Date:
July 27, 2020
Issue #:
1603
Summary:
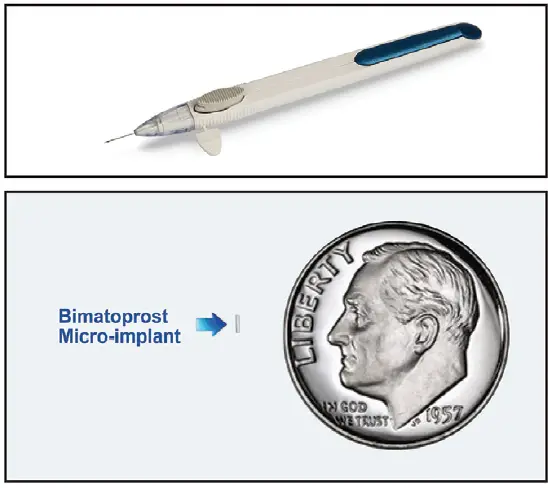
The FDA has approved an intracameral implant
containing the prostaglandin analog bimatoprost
(Durysta – Allergan) for reduction of intraocular
pressure (IOP) in patients with open-angle glaucoma
or ocular hypertension. Bimatoprost is also
available in 0.01% (Lumigan) and 0.03% (generics)
ophthalmic solutions for the same indication and in
a 0.03% solution (Latisse, and generics) for eyelash
enhancement. Durysta is the first ocular implant to
become available in the US for treatment of glaucoma.
containing the prostaglandin analog bimatoprost
(Durysta – Allergan) for reduction of intraocular
pressure (IOP) in patients with open-angle glaucoma
or ocular hypertension. Bimatoprost is also
available in 0.01% (Lumigan) and 0.03% (generics)
ophthalmic solutions for the same indication and in
a 0.03% solution (Latisse, and generics) for eyelash
enhancement. Durysta is the first ocular implant to
become available in the US for treatment of glaucoma.