
Project seeks to chart regulatory paths for digital products
The nonprofit Digital Medicine Society is partnering with the likes of Google and the FDA to develop a free resource designed to help digital health […]

The nonprofit Digital Medicine Society is partnering with the likes of Google and the FDA to develop a free resource designed to help digital health […]

Abbott is already a big player in the wearable glucose-monitoring sector, and has started working on a new device that will measure both glucose and […]

Abbott has joined forces with two other companies to bring an automated insulin delivery (AID) system to people with diabetes in Europe that will monitor […]

The latest version of Abbott’s FreeStyle Libre continuous glucose monitor is now available via the NHS to eligible people with diabetes in the UK. Abbott’s […]

The FDA has approved expanded labelling for Abbott’s CardioMEMS HF System, an implantable sensor that provides an early warning of worsening heart failure, that could […]

Billed as the world’s most influential technology event, this year’s CES show saw its first ever keynote from a healthcare company when Abbott CEO Robert […]

Abbott wants to leap into the world of consumer wearables to track various health signals for better health for consumers and better growth of its […]

US medical device company Abbott has said it plans to launch a new range of ‘biowearable’ health devices aimed at consumers, in a move that […]

A recent webinar hosted by MedCity News and sponsored by BioT shared insights on some of the ingredients to successful creation and execution of remote […]

In an interview with PharmaShots, Suela Sulo, Ph.D., M.S., Director of Global Health Economics, Outcomes Research and Policy at Abbott shared her views on how […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

A study published in August found that women experienced more adverse events from Boston Scientific’s Watchman implant than men. The FDA said it plans to […]

Everest’s SPR206 Receives the NMPA’s IND Approval for the Treatment of MDR Gram-Negative Bacterial Infections Published: Sept 24, 2021 | Tags: Everest, SPR206, NMPA, IND, […]

Shots: The US FDA has approved Abbott’s Epic Plus & Epic Plus Supra Stented Tissue valves to treat patients with aortic or MV disease who […]

With the results of a recent trial Abbott is seeking an expanded indication from the FDA for its CardioMEMS device, intended to prevent hospitalization of […]

In an interview with PharmaShots, Michael Dale, Senior Vice President of the Structural Heart division at Abbott shared his views on the Navitor’s approval in […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Shots: The acquisition will accelerate Abbott’s vascular portfolio with the addition of Walk Vascular’s peripheral thrombectomy systems to treat patients with peripheral blood clots Walk […]

Shots: The head-to-head Amulet IDE study compares Amplatzer Amulet Left Atrial Appendage (LAA) Occluder with the Watchman device to treat patients with AFib at an […]

Janssen Presents Results of Rybrevant (amivantamab-vmjw) in P-I CHRYSALIS Study for Advanced NSCLC with METex14 Mutations at WCLC Published: Aug 20, 2021 | Tags: Astellas, […]

Medtronic to Acquire Intersect ENT for ~$1.1B Published: Aug 6, 2021 | Tags: Medtronic, Intersect ENT, ~$1.1B Kite Signs a License Agreement with Appia to […]

Abbott received FDA clearance for an app to pair with its Freestyle Libre 2 glucose monitor. The app lets people get readings directly on their […]

The FDA has approved an artificial intelligence-based imaging software developed by Abbott that is used to help physicians carry out procedures like implanting stents to […]

Shots: Abbott launches its latest Jot Dx ICM with new features to help clinicians for a constant flow of heart rhythm data to treat atrial […]

Neuros Medical Receives FDA Breakthrough Device Designation for its Novel Altius® High-Frequency Nerve Block System On June 29, 2021, the US Food and Drug Administration […]

BeiGene Reports Results of Tislelizumab + CT in P-III RATIONALE 309 Study as 1L Treatment for Recurrent or Metastatic Nasopharyngeal Cancer Published: May 21, 2021 […]

The medical devices industry is a fast-growing market driven by the complex needs of patients encouraging life science companies to develop innovative therapies to improve […]

Shots: Abbott has received CE Mark for Navitor making the minimally invasive device available for people in Europe with severe aortic stenosis who are at […]

In an interview with PharmaShots, Dr. Drew Falconer, Neurologist and Director at Inova Parkinson’s and Movement Disorders Center in Fairfax, Va. shares insights on the […]

Shots: Abbott’s new imaging platform powered by Ultreon 1.0 software has received a CE mark in the EU. The new imaging platform merges optical coherence […]

In an interview with PharmaShots, Allen Burton, M.D., Medical Director at Abbott Neuromodulation shared his views on the NeuroSphere myPath App which is designed to […]

PhaseBio Signs a Supply Agreement with BioVectra to Support the Development and Commercialization of Bentracimab Published: Mar 12, 2021 | Tags: PhaseBio, Signs, Supply, Agreement, […]

Shots: The coalition builds on Abbott’s excellence in virus surveillance and aid in analyzing virus samples for unknown diseases and detects mutations and variants including […]

Shots: The US FDA has approved NeuroSphere Virtual Clinic that allows a patient to communicate with a physician and remotely receive stimulation settings in real […]

Abbott announced the launch of the newly-approved NeuroSphere Virtual Clinic by which deep brain stimulation patients can have their devices programmed and reset remotely, from […]

Roche Collaborate with Cambridge Quantum Computing to Develop Quantum Algorithm for Drug Discovery and Development Published: Jan 29, 2020 | Tags: Roche, Collaborate, Cambridge Quantum Computing, […]

Shots: Abbott received a CE mark for asymptomatic testing and self-swabbing using its Panbio COVID-19 Ag test to detect SARS-CoV-2. Abbott has shipped 200M test […]

What You Should Know: – Abbott launches new app, NeuroSphere myPath that will allow patients to record their pain relief during the device trial period […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

Shots: The app will record pain relief during the device trial period with SCS or DRG therapy, simplifying reporting outcomes & connect patients to physicians […]

Sandoz to Launch Hyrimoz (biosimilar, adalimumab) in Canada Published: Dec 17, 2020 | Tags: Authorization, Biosimilar, canada, health, Humira, Hyrimoz, Launch, receives, Sandoz Amgen’s Riabni […]

Pawel Czech, Founder of Nex.D While countries around the world continue to flounder with regards to COVID-19 testing, everyone is wondering how national governments could […]

Shots: Abbott received CE Mark for its new quantitative SARS-CoV-2 IgG lab-based serology test. The test measures levels of IgG Abs to help in the […]

What You Should Know: – The digital diabetes market is on track to reach $1.5 billion dollars by 2024, according to a new report by Research2Guidance. […]

Richter Acquires Janssen’s Evra Transdermal Contraceptive Patch Assets for $263.5M Published: Dec 3, 2020 | Tags: Asset Purchase Agreement, Evra, Gedeon Richter, Janssen Pharmaceutical, Signs, […]

Shots: Abbott’s next-generation, sensor-based glucose monitoring technology, FreeStyle Libre 2, received Health Canada’s approval for adults & children with diabetes The system continuously measures glucose […]

What You Should Know: – White house coronavirus task force doubles down on rapid testing strategy to fight the coronavirus as some states say they […]

Qiagen Launches Portable Digital SARS-CoV-2 Antigen Test in the US Published: Nov 13,2020 | Tags: Antigen Test, Digital, Launches, Portable, Qiagen, SARS-CoV-2, US GSK and […]

Shots: Abbott has launched the IonicRF Generator, which is used to deliver a non-surgical, minimally invasive treatment for the management of pain in the nervous […]

AstraZeneca’s Brilinta (ticagrelor) Receives the US FDA’s Approval to Reduce the Risk of Stroke in Patients with an Acute Ischemic Stroke or High-Risk Transient Ischemic […]

Abbott recently released global research on vascular patient care, designed to shine a light on the vascular patient journey. The report called “Beyond Intervention” uncovers […]

Shots: The company has received CE mark in EU and approval in Australia for its new EnSite X EP system which offers the option to […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

Novartis’s Luxturna (voretigene neparvovec) Receives Health Canada Approval as the First Gene Replacement Therapy for Inherited Retinal Disease Published: Oct 15, 2020 | Tags: approval, […]

1. Taysha Gene Therapies’ TSHA-101 Receives the US FDA’s Orphan Drug Designation and Rare Pediatric Disease Designation for GM2 Gangliosidosis Published: Aug 27, 2020 | […]
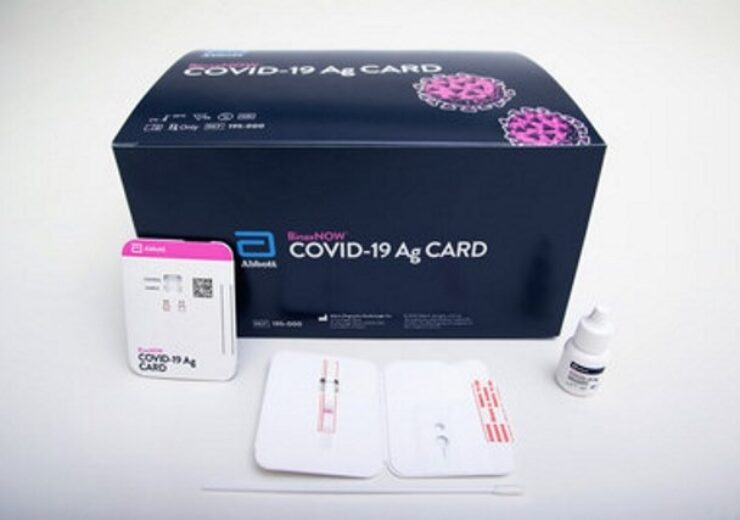
Shots: The US FDA has issued EUA for Abbott’s BinaxNOW COVID-19 Ag Card rapid test for the detection of COVID-19. The test will be available […]

Shots: The US FDA approves Abbott’s patient controller app for use on Apple’s smartphone devices, allowing patients with neurological conditions such as chronic pain or […]

Eric Stone, Co-Founder & CEO at Velano Vascular Prior to the outbreak of COVID-19, the healthcare industry in the United States was in the midst […]
Copyright © 2024 | WordPress Theme by MH Themes