
Roche to co-develop remdesivir rival in partnership with Atea
Roche may not be the first company that comes to mind for a COVID-19 treatment, but the Swiss firm is taking a big swing at […]

Roche may not be the first company that comes to mind for a COVID-19 treatment, but the Swiss firm is taking a big swing at […]

What You Should Know: – Babyscripts today announced a commercial partnership with Roche Diagnostics, a division of the world’s largest biotech company and a global […]

Shots: The companies will advance the prasinezumab into a P-IIb study in patients with early Parkinson’s disease based on positive signals of efficacy consistent with […]

Shots: Genesis to receive an up front and is eligible to receive pre/ clinical and regulatory milestones along with royalties on sales of approved drugs […]

The deal with Roche subsidiary Genentech is likely not the last for Genesis Therapeutics, which is based on AI research undertaken at Stanford. Such partnerships […]

Combination therapies involving Roche and AbbVie’s cancer drug Venclexta have been formally approved by the FDA in acute myeloid leukaemia, following supportive data from late-stage […]

Novartis’s Luxturna (voretigene neparvovec) Receives Health Canada Approval as the First Gene Replacement Therapy for Inherited Retinal Disease Published: Oct 15, 2020 | Tags: approval, […]

Roche’s pharmaceutical revenues have been hit by falling sales in its pharma division in Q3 but a spike in demand for COVID-19 tests spared the […]

Shots: Dyno to receive upfront and is eligible to receive ~$1.8B milestones including development and commercial milestones along with royalties on any product emerges during […]

Supplies of swabs for coronavirus and other critical NHS tests for diseases like cancer, diabetes and heart disease have been threatened by a failure at […]

Supplies of swabs for coronavirus and other critical NHS tests for diseases like cancer, diabetes and heart disease have been threatened by a failure at […]
Roche’s Genentech unit has paid $200 million to develop an individualised cancer vaccine with Norwegian biotech Vaccibody, which focuses on targets known as neoantigens that […]

1. BioMarin Reports NDA Submission to the US FDA for Vosoritide to Treat Children with Achondroplasia Published: Aug 20, 2020 | Tags: BioMarin, Reports, NDA, […]

New insights into smell and taste loss symptoms, artificial intelligence and further encouraging data from a vaccine candidate hit the headlines this week as the […]

It’s mission accomplished for Novartis after the FDA approved ofatumumab for multiple sclerosis, completing a project where the former cancer drug has been repurposed. The […]

NICE has recommended regular NHS funding for Roche’s Polivy in certain lymphoma patients in final draft guidance, overturning a previous rejection. The new guidance recommends […]

Roche has agreed to help Regeneron manufacture its COVID-19 antibody cocktail REGN-COV2, which started late-stage clinical testing a few weeks ago, in a deal that […]
The deal brings Roche’s considerable manufacturing heft to Regeneron’s efforts to develop the two-antibody cocktail, REGN-COV2. Regeneron will distribute the drug in the U.S., while […]

Shots: The approval is based on P-III IMbrave150 study assessing Tecentriq (1200 mg) + bevacizumab (15 mg/kg, q3w, IV) vs sorafenib (400mg, bid) in 501 […]

Shots: The two global companies collaborated to develop, manufacture, and distribute REGN-COV2 across the globe. The agreement is expected to increase the supply of REGN-COV2 […]

New strategy, new outlook: Sanofi takes another step, gains full control over the Principia’s MS treatment Sanofi has inked a definitive agreement with Principia Biopharma […]
The agency approved Enspryng for the disease, which is often misdiagnosed as multiple sclerosis and can cause blindness, muscle weakness and paralysis.
Shots: The approval is supported by P-III SAkuraStar and SAkuraSky studies involve assessing Enspryng (120mg, SC, q4w) as a monothx. & as an add-on therapy […]

The FDA has approved Roche’s satralizumab for the rare autoimmune disorder Neuromyelitis Optica Spectrum Disorder (NMOSD), under the brand name Enspryng. Roche is taking on […]

1. Roche’s Evrysdi (risdiplam) Receives the US FDA’s Approval for SMA in Adults and Children Published: Aug 10, 2020 | Tags: Roche, Evrysdi, risdiplam, Receives, US, […]

The FDA has accepted TG Therapeutics’ filing for its lymphoma drug umbralisib, which if approved could compete against a combination therapy from Bristol-Myers Squibb and […]

Shots: The US FDA has accepted the sBLA for a new self-administration option for Xolair across all approved indications in the US. The company anticipates […]
Shots: Celleron will receive an exclusive global right for the clinical development, manufacturing, and commercialization of emactuzumab (formerly RG7155 and RO5509554). The companies expect the […]
UK biotech Celleron Therapeutics has bought a licence to develop, manufacture and market emactuzumab, an unwanted drug from Roche’s pipeline that could be used in […]
Roche Set to Compete With Biogen and Novartis in the SMA Market With its First Oral Therapy The US FDA has recommended the use of […]

Analysts noted that Evrysdi would be priced significantly lower than Biogen’s Spinraza or Novartis’ Zolgensma. While this will provide a market advantage, it would also […]
Roche’s late-stage trial programme for ulcerative colitis candidate etrolizumab has generated some positive and negative results, but leans firmly towards the latter. The Swiss drugmaker […]

Shots: In the HIBISCUS I induction study, in patients without prior anti-TNF treatment, etrolizumab met its 1EPs while in HIBISCUS II study in the same […]

Roche is hoping to undercut hugely expensive rivals after the FDA approved its oral spinal muscular atrophy (SMA) drug Evrysdi (risdiplam). Evrysdi is the third […]

Shots: The US FDA has approved Evrysdi to treat SMA in adults and children ≥ 2mos. The approval is based on two clinical studies designed […]
Roche’s hopes of extending the use of its PD-L1 inhibitor Tecentriq in triple-negative breast cancer (TNBC) have been dashed by a late-stage trial failure looking […]

Shots: The P-III IMpassion131 study involves assessing of Tecentriq + paclitaxel vs PBO + paclitaxel, in 651 people in a ratio (2:1) with previously untreated, […]

Shots: The US FDA has granted de novo class II for cobas EBV test, providing HCPs a tool for monitoring transplant patients at risk for […]
Shares in Redx Pharma were soaring this morning after it licensed its preclinical fibrotic disease drug RXC006 to AstraZeneca. The company’s shares were valued as […]

Roche’s tumour-agnostic therapy Rozlytrek has been approved in the European Union for people with tumours with the NTRK mutation, meaning Bayer’s class rival faces competition […]

Shots: The approval is based on integrated analysis including data from 74 people with LA/ m-NTRK fusion-positive solid tumors (14 tumor types) & 161 people […]

1. Oxford Biomedica Signs Three Year Clinical Supply Agreement with Axovant to Manufacture and Supply AXO-Lenti-PD for Parkinson’s Disease Published: Jul 31, 2020 | Tags: Oxford […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]
Shots: The US FDA has approved the new VENTANA HER2 Dual ISH DNA Probe Cocktail assay to detect HER2 biomarker in BC and as a […]
Roche’s checkpoint inhibitor Tecentriq has been cleared for another new use in the US – in melanoma – but could struggle to displace rival drugs […]

Shots: The approval is based on P-III IMspire150 study assessing Tecentriq (atezolizumab) + Cotellic (cobimetinib) + Zelboraf (vemurafenib) vs PBO + Cotellic + Zelboraf in […]

Roche already has one Alzheimer’s candidate in its pipeline targeting tau, but has added a second via a deal with Belgian drugmaker UCB worth up […]

The company said that the COVACTA trial of Actemra in Covid-19 pneumonia had failed to meet its primary endpoint. Earlier this month, Regeneron and Sanofi […]
Shots: The P-III COVACTA study involves assessing of Actemra/RoActemra (IV) + SOC vs PBO + SOC in adult patients hospitalized with severe COVID-19 associated pneumonia. […]

Roche’s anti-inflammatory drug Actemra has failed in a phase 3 clinical trial that was testing to see if it improved the condition of patients with […]

Shots: UCB to receive $120M and is eligible to receive $2B as cost reimbursement, development, and commercial milestones as well as royalties on sales of […]

1. Gilead and Galapagos’ Jyseleca (filgotinib) Receive the CHMP’s Positive Opinion for Moderate to Severe Rheumatoid Arthritis Published: Jul 24, 2020 | Tags: Gilead, Galapagos, Jyseleca, […]
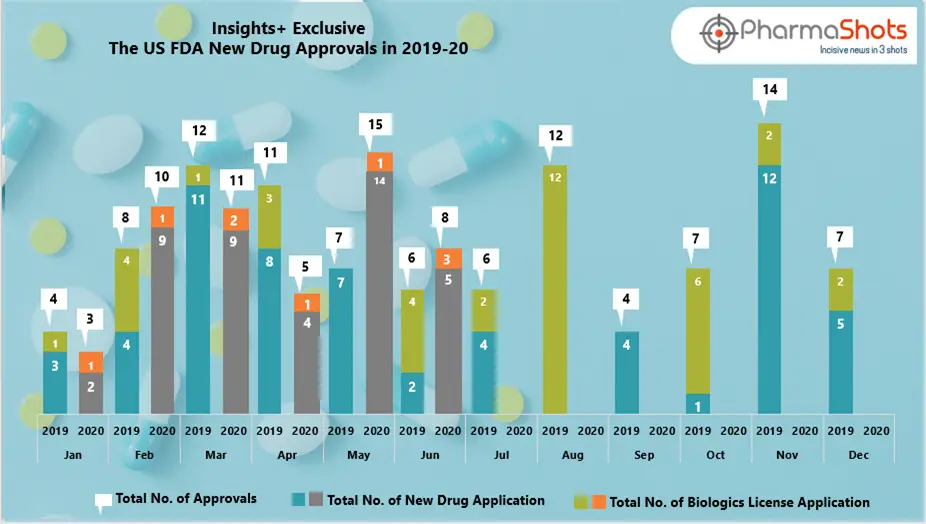
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]

Roche has announced half-year results showing the COVID-19 pandemic affected sales of its key products, with currency headwinds also weighing on revenues. The Swiss pharma’s […]

The companies will test Biofourmis’ sensor and artificial intelligence system in a clinical trial of more than 120 women with endometriosis.

Shots: Jnana to receive $40M upfront payment in cash and is eligible to get ~$1B as research funding, preclinical, development, and commercialization milestone along with […]

Shots: The real-world evidence partnership will allow the Roche to access PicnicHealth’s set of de-identified patient records to gain insights on certain diseases and treatments […]
1. Eli Lilly Reports Results of Mirikizumab in P-III OASIS-2 Study to Treat Moderate to Severe Plaque Psoriasis Published: Jul 17, 2020 | Tags: Eli Lilly, […]

The companies will use a combination of more than seven years of medical record data with five years of prospective data on 5,000 MS patients, […]

Under the deal, Blueprint will additionally be eligible for more than $900M in milestone payments plus royalties for the RET inhibitor pralsetinib. The deal includes […]

Pharma knows there is a need for clean, reusable, FAIR data to fuel effective machine learning-driven R&D – but driving effective information and knowledge management […]
Copyright © 2024 | WordPress Theme by MH Themes