
Second unanimous FDA adcomm vote boosts bluebird bio
bluebird bio started last week on tenterhooks, as FDA advisors cast their eyes over data for two gene therapy candidates that are key to its […]

bluebird bio started last week on tenterhooks, as FDA advisors cast their eyes over data for two gene therapy candidates that are key to its […]

The FDA may have safety concerns abut bluebird bio’s gene therapy for rare, fatal disease cerebral adrenoleukodystrophy (CALD), but its advisors believe its benefits far […]

US cost-effectiveness watchdog ICER has handed bluebird bio some good news ahead of its FDA advisory committee meeting for rare blood disorder gene therapy beti-cel […]

Gene therapy specialist bluebird bio has said 30% of its workforce will lose their jobs, in a bid to conserve cash as it tried to […]

As bluebird bio struggles to get its gene therapies over the finishing line, the company says its financial position raises “substantial doubt” about its ability […]

Europe market has proved so hostile to gene therapies when it comes to pricing and reimbursement for gene therapies that bluebird bio has decided to […]

Bluebird bio has its first approval for Skysona (formerly Lenti-D), its gene therapy for children with the rare disease cerebral adrenoleukodystrophy (CALD), after getting the […]

In an interview with PharmaShots, Rich Colvin, M.D., Ph.D., Interim Chief Medical Officer at bluebird bio shares his views on the Skysona and its working […]

The EU medicines regulator has said that there is some evidence to suggest a possible link between mRNA-based vaccines for COVID-19 and rare cases of […]

Johnson & Johnson has secured a six-month FDA review for its multiple myeloma CAR-T therapy ciltacabtagene autoleucel (cilta-cel), narrowing the lead for Bristol-Myers Squibb and […]

The UK has been a global leader in introducing cell and gene therapies that can potentially transform the lives of patients with debilitating diseases – […]

US biotech bluebird bio has had a challenging time in the last few months, so a recommendation for EU approval of its gene therapy for […]

The FDA has approved a Bristol Myers Squibb cell therapy made by engineering a patient’s immune cells to target a cancer protein called BCMA. The […]

Shots: The approval is based on the P-II KarMMa trial that involves assessing Abecma in 127 patients with r/r MM prior treated with 3L+ therapy […]

bluebird bio has announced new clinical trial data from its gene therapy for patients with cerebral adrenoleukodystrophy (CALD), including long-term follow-up data. CALD is the […]

Bluebird bio said independent analyses found that the gene therapy inserted its payload at a site not known to cause cancer. Based on the results, […]

bluebird bio is to ask regulators to restart clinical studies of its LentiGlobin for sickle cell disease, after an investigation concluded that a case of […]

Bluebird Bio’s decision to hit pause on the launch of Zynteglo for beta thalassaemia after two cases of cancer were seen in a clinical trial […]

Cancer is a known risk of lentiviral-based gene therapies. Though bluebird says its gene therapy is engineered to avoid that risk, the company is suspending […]

It’s back to the drawing board for bluebird bio and its discussions with NICE, which has rejected its beta thalassaemia gene therapy Zynteglo for regular […]

Medivir, IGM Biosciences enters into an exclusive licensing agreement for Birinapant Medivir AB has entered into an exclusive licensing agreement with IGM Biosciences to receive […]

Shots: Bluebird plans to separate its severe genetic disease & oncology businesses into differentiated and independent publicly traded companies. The company will retain focus on […]

US biotech bluebird bio has announced plans to split into two this year, with a separate oncology business spinning off as the company prepares to […]

bluebird bio has presented long-term data from its Zinteglo one-time gene therapy for the blood disorder beta-thalassaemia, as the company continues talks with payers in […]

Bluebird bio could be just a few months away from approval of its gene therapy for rare disease cerebral adrenoleukodystrophy (CALD) in the EU, after […]
The companies’ resubmission of their application Wednesday seeking approval for idecabtagene vicleucel was in line with the timeline they provided in May, when the FDA […]
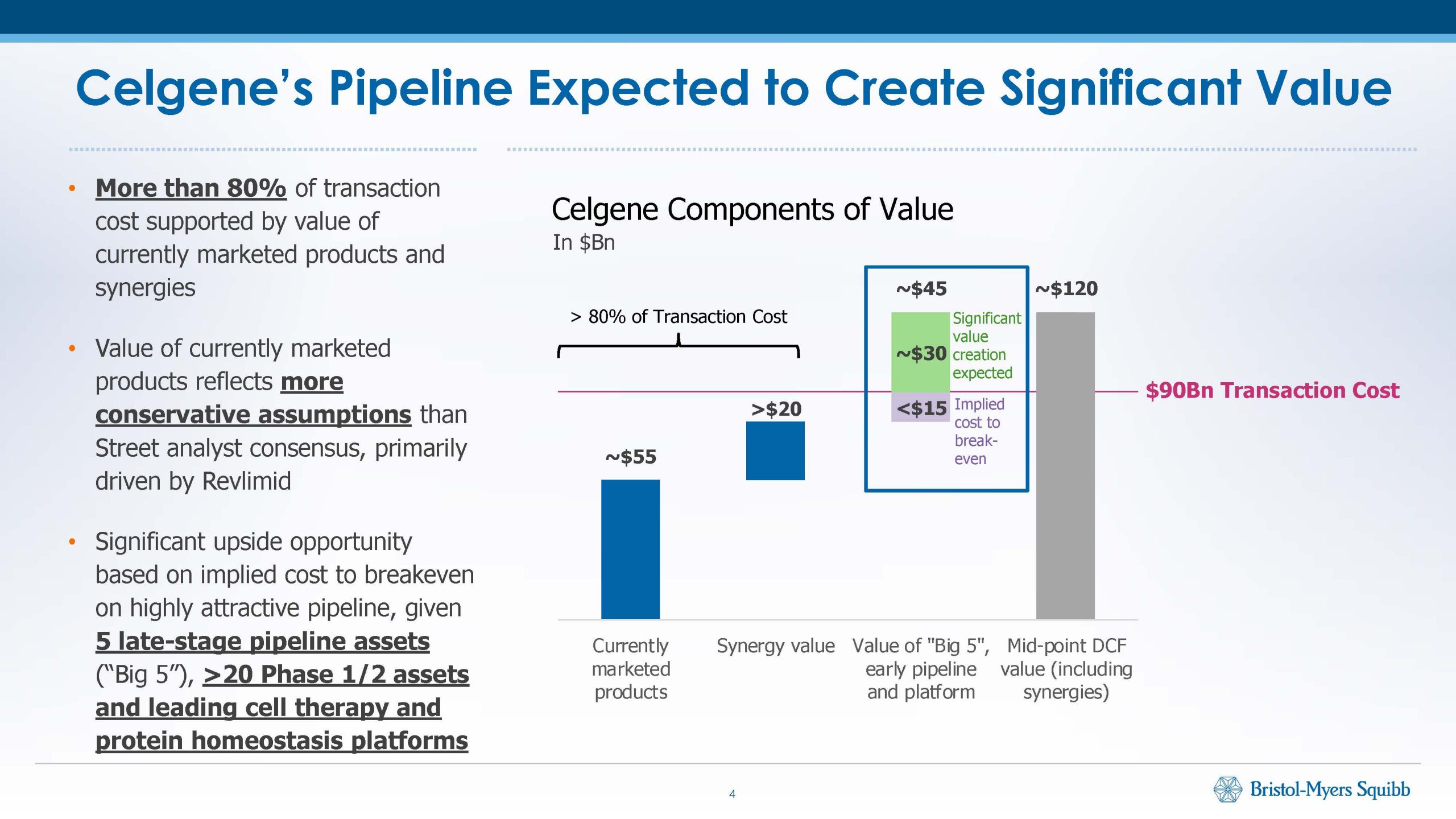
Bristol-Myers Squibb and bluebird bio have refiled their CAR-T therapy for multiple myeloma, leaving the outcome of a three drug “bet” with former Celgene shareholders […]
Copyright © 2024 | WordPress Theme by MH Themes