
Aetion completes $82M Series B round with $19M extension
The healthcare analytics company said it had raised the extension funding from three new investors and would use it to accelerate development of its real-world […]

The healthcare analytics company said it had raised the extension funding from three new investors and would use it to accelerate development of its real-world […]

In an interview, Peter Meath, J.P. Morgan’s co-head of Healthcare and Life Sciences, Middle Market Banking & Specialized Industries, says the Covid-19 pandemic has cast […]

The FDA commissioner took to Twitter Monday night to defend his performance at the agency, which came under sharp criticism amid accusations that the authorization […]

The biotech company reached a deal with the European Commission to supply an initial 80 million doses, while European Union member states will have the […]

While the emergency use authorization granted Sunday was not seen as scientifically unwarranted, it drew concerns from some experts that it could hinder enrollment in […]

Over the weekend, Trump called the agency, without evidence, part of a “deep state” that was slow-walking vaccines and drugs for Covid-19. The next day, […]
The companies also said their Phase II/III study, slated to enroll about 30,000 participants, had already dosed more than 11,000.

The clinical trial had already been placed on hold in connection with the two prior patient deaths, which were linked to the higher dose of […]
The New York Times reported that the FDA was prepared to issue an emergency use authorization for the treatment last week, but NIH Director Francis […]

The drugmaker emphasized the full global rights it would acquire to Momenta’s lead asset, nipocalimab, which it is developing for several autoantibody-driven diseases and that […]

In a CRL issued for valoctocogene roxaparvovec, the FDA told BioMarin that differences between its Phase I/II and Phase III studies limited the ability to […]

While there had been some risks to filgotinib’s approval cited before, analysts expressed surprise at the decision, with one writing it would likely delay approval […]
The deal brings Roche’s considerable manufacturing heft to Regeneron’s efforts to develop the two-antibody cocktail, REGN-COV2. Regeneron will distribute the drug in the U.S., while […]

The update to the partnership includes opt-in rights for Gilead on 15 drug targets, up from five under the original 2018 deal. Gilead will pay […]

Nearly all of the states, plus five territories and the District of Columbia, are seeking an aggregate of $2.156 trillion from the maker of the […]

Pleiogenix is a biopharma startup developing treatments for liver diseases as well as Covid-19 and non-alcoholic steatohepatitis (NASH). It recently emerged as the winner of the […]
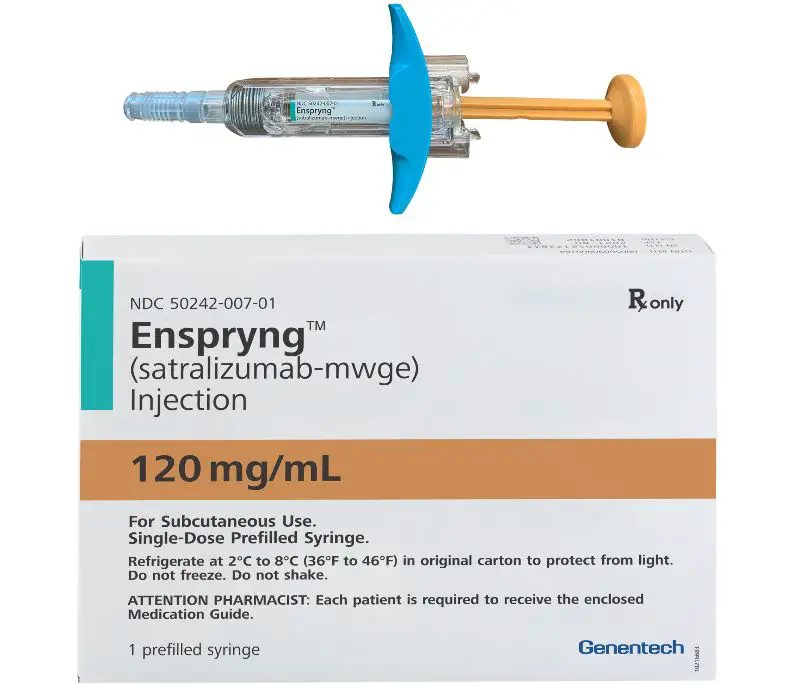
The agency approved Enspryng for the disease, which is often misdiagnosed as multiple sclerosis and can cause blindness, muscle weakness and paralysis.

The French drugmaker will take full control of SAR442168, a BTK inhibitor developed for multiple sclerosis that it in-licensed from the California biotech company in […]

Earlier this month, Ohio Gov. Mike DeWine received a false-positive result from a rapid antigen test for Covid-19, raising concerns about the tests’ accuracy. The […]

The company plans to train 250 new racially and ethnically diverse clinical trial investigators and extend the reach of its trials to underserved rural and […]
UnitedHealthcare sent a notice to plan members stating that it would no longer cover Descovy to prevent HIV, while Truvada – after it goes generic […]
In a commentary, physicians and pharmacists wrote that percentages of Black, Latino and Native American patients in two Phase III studies of the drug that […]

The company’s software system is designed to streamline the work done by staff at clinical trial sites, such as manually copying information about patient recruitment […]
The agreement, which includes 100 million doses of mRNA-1273 and an option to acquire 400 million more, brings the total amount of money the biotech […]

KaNDy’s lead product candidate, NT-814, is a non-hormonal treatment for symptoms of menopause. The drug completed Phase IIb testing and is slated to start Phase […]
Russian President Vladimir Putin said a vaccine developed at a Moscow research institute that appears to still be in Phase I development among fewer than […]

Analysts noted that Evrysdi would be priced significantly lower than Biogen’s Spinraza or Novartis’ Zolgensma. While this will provide a market advantage, it would also […]

In a phone interview, Guardant Health CEO Helmy Eltoukhy said he expected the approval to encourage coverage by those payers still on the sidelines.

News from innovative healthcare startups from Specifica, Aerovate Therapeutics and more.
Gilead Sciences had said in its second-quarter earnings that it would partner with companies in North America, Europe and Asia to manufacture the antiviral drug, […]

Biogen and Eisai said the FDA had accepted and given priority review to their application for aducanumab. The drug will undergo an advisory committee meeting […]
Blenrep, an antibody-drug conjugate, is the first drug approved for multiple myeloma that targets the antigen BCMA. Several other companies are also developing BCMA-targeting therapies […]
As we look forward to the back half of the year, it’s clear that the desire for companies to commence their IPO process with organizational […]
The drug has consistently failed to show a benefit when tested in randomized, controlled clinical trials. Social science may explain why laypeople and doctors alike […]
The company said the study of Kymriah in follicular lymphoma met its primary endpoint, though it did not disclose the trial data. It plans to […]

Under the agreement with BARDA and the Department of Defense, the drugmaker would supply 100 million doses of its vaccine, for which it published preclinical […]

With this new cash infusion, Sight Diagnostics, which uses machine learning and AI to analyze images of blood samples, has raised a total of $124 […]
Hanmi will retain options for the drug, efinopegdutide, in Korea under the deal, in which Merck is paying the Seoul-based company $10 million upfront and […]

Amplified Sciences emerged as the winner of the diagnostics track for the MedCity INVEST conference’s Pitch Perfect contest with PanCystPro, the test it is developing […]

Three of the companies making drugs used in the Phase II I-SPY COVID-19 study – Amgen, AbbVie and Takeda – announced the patient enrollments Monday. […]
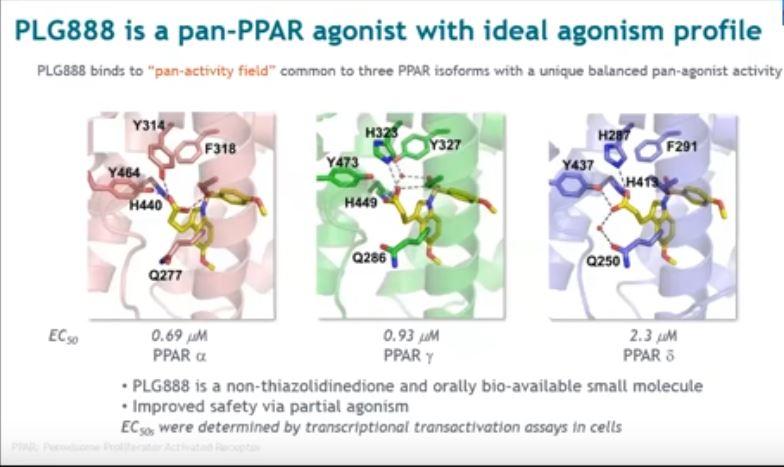
The company is hoping to close its seed funding round in the next six months and aims to be in a position to seek regulatory […]
The FDA approved Monjuvi for second-line diffuse large B-cell lymphoma. Though the drug has the same antigen target as approved CAR-T cell therapies, it is […]

Financial terms of the deal were not disclosed, but it follows a $1.95 billion contract between the two companies and the U.S. government. Assuming clinical […]
The companies will provide 100 million doses of their jointly developed vaccine. But with a Phase I/II study planned in September, it is behind in […]

While many of us have avoided exposure to Covid-19 by sheltering in place, patients with […]
The company said in its 2Q earnings that it would halt development of ALXN2040 for C3 glomerulopathy, or C3G, citing the drug’s lack of potency. […]
The companies’ resubmission of their application Wednesday seeking approval for idecabtagene vicleucel was in line with the timeline they provided in May, when the FDA […]

The company said that the COVACTA trial of Actemra in Covid-19 pneumonia had failed to meet its primary endpoint. Earlier this month, Regeneron and Sanofi […]
The study design is still being finalized, and the company is thus not yet divulging too many details. However, CEO David Daly said it would […]
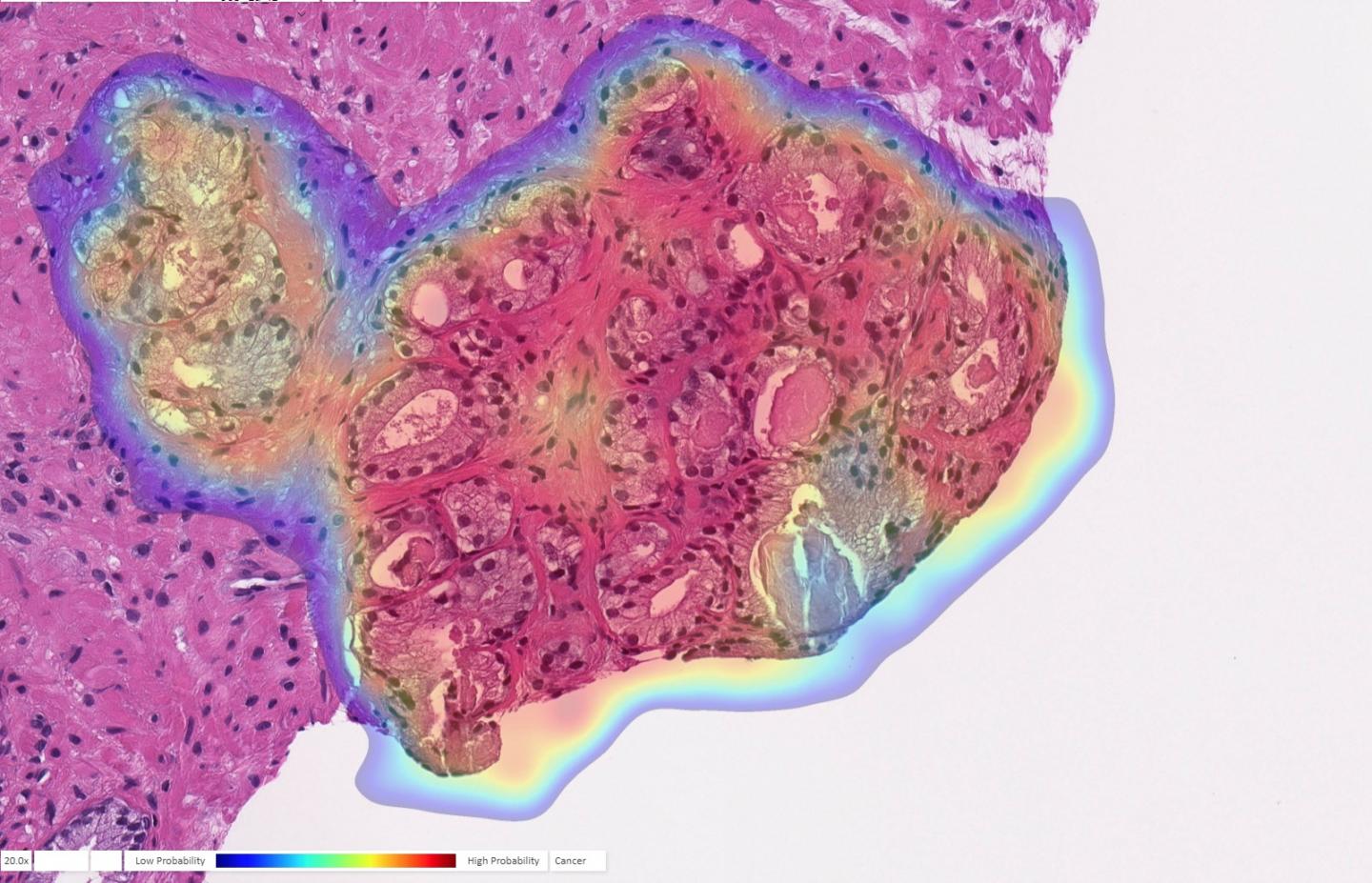
Tel Aviv-based Ibex Medical Analytics published data on what it called the first algorithm to go beyond detection and into areas like tumor grading and […]
The companies said they had started a Phase II/III trial BNT162b2. The news comes the day after Moderna announced the launch of its Phase III […]

The companies will share development and commercialization of DS-1062 in solid tumors. They already have a similar deal in place, the $6.9 billion agreement signed […]
The company had received $483 million from BARDA in April to fund the development of the vaccine, mRNA-1273, through Phase III, but that amount was […]

The study adds to the growing body of evidence that the drug, promoted early in the pandemic by President Trump, is ineffective, despite its getting […]
The agency approved Tecartus, previously developed under the name KTE-X19, as the first CAR-T therapy for mantle cell lymphoma. The company had previously won approval […]

A successful cocktail would also serve us in the long-run to treat the impacts of Covid-19 and keep those who are most vulnerable, the elderly […]
The company said it was formally terminating the Phase III study of the drug elafibranor in NASH and refocus its development of the drug on […]

The company said the CareStart Covid-19 MDx RT-PCR test is designed to detect RNA from the SARS-CoV-2 virus and produce results within 83 minutes.

The companies will test Biofourmis’ sensor and artificial intelligence system in a clinical trial of more than 120 women with endometriosis.

In an interview, TriNet’s Vice President of Life Sciences, Maria Abouseif, talked about the role Professional Employer Organizations play in helping life science companies navigate […]
The company plans to enroll 60 patients who have received its drug, Spinraza, following suboptimal response to Novartis’ Zolgensma, which was approved last year. The […]

The company said that patients receiving the drug, SNG001, had a 79% lower risk of requiring ventilation or dying compared with those on placebo, sending […]
The company announced the publication of interim data from the Phase I/II study of a vaccine it is developing with the University of Oxford. The […]

Amid fears of a premature emergency use authorization for a Covid-19 vaccine granted for political reasons, the guidance that the FDA released at the beginning […]
A joint report by cybersecurity agencies in the U.K. and Canada, endorsed by their U.S. counterparts, states that the hacker group Cozy Bear had been […]

The company, whose drug-discovery efforts are focused on protein motion and detection of interactions that occur anywhere on a protein rather than only at active […]
Opioid abuse is a widely recognized public health crisis in the United States and the […]

Evaluation of durability is still under evaluation, but the responses to mRNA-1273 show neutralizing antibody titers multiple times higher than those of recovered patients. Additionally, […]
The company is developing biosensors, initially for NASH, that could be used in place of liver biopsy and have applicability in development of drugs for […]
The Oncologic Drugs Advisory Committee voted 12-0 that the benefit-risk profile of belantamab mafodotin supported the drug. The vote came despite concerns expressed by the […]

Under the deal, Blueprint will additionally be eligible for more than $900M in milestone payments plus royalties for the RET inhibitor pralsetinib. The deal includes […]
The companies anticipate releasing more clinical data from the BNT162 messenger RNA vaccine program and potentially starting a 30,000-participant Phase IIb/III study this month.

The study is enrolling 60 healthy volunteers aged 18-45, but the company hopes it will form the basis of trials of the drug as an […]

The FDA’s Oncologic Drugs Advisory Committee will convene Tuesday to discuss the drug, belantamab mafodotin. Despite the drug’s first-in-class potential, the briefing document for the […]

Research on the drug’s molecular target dates back to the 1950s. A spokesperson for Gilead noted that the company anticipates potentially investing more than $1 […]

The company said it is on track to start its 30,000-participant Phase III trial this month and has finished manufacturing enough supplies of the vaccine […]

Bari Kowal, vice president of global clinical operations at Regeneron Pharmaceuticals, gives an update on the clinical trials ongoing to develop drugs to fight Covid-19 […]

The company is currently running a Phase I study of its lead candidate, the antibody-drug conjugate VLS-101, in patients with lymphomas and leukemias. It has […]

The FDA has 60 days to decide whether or not to accept the application. An analyst wrote that the agency accepting it with a priority […]

The company said Tuesday that a final analysis of its Phase IIb/III study of cabotegravir showed greater efficacy in HIV prevention than Gilead’s Truvada. The […]

The patient, who had been enrolled in the Phase I study at the higher dose level of UCARTCS1A, suffered a cardiac arrest, the causes of […]

The contract covers manufacture and supply of the two-antibody drug cocktail REGN-COV2. The company had announced Monday that it entered the drug into three late-stage […]

The company is running a Phase I/II study that started in May in Australia and plans to open a Phase III study of its vaccine […]

The company also said that the Phase III study of the autoimmune disease drug Kevzara that it was running with Sanofi failed to meet either […]
Copyright © 2024 | WordPress Theme by MH Themes