s disease drug Aduhelm has been revised to clarify that its use is for patients with mild forms of the […]

Computer – the most powerful in Britain – will use AI to find new cures and deepen understanding of diseases The UK’s most powerful supercomputer, […]

Fresh off its first FDA approval, Apellis Pharmaceuticals is teaming up with Beam Therapeutics, a partnership that will use Beam’s base-editing technology to develop new […]

Intellia Therapeutics has early clinical trial data showing that its CRISPR-based therapy for a rare disease can edit genes inside the body safely and effectively. […]

Lyell Immunopharma and Verve Therapeutics are still preclinical, but they’re bring novel genetic approaches to the treatment of cancer and cardiovascular disease. Now the two […]

CAMP4 Therapeutics is developing a new kind of RNA therapy that treats disease by upregulating gene expression. The startup has raised $45 million as CEO […]

A Sage Therapeutics depression drug that failed a pivotal study in 2019 has positive preliminary results from a new Phase 3 clinical trial, but the […]

Senda Biosciences is developing drugs based on an understanding of intersystems biology—the way that humans interact with bacteria and plants. The startup, founded by Flagship […]

Biogen is still discussing with the FDA the clinical trial design to confirm the benefit offered by its newly approved Alzheimer’s disease drug, Aduhelm. According […]

A Biogen Alzheimer’s drug that clears amyloid plaque from the brain has been granted accelerated approved by the FDA. The drug, which will be marketed […]

MorphoSys is acquiring Constellation Pharmaceuticals and its late-stage myelofibrosis drug in a deal that values the epigenetics biotech at $1.7 billion. Germany-based MorphoSys will finance […]

Moderna released preliminary clinical data showing its Covid-19 vaccine produced efficacy in adolescents that was comparable to what was seen in adults. The company plans […]

Biogen is reaching across the Charles River, striking up a gene therapy manufacturing alliance with synthetic biology firm Ginkgo Bioworks. Ginkgo will use its technology […]

BlueRock Therapeutics, a Bayer subsidiary, is teaming up with Opsis Therapeutics and Fujifilm Cellular Dynamics in an R&D pact focused on developing stem cell therapies […]

A Biogen gene therapy for a rare, inherited eye disorder that leads to blindness has failed a key clinical trial. The preliminary results are the […]

Biogen paid $18 million to acquire a drug that TMS Co. of Japan tested in patients with acute ischemic stroke. The acquisition follows the report […]

Incubated by Flagship Pioneering, biotech firm Laronde is developing endless RNA, or eRNA, a new class of medicines that goes beyond messenger RNA-based therapies. Flagship […]

Dyno Therapeutics’ technology for designing viral vectors that deliver gene therapies has led to partnerships with Novartis, Sarepta Therapeutics, and Roche. With the Series A […]

Neuroelectrics is planning a pivotal test of its wearable medical device, which delivers brain electrical stimulation to treat epilepsy. The startup’s Series A round of […]

Vaccitech and Werewolf Therapeutics joined the public markets, raising a combined $230 million. The vaccine developer and the cancer immunotherapy biotech will apply the IPO […]

Alnylam Pharmaceuticals has filed for FDA approval of vutrisiran, which could become the biotech’s fourth drug that works by RNA interference. Meanwhile the company is […]

Vertex Pharmaceuticals is collaborating with Obsidian Therapeutics to develop up to five new gene editing therapies. Obsidian’s technology precisely controls protein expression, a capability the […]

The FDA placed a clinical hold on a KalVista Pharmaceuticals drug being readied for mid-stage studies in a rare, inherited disorder. Meanwhile, the FDA has […]

Relay Therapeutics has its own technology that applies artificial intelligence and machine learning techniques to drug discovery. Its $85 million acquisition of ZebiAI gives it […]

Amylyx’s ALS drug met the goals of a mid-stage study, paving the way for regulatory submissions in Europe and Canada. But the FDA is requiring […]

Tango Therapeutics’ SPAC merger will infuse the biotech with $353 million to fund clinical trials for its cancer drugs based on synthetic lethality. Tango’s lead […]

With two cancer programs already making progress, Repertoire Immune Medicines will use the Series B financing to expand its immune synapse research to autoimmune disorders […]

The FDA increased the maximum number of doses permitted for each vial of Moderna’s Covid-19 vaccine, a change intended to make more doses available. The […]

Omega Therapeutics has raised more than $210 million in the past eight months for R&D of medicines that regulate genes. The company’s lead therapeutic candidate […]

AstraZeneca reported that its Covid-19 vaccine was 79% effective at preventing severe infection and 100% effective at preventing hospitalization. The company expects to file a […]

In two pivotal studies years apart, Aveo Oncology’s drug tivozanib did not help kidney cancer patients live longer overall compared to the standard of care. […]

The FDA decision gives Sarepta Therapeutics its third approved drug for Duchenne muscular dystrophy. The accelerated approval requires the biotech to conduct additional clinical testing […]

Cancer is a known risk of lentiviral-based gene therapies. Though bluebird says its gene therapy is engineered to avoid that risk, the company is suspending […]

The Neurocrine decision follows a clinical hold placed on the Parkinson’s program in December. It’s Voyager’s second loss of an R&D partner in the past […]

The latest funding brings total investment in the company to $750 million. The money has gone, in part, toward licensing of promising drug candidates in […]
The company published interim data from the Phase I study of mRNA-1273 in 40 older and elderly adults, showing responses comparable to those seen in […]

The six-year-old Google-backed company was founded by three MIT physicists and hopes to use its technology to make drug discovery faster and less cumbersome by […]

The company announced Wednesday the FDA approval of its liquid biopsy test, the second to win an agency nod in less than a month, saying […]

The biotech company reached a deal with the European Commission to supply an initial 80 million doses, while European Union member states will have the […]

The drugmaker emphasized the full global rights it would acquire to Momenta’s lead asset, nipocalimab, which it is developing for several autoantibody-driven diseases and that […]
The agreement, which includes 100 million doses of mRNA-1273 and an option to acquire 400 million more, brings the total amount of money the biotech […]

Biogen and Eisai said the FDA had accepted and given priority review to their application for aducanumab. The drug will undergo an advisory committee meeting […]
The companies’ resubmission of their application Wednesday seeking approval for idecabtagene vicleucel was in line with the timeline they provided in May, when the FDA […]
The study design is still being finalized, and the company is thus not yet divulging too many details. However, CEO David Daly said it would […]
The company had received $483 million from BARDA in April to fund the development of the vaccine, mRNA-1273, through Phase III, but that amount was […]
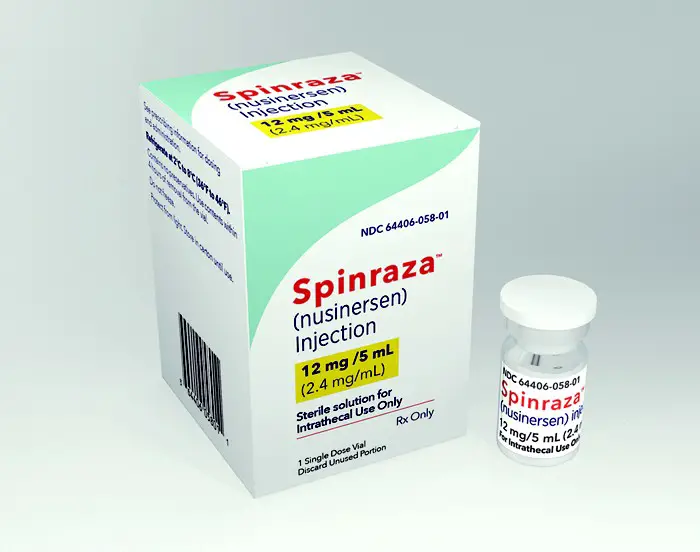
The company plans to enroll 60 patients who have received its drug, Spinraza, following suboptimal response to Novartis’ Zolgensma, which was approved last year. The […]

The company, whose drug-discovery efforts are focused on protein motion and detection of interactions that occur anywhere on a protein rather than only at active […]

Evaluation of durability is still under evaluation, but the responses to mRNA-1273 show neutralizing antibody titers multiple times higher than those of recovered patients. Additionally, […]

Under the deal, Blueprint will additionally be eligible for more than $900M in milestone payments plus royalties for the RET inhibitor pralsetinib. The deal includes […]

The company said it is on track to start its 30,000-participant Phase III trial this month and has finished manufacturing enough supplies of the vaccine […]

The FDA has 60 days to decide whether or not to accept the application. An analyst wrote that the agency accepting it with a priority […]

Thanks to the success of the Human Genome Project, 20 years ago this week, scientists can track biology and disease at a molecular level Twenty […]
Copyright © 2024 | WordPress Theme by MH Themes