
Aduhelm’s loss is a win for value-based drug pricing
Value-based pricing could have shown us with more certainty the real level of effectiveness of Aduhelm. Maybe it is worth paying something for; and value-based […]

Value-based pricing could have shown us with more certainty the real level of effectiveness of Aduhelm. Maybe it is worth paying something for; and value-based […]

Merck is paying $25 million to kick off a research partnership with Cerevance, a startup that analyzes donor brain tissue to find novel targets for […]

Acadia Pharmaceuticals drug Nuplazid failed to win FDA approval for the treatment of psychosis in Alzheimer’s disease patients. The regulator said that the data submitted […]

In May, another amyloid targeting drug for Alzheimer’s disease – AC Immune and Roche’s crenezumab – missed the mark in another clinical trial and looked […]

Memory Health CEO Edward Shehab talked about taking a preventive approach to neurodegenerative conditions.

Neurodegenerative diseases like Parkinson’s and Alzheimer’s affect millions of people worldwide, and their prevalence is only increasing, according to the World Health Organization. And while […]

AbbVie is pulling out of its collaboration with Alector on the development of Alzheimer’s disease candidate AL003, which aims to tackle the neurodegenerative disease by […]

AbbVie paid Alector $205 million up front five years ago to begin an R&D partnership on two drugs intended to bring an immunotherapy approach to […]

The FDA has started an accelerated review of Eisai and Biogen’s a new amyloid-targeting antibody lecanemab for Alzheimer’s disease, with a 6 January deadline that […]

Aniket Singh Rajput, Founder and CEO of Neuroglee Thanks to COVID-19 and numerous other factors, the adoption of digital health technologies used by patients in […]

An Alzheimer’s disease drug from Athira Pharma has failed a Phase 2 clinical trial and the biotech has a surprising theory why. Athira says standard […]

Alzheimer’s disease could be diagnosed from a single MRI brain scan, according to a new study by Imperial College London. Using machine learning technology, researchers […]

Acadia Pharma’s attempts to extend the indications of its Nuplazid drug in the US to include Alzheimer’s disease-related psychosis have suffered another major setback. Nuplazid […]

The drug, crenezumab, failed to prevent early symptoms or slow cognitive decline, the latest setback in the long quest to find effective therapies for the […]

The drug, crenezumab, failed to prevent early symptoms or slow cognitive decline, the latest setback in the long quest to find effective therapies for the […]

The drug, crenezumab, failed to prevent early symptoms or slow cognitive decline, the latest setback in the long quest to find effective therapies for the […]

The litany of failed trials of amyloid-targeting drugs for Alzheimer’s disease has added another verse, as Roche concedes that its crenezumab candidate was unable to […]

Cumulus Neuroscience has started a pair of clinical trials to gauge the value of a panel of physiological and digital biomarkers in tracking the course […]

Here we go again. With Aduhelm largely sidelined, Biogen and Eisai are hoping for a better fortunes with their next Alzheimer’s disease therapy lecanemab, which […]

The US FDA has authorised a new test that can detect amyloid plaques in early-stage Alzheimer’s disease without the use of PET scans, which expose […]

The tests could prove useful in screening patients that coud benefit from therapies being tested for Alzheimer’s. But uptake will depend, in part, on reimbursement, […]

The drug company will effectively give up on marketing Aduhelm, which comes with unproven benefits and serious safety risks.

The drug company will effectively give up on marketing Aduhelm, which comes with unproven benefits and serious safety risks.

The drug company will effectively give up on marketing Aduhelm, which comes with unproven benefits and serious safety risks.

The drug company will effectively give up on marketing Aduhelm, which comes with unproven benefits and serious safety risks.

The drug company will effectively give up on marketing Aduhelm, which comes with unproven benefit and serious safety risks.

Biogen has announced a major revamp of its business that will see chief executive Michel Vounatsos depart the company, and all commercial activities related to […]

Chicken, fish, and egg powder in processed foods present greater risk from cholesterol oxidation byproducts, but there are things you can do to reduce exposure. […]

Oxidized cholesterol can be a hundred times more toxic than regular cholesterol, raising additional concerns about foods such as ghee, canned tuna, processed meat, and […]

Biogen has given up on seeking approval for its Alzheimer’s disease therapy Aduhelm in the EU, saying that discussions with EU regulators had made it […]

Biogen withdrew an application seeking approval in Europe for Alzheimer’s disease drug Aduhelm, a move that follows a negative opinion on the therapy issued by […]

Tom Zaubler, MD, Chief Medical Officer of NeuroFlow Since the passage of the Medicare Improvements for Patients & Providers Act in 2008, the U.S. healthcare […]

Studies linked to Cassava Sciences, once a stock market favorite, have been retracted or challenged by medical journals.

New technologies for quantifying behavioral information and tracking neurological patterns have the potential to change treatment—but we must connect the data.

Biogen’s Alzheimer’s disease drug Aduhelm will only be covered for Medicare beneficiaries participating in a clinical trial. The final coverage determination will have the effect […]

The US Centres for Medicare and Medicaid Services (CMS) has firmed up its draft decision to limit reimbursement of Biogen’s Alzheimer’s disease therapy Aduhelm to […]

Officials cited data showing the new Alzheimer’s drug has serious safety risks and may not help patients.

The Alzheimer’s Association has pushed relentlessly to get broad access to Aduhelm, despite safety risks and uncertain evidence that it helps patients.

The largest-ever study of genetics in Alzheimer’s disease patients has identified 42 new genes that appear to be linked to the neurodegenerative disorder. The new […]

University College Dublin spinout Kinesis Health Technologies, which has developed a range of wearable sensors to assess the physical capabilities of patients, has been bought […]

After months of debate about the validity of its phase 3 clinical trials for Alzheimer’s therapy Aduhelm, Biogen has published the results in a peer-review […]

A group of pharma companies and academic groups have joined forces with the Digital Medicine Society (DiMe) to develop standardised, digital clinical measures that can […]

Japanese pharma Eisai has backed away from its 50:50 profit-sharing arrangement with Biogen for troubled Alzheimer’s therapy Aduhelm, handing over full responsibility to its US […]

What You Should Know: – Aetion, real-world evidence (RWE) tech and analytics provider, and Cognito Therapeutics, a pioneer in neuromodulation therapeutics for neurodegenerative diseases like Alzheimer’s, are partnering […]

With prospects of an early, accelerated approval for Roche’s Alzheimer’s disease candidate gantenerumab looking diminished, the drugmaker has launched a new four-year trial in the […]

Four years after spinning out from Belgium’s UCB, Syndesi Therapeutics and its SV2A modulator programme has been bought by AbbVie for $130 million upfront. The […]

AbbVie has acquired Syndesi Therapeutics, an early clinical neuroscience startup developing drugs that have potential applications in a range of cognitive disorders. Syndesi is based […]

Swiss digital health company MindMaze has raised $105 million to help take its digital therapeutics (DTx) for neurological diseases like stroke onto the global stage, […]

Japanese drugmaker Eisai has published comments filed with the US Centers for Medicare & Medicaid Services over its restrictive coverage proposal for beleaguered Alzheimer’s drug […]

Eli Lilly has backed away from plans to file Alzheimer’s drug donanemab in the first quarter of this year, saying the proposal to limit reimbursement […]

What evidence is there that our meat-sweet diets play a cause-and-effect role in dementia? What is behind the dramatic increase in dementia in Japan over […]

Eli Lilly has said it is seeking permission to build a new biopharmaceutical manufacturing facility in Ireland that will be used to produce active ingredients […]

Biogen and Eisai have increased the earning potential of their Alzheimer’s drug Aduhelm by expanding the post-marketing trial of the drug required by the FDA […]

The FDA placed a clinical hold on Denali Therapeutics’ application to begin Phase 1 testing of an antibody drug for Alzheimer’s disease. The Denali drug […]

Biogen’s difficult launch of controversial Alzheimer’s therapy Aduhelm has been made even harder by a proposal by Medicare to cover the drug only for certain […]

If the preliminary decision is finalized this spring, it would restrict coverage to patients in randomized clinical trials.

If the preliminary decision is finalized this spring, it would restrict coverage to patients in randomized clinical trials.

The Center for Medicare and Medicaid Services proposes covering Biogen Alzheimer’s disease drug Aduhelm only for people enrolled in a clinical trial. In explaining this […]

If the preliminary decision is finalized this spring, it would sharply limit the number of patients who use the expensive drug.

Medicare officials are trying to determine whether to cover Aduhelm, which the F.D.A. approved despite unclear evidence that it helps patients.

Biogen and Eisai head towards the end of the year with some much-needed good news in their Alzheimer’s programmes, as the FDA awards a fast-track […]

A group of Alzheimer’s experts and health advocates called on the F.D.A. to withdraw its approval of the drug, the latest of several setbacks for […]

A group of Alzheimer’s experts and health advocates called on the F.D.A. to withdraw its approval of the drug, the latest of several setbacks for […]

A group of Alzheimer’s experts and health advocates called on the F.D.A. to withdraw its approval of the drug, the latest of several setbacks for […]

A group of Alzheimer’s experts and health advocates called on the F.D.A. to withdraw its approval of the drug, the latest of several setbacks for […]

The price of Biogen Alzheimer’s disease drug Aduhelm will be cut in half at the start of 2022, a move that comes as a Medicare […]

Biogen has cut the list price of its Alzheimer’s disease therapy in half from $56,000 to $28,200 a year, before the outcome of an ongoing […]

While the era of using digital health tools to manage MCI and perhaps prevent greater cognitive decline is just beginning, it is a promising new […]

The EMA’s human medicines committee has said it cannot approve Biogen and Eisai’s Alzheimer’s disease therapy Aduhelm, saying it was unconvinced by the data submitted […]

Biogen and partner Eisai have said they will provide details next March for the confirmatory trial that will be needed to upgrade the drug’s accelerated […]

Six months after Biogen was awarded regulatory approval for its Alzheimer’s disease drug Aduhelm, the company the company is laying out a timeline for the […]

We now know how important white matter is to our overall brain health and cognitive ability, as well as how declines in white matter structure […]

Scientists in the US have found that people who use Viagra for erectile dysfunction seem to have a lower risk of developing Alzheimer’s disease – […]

GlaxoSmithKline has formed a five-year partnership with Oxford University to set up a new institute that will apply machine learning and functional genomics to the […]

Japanese drugmaker Eisai has teamed up with Fujitsu spin-out FCNT, which makes smartphones aimed at older consumers, on digital tools to support people with dementia. […]

The first patients are now being treated as part of a first-in-human trial of a vaccine, administered as a nasal spray, that aims to slow […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Newly published safety data shows that 41 percent of patients in key clinical trials of the Alzheimer’s drug experienced brain bleeding or swelling, though many […]

Bill Martin, the Global Therapeutic Area Head of Neuroscience at The Janssen Pharmaceutical Companies of Johnson & Johnson, shared some of the promising developments in […]

The drug, Aduhelm, has been mired in controversy since it was in approved in the United States in June.

The drug, Aduhelm, has been mired in controversy since it was in approved in the United States in June.

The drug, Aduhelm, has been mired in controversy since it was in approved in the United States in June.

The drug, Aduhelm, has been mired in controversy since it was in approved in the United States in June.

Biogen and Eisai have said that the EMA’s human medicines committee looks unlikely recommended approval of their Alzheimer’s drug Aduhelm when it comes up for […]

Vaxxinity, a clinical-stage biotech with vaccine technology that gets the body to produce therapeutic or protective antibodies, raised $88 million from an IPO that priced […]

Eli Lilly has followed through on its plan to file for approval of its Alzheimer’s disease candidate donanemab, and is planning to take on Biogen […]

The FDA has approved an artificial intelligence-based test for early detection of dementia that can be carried out on an iPad in five minutes. The […]

Analysts were predicting sales of Biogen’s recently approved Alzheimer’s therapy Aduhelm may start to gather a little momentum in the third quarter, but the drug […]

The drug, Aduhelm, brought in $300,000 in revenue in its first full three months of availability. The company expects the drug to generate minimal revenue […]

The drug, Aduhelm, brought in $300,000 in revenue in its first full three months of availability. The company expects the drug to generate minimal revenue […]

A software platform developed by Altoida that uses augmented reality and artificial intelligence to diagnose neurological diseases in their earliest stages is being tested in […]

A data-mining study conducted by researchers in the US has found that an already-approved diuretic drug could have potential as a treatment for some patients […]

Shots: The designation is based on the data from an ongoing SCarlet RoAD, Marguerite RoAD OLE trials along with other studies evaluating gantenerumab (anti-amyloid beta […]

Pfizer’s tyrosine kinase inhibitor Inlyta is already a cornerstone of treatment for some forms of cancer, but could have potential as a therapy for Alzheimer’s […]

Eisai is seeking accelerated FDA approval of lecanemab, an Alzheimer’s disease drug that is part of a research alliance with Biogen. The drug is following […]

Biogen and Eisai’s first Alzheimer’s disease drug Aduhelm is facing an uphill battle in the US market, but the partners are forging ahead with their […]

Shares in Biogen have come under pressure after chief executive Michel Vounatsos admitted that the company is facing a much slower than anticipated rollout of […]

A digital health company focusing on neurodegenerative disorders – Neuroglee Therapeutics – has raised $10 million in first-round funding to roll out a virtual neurology […]

The agency approved Biogen’s Aduhelm “despite concerns raised by experts,” committee leaders said in a letter.

The agency approved Biogen’s Aduhelm “despite concerns raised by experts,” committee leaders said in a letter.

The agency approved Biogen’s Aduhelm “despite concerns raised by experts,” committee leaders said in a letter.

Shots: The P-II Lauriet study evaluates the safety & efficacy of semorinemab (anti-tau mAb) vs PBO in 272 adult patients with AD across 43 centers […]

Shares in AC Immune leaped today after the company said its tau-targeting Alzheimer’s disease candidate semorinemab hit one of its objectives in a phase 2 […]

Test your knowledge of this week’s health news.

Test your knowledge of this week’s health news.

For an increasingly important set of drugs, Medicare has been driving up prices. It knows how to do better.

For an increasingly important set of drugs, Medicare has been driving up prices. It knows how to do better.

Health insurer Excellus BlueCross BlueShield has said it will not cover treatment with Biogen and Eisai’s new Alzheimer’s disease drug Aduhelm because it has not […]

Researchers at Cambridge University in the UK are trialling an artificial intelligence system that they think could spot the signs of dementia after a single […]

The FDA’s approval of Biogen’s Aduhelm for Alzheimer’s disease emboldened Eli Lilly to move forward plans to file its own candidate donanemab later this year, […]

Patients taking new Alzheimer’s disease drug Aduhelm will be evaluated in a real-world study lasting up to five years. Biogen announced plans for the observational […]

With a string of new drugs for Alzheimer’s disease making their way through late-stage clinical testing, the demand for suitable subjects to enrol into trials […]

Biogen and partner Eisai have said they will run a large-scale observational study of Alzheimer’s drug Aduhelm in the US to build evidence for the […]

Biogen has reported $2m in sales for newly-launched Alzheimer’s therapy Aduhelm that came in below its expectations, prompting the company to issue a rebuke to […]

The drug from Biogen, called Aduhelm, is expected to generate billions in sales. It could also strain Medicare’s budget within a few years.

Merck and Evidation Health launched a study to see if data from smartphones and wearables could be used to detect and monitor the treatment of […]

The drug from Biogen, called Aduhelm, is expected to generate billions in sales. It could also strain Medicare’s budget within a few years.

Phase 3 results with Roche’s amyloid-targeting Alzheimer’s drug gantenerumab may not be due until the second half of last year, but the company is already […]

Though some of its own senior officials said there was little evidence of benefit for patients, the F.D.A. nonetheless greenlighted Biogen’s Aduhelm, or aducanumab.

Though some of its own senior officials said there was little evidence of benefit for patients, the F.D.A. nonetheless greenlighted Biogen’s Aduhelm, or aducanumab.

The major medical center’s rejection of the new Alzheimer’s drug is one of the starkest signs of concern over its effectiveness.

Three FDA figures at the centre of the fallout over the approval Biogen and Eisai’s Alzheimer’s disease therapy Aduhelm have defended their actions, arguing that […]

CMS plans to review and determine coverage for Biogen’s Aduhelm, the first new Alzheimer’s disease treatment in decades. But the jury is still out on […]

Diagnosing Alzheimer’s disease is still a challenge, particularly in its earliest stages, but a new study suggests that subtle changes in behaviour whilst driving could […]

The US government has started a review of how Biogen and Eisai’s pricey new Alzheimer’s disease therapy Aduhelm will be covered by Medicare, to see […]

In another twist to the Aduhelm approval tale, FDA’s Janet Woodcock has called for an independent investigation into the relationship between agency staffers and Biogen […]

And how to learn more about the issue dividing school districts across the country.

The agency’s acting head said a review should look into whether any interactions between agency staff and the drug developer, Biogen, broke F.D.A. rules.

One of the criticisms levelled at the FDA over its approval of Biogen and Eisai’s Alzheimer’s disease drug Aduhelm was its decision to clear use […]

The approval of Aduhelm to treat Alzheimer’s disease has raised hope among older adults, but many doctors wonder if it is warranted.

The agency faced criticism for approving Aduhelm for all Alzheimer’s patients. Now it recommends that the drug be given only to those with mild symptoms.

The agency faced criticism for approving Aduhelm for all Alzheimer’s patients. The new label recommends that the drug be given only to patients with mild […]

The approval of Aduhelm to treat Alzheimer’s disease has raised hope among older adults, but many doctors wonder if it is warranted.

The F.D.A. shocked the world of medicine when it approved a much-debated drug for Alzheimer’s. What does the decision mean for patients and their families?

The F.D.A. shocked the world of medicine when it approved a much-debated drug for the disease. What does the decision mean for patients and their […]

GlaxoSmithKline is paying Alector $700 million up front to share in the development of the biotech’s two lead drugs, which offer potentially broad application in […]

I would like to recommend this piece at Stat about the details of the Biogen aducanumab approval. It’s behind the paywall, but it’s a deeply […]

Acumen Pharmaceuticals’ IPO raised $160 million to fund clinical development of its Alzheimer’s disease drug candidate. Though that drug goes after the same target as […]

The Institute for Clinical and Economic Review (ICER) was very damning of Biogen’s new Alzheimer’s disease therapy Aduhelm when it issued its first report on […]

The Institute for Clinical and Economic Review (ICER) was very damning of Biogen’s new Alzheimer’s disease therapy Aduhelm when it issued its first report on […]

Even those who supported the F.D.A.’s approval of the controversial new drug said authorizing it for anyone with Alzheimer’s disease was much too broad.

If you have occasion to study neurodegeneration, you will be struck by how many terrible high-profile diseases in this area seem to share a common […]

It should be obvious, given previous posts here, that I think that the FDA approval of Biogen’s aducanumab for Alzheimer’s was a mistake. It is […]

Biogen has four other Alzheimer’s candidates in its clinical pipeline behind recently-approved Aduhelm, but one of them – anti-tau antibody gosuranemab – has just been […]

With the first patient set to receive Biogen’s new Alzheimer’s drug Aduhelm in Rhode Island today, US consumer advocacy organisation Public Citizen is deeply unhappy […]

The F.D.A.’s approval of the Alzheimer’s drug aducanumab is inexplicable.

Date: July 12, 2021 Issue #: 1628 Summary: Aducanumab-avwa (Aduhelm – Biogen/Eisai), an IV amyloid beta-directed monoclonal antibody, has received accelerated approval from the FDA […]

The F.D.A.’s approval of the Alzheimer’s drug aducanumab is inexplicable.

The fallout over the FDA’s decision to approved Biogen’s Alzheimer’s therapy Aduhelm continues to be felt, with a third member of its advisory committee resigning […]

Despite scant evidence that it works, the drug, Aduhelm, is predicted to generate billions of dollars in revenue, much of it from Medicare.

The drug, Aduhelm, a monthly infusion priced at $56,000 per year, was approved this week despite weak evidence that it helps patients.

The drug, Aduhelm, a monthly infusion priced at $56,000 per year, was approved this week despite weak evidence that it helps patients.

Biogen is still discussing with the FDA the clinical trial design to confirm the benefit offered by its newly approved Alzheimer’s disease drug, Aduhelm. According […]

As the world knows, the FDA approved Biogen’s anti-amyloid antibody today, surely the first marketed drug whose Phase III trial was stopped for futility. I […]

Aducanumab, or Aduhelm, is the first new Alzheimer’s treatment in 18 years and the first to attack the disease process. But some experts say there’s […]

The FDA “has failed in its responsibility to protect patients and families from unproven treatments with known harms” in approving Biogen’s Alzheimer’s disease drug Aduhelm. […]

Despite scant evidence that it works, the drug, Aduhelm, is predicted to generate billions of dollars in revenue, much of it from Medicare.

A Biogen Alzheimer’s drug that clears amyloid plaque from the brain has been granted accelerated approved by the FDA. The drug, which will be marketed […]

Aducanumab, or Aduhelm, is the first new Alzheimer’s treatment in 18 years and the first to attack the disease process. But some prominent experts say […]

Biogen and Eisai have defied the naysayers and secured an historic FDA approval for their amyloid-targeting antibody aducanumab as the first-ever disease-modifying treatment for Alzheimer’s […]

Aducanumab is the first new Alzheimer’s treatment in 18 years and the first to attack the disease process. But some prominent experts say there’s not […]

An F.D.A. sign-off for aducanumab would make finding a good treatment harder.

If the agency approves it, aducanumab would be the first new Alzheimer’s treatment since 2003. Patients are desperate for new options, but some scientists say […]

If the agency approves it, aducanumab would be the first new Alzheimer’s treatment since 2003. Patients are desperate for new options, but some scientists say […]

If the agency approves it, aducanumab would be the first new Alzheimer’s treatment since 2003. Patients are desperate for new options, but some scientists say […]

There’s a big FDA decision coming up in the next few days: whether or not to approve the Biogen antibody for Alzheimer’s (aducanumab). I’ve had […]

The spice saffron is pitted head-to-head against the leading drug for severe Alzheimer’s disease. What’s the latest on treating memory disorders with the spice saffron? […]

An F.D.A. sign-off for aducanumab would make finding a good treatment harder.

An F.D.A. sign-off for aducanumab would make finding a good treatment harder.

An F.D.A. sign-off for aducanumab would make finding a good treatment harder.

Exscientia has advanced a drug candidate for Alzheimer’s disease into human testing, the third clinical-stage project for the UK specialist in artificial intelligence (AI) based […]

The neuroQWERTY technology can analyse people’s typing for signs of motor conditions like Parkinson’s disease – and now the team behind it is looking to […]

Standard cost-effectiveness measures the value of a treatment based on the cost for an additional unit of health where units of health are measured in […]

What happened when turmeric curcumin was put to the test to see if it could reverse DNA damage caused by arsenic exposure? Arsenic is a […]

That’s a post title that I could have used eight or ten times over the lifetime of this blog – Eli Lilly has been hammering […]

Digital health startup Cognoa shared results of a Phase 2 trial of its device, intended to treat Alzheimer’s disease using gamma frequency light and sound. […]

With so many novel drug candidates for Alzheimer’s disease failing in clinical development, researchers in the US have started using artificial intelligence (AI) to screen […]

CNS disorders are a wide-ranging set of disorders in which the brain loses its normal functioning, limiting everyday ability to function. These may arise from […]

Shots: The alliance will investigate the effectiveness of LPC-bound EPA/DHA in preventing cognitive decline linked to AD. Aker BioMarine will supply the research team with […]

Medical charity Alzheimer’s Research UK has teamed up with artificial intelligence specialist Exscientia to find new drug treatments for the devastating neurodegenerative disease. The alliance […]

Shares in Swiss biotech AC Immune have risen sharply after it said a vaccine in development for Alzheimer’s disease (AD) hit the mark in a […]

Hot on the heels, Aurinia launches its drug in the Lupus market after GSK Aurinia Pharmaceuticals has recently got the USFDA approval for its Lupkynis […]

Biohaven’s Troriluzole Dwindles Again In Alzheimer’s After Anxiety Biohaven Pharmaceuticals had put too much faith in its third-generation prodrug, Troriluzole. The company has tested the […]

When Eli Lilly reported results for donanemab in Alzheimer’s disease earlier this week it was hailed as a rare win for the amyloid hypothesis, although […]

Participants with the disease still declined, but much more slowly than those receiving a placebo, investigators say.

Participants with the disease still declined, but much more slowly than those receiving a placebo, investigators say.

Participants with the disease still declined, but much more slowly than those receiving a placebo, investigators say.

Jenifer Leaf Jaeger, MD, MPH, Senior Medical Director, HealthEC Senior isolation is a health risk that affects at least a quarter of seniors over 65. […]

The FDA may have been minded to approve Biogen and Eisai’s Alzheimer’s candidate aducanumab, but its clinical advisors have little doubt that the evidence for […]

The nonbinding panel recommendation to reject the drug goes against what FDA scientists presented but they throw up another obstacle for the drug, which is […]

Regulatory reviews of Biogen’s Alzheimer’s drug aducanumab are now ongoing on both sides of the Atlantic, but debate is still ongoing about whether the data […]

Improving cognitive performance with aromatherapy in young, healthy volunteers is one thing, but how about where it really matters? As I discuss in my video […]
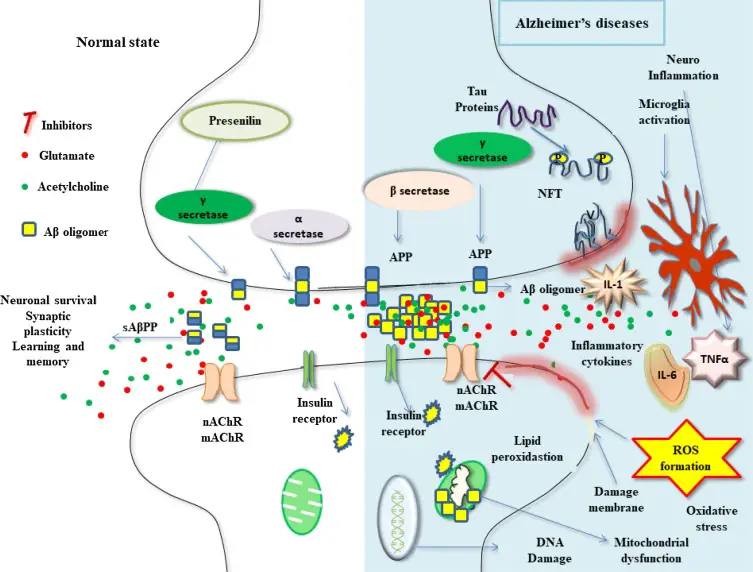
Recent Advances in Alzheimer’s Disease Prevention A prominent neurologist reviews what is known (and not known) about recent evidence-based advances in Alzheimer’s Disease prevention. The […]

Biogen and Eisai said the FDA had accepted and given priority review to their application for aducanumab. The drug will undergo an advisory committee meeting […]

Roche already has one Alzheimer’s candidate in its pipeline targeting tau, but has added a second via a deal with Belgian drugmaker UCB worth up […]

Shots: UCB to receive $120M and is eligible to receive $2B as cost reimbursement, development, and commercial milestones as well as royalties on sales of […]
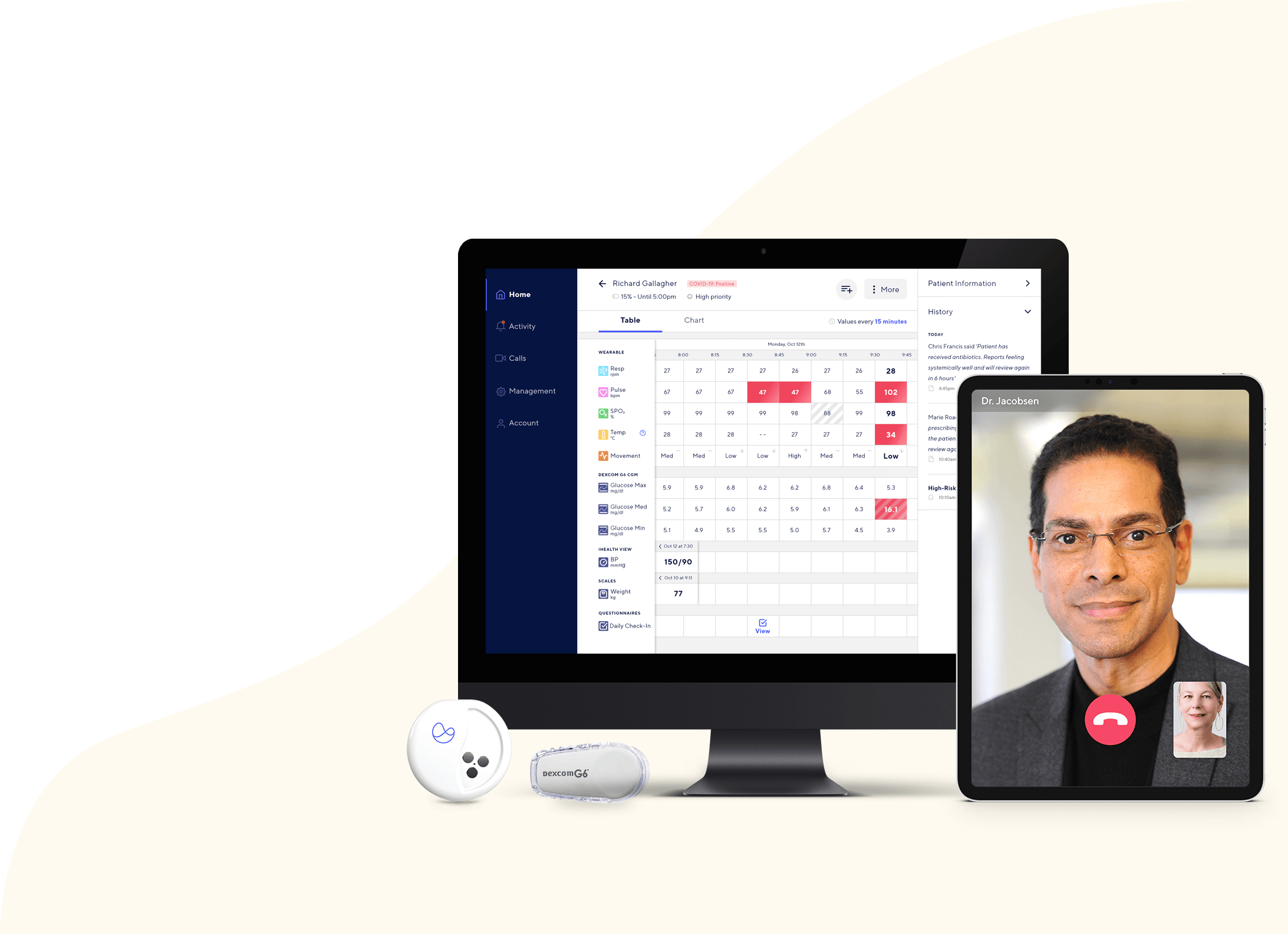
What You Should Know: – Current Health has partnered with Dexcom to add continuous glucose monitoring (CGM) capabilities to its remote patient monitoring (RPM) platform […]

The FDA has 60 days to decide whether or not to accept the application. An analyst wrote that the agency accepting it with a priority […]
Having hypertension in midlife (ages 40 through 60) is associated with elevated risk of cognitive impairment and Alzheimer’s dementia later in life, even more so […]
Copyright © 2024 | WordPress Theme by MH Themes