
BMS cancer drug’s expanded approval gives it an edge over a Gilead cell therapy
The new FDA approval for Breyanzi moves the Bristol Myers Squibb cancer cell therapy into an earlier line of treatment for an aggressive type of […]

The new FDA approval for Breyanzi moves the Bristol Myers Squibb cancer cell therapy into an earlier line of treatment for an aggressive type of […]

As an oncologist, Kristen Hege first encountered cancer cell therapy research in the mid-1990s. Now as a Bristol Myers Squibb executive, she oversees efforts to […]

CAR T cell therapies for cancer still pose challenges in manufacturing, safety, and the ability to address solid tumors. A panel at the World Medical […]

With the undeniable excitement in the BCMA field over the past few months, the BCMA Targeted Therapies Summit returns to the space to equip you […]

Approval of the Johnson & Johnson and Legend Biotech cell therapy, Carvykti, marks the second CAR T-cell therapy to clear the regulatory bar for multiple […]

Clinical-stage Arcellx raised $123.8 million from its IPO, which the company will use to advance to a pivotal test for its lead program, a CAR […]

Despite the targeted approach of some cancer treatments, tumors can find ways to escape, leading patients to relapse. ImmPACT Bio will apply its Series B […]

The cell therapy field is showing no signs of slowing down, with huge investment and excitement around next generation products and their clinical validation in solid tumour […]

A phase 1 trial of Allogene’s of-the-shelf CAR-T for lymphoma has been placed on hold by the FDA, after a “chromosomal abnormality” was seen in […]

In 2018, the former leadership of team from Kite Pharma founded Allogene, on a mission to outdo the CAR-T cancer cell therapies they had just […]

Dr. Anne Kerber, VP, Head of Clinical Development, Europe at Kite, a Gilead Company, explains how the company works with researchers and physicians across Europe […]

The FDA has approved Bristol-Myers Squibb’s Abecma (idecabtagene vicleucel) for multiple myeloma, the first cell therapy to treat the disease. Abecma’s review was delayed after […]

Lymphoma Action is the only UK health charity dedicated to people affected by lymphoma. Chief Executive Ropinder Gill and Director of Operations and External Affairs […]

NHS patients in England will be among the first in the world to receive Gilead’s Tecartus cancer cell therapy for certain types of lymphoma, after […]

Louis van de Wiel, Vice President, Site Head EU Manufacturing, Kite, a Gilead Company, reveals the complexity that sits behind the process of individualised cell […]

Accelerate the Bench to Bedside Development of Novel CAR Immunotherapies for Safe, Effective & Affordable Advanced Therapies With CAR-TCR therapies on the cusp of achieving […]
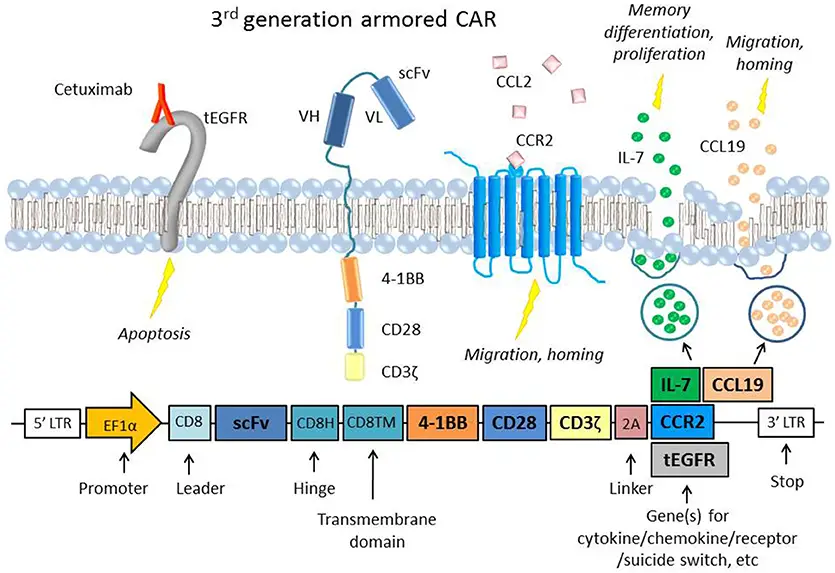
The findings may make it possible to identify a subset of patients who experience poorer outcomes or more serious side effects, especially neurotoxicity.
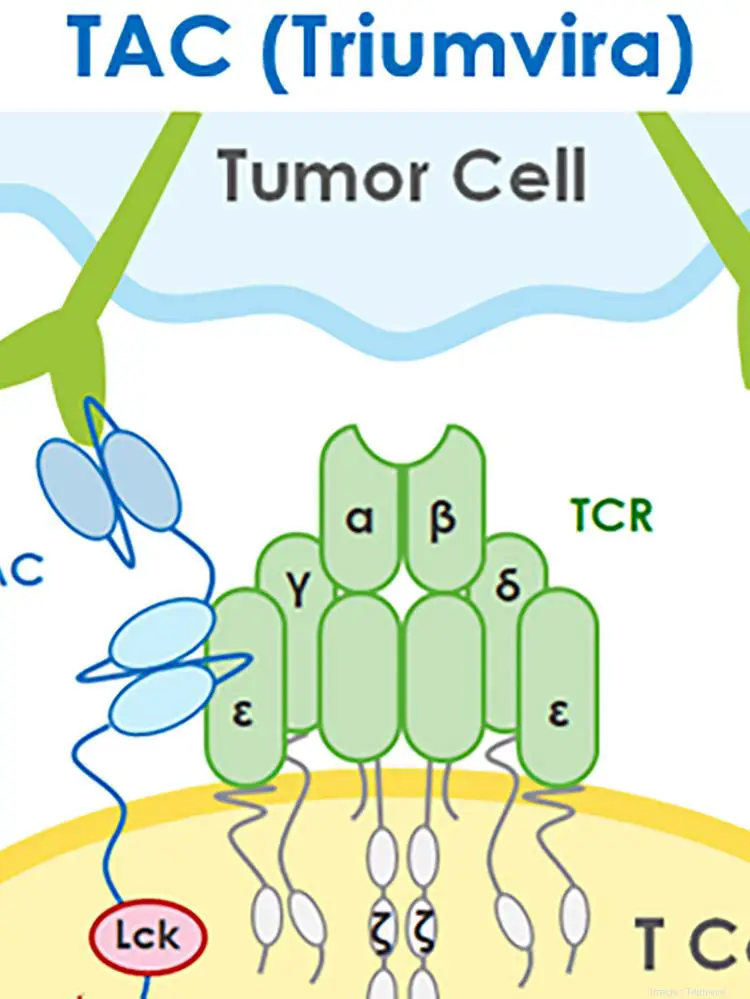
The company hopes to use the money to bring its T-cell antigen coupler cell therapy technology – currently in preclinical development – into human clinical […]
The company said the study of Kymriah in follicular lymphoma met its primary endpoint, though it did not disclose the trial data. It plans to […]
Cell and gene therapies offer some of the most groundbreaking advancements in patient care the pharma industry has ever seen. However, to fully realise the […]
The companies’ resubmission of their application Wednesday seeking approval for idecabtagene vicleucel was in line with the timeline they provided in May, when the FDA […]
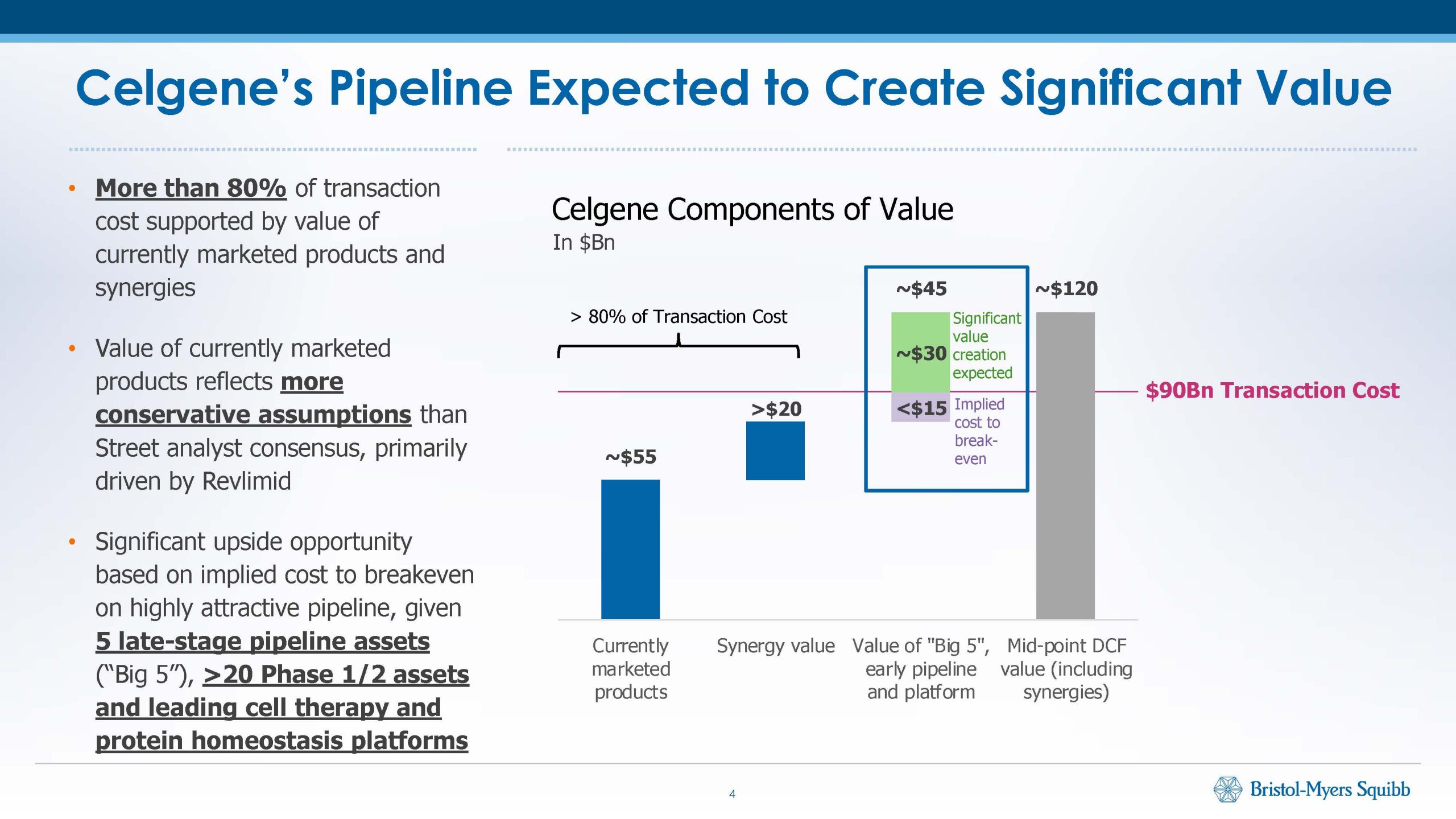
Bristol-Myers Squibb and bluebird bio have refiled their CAR-T therapy for multiple myeloma, leaving the outcome of a three drug “bet” with former Celgene shareholders […]
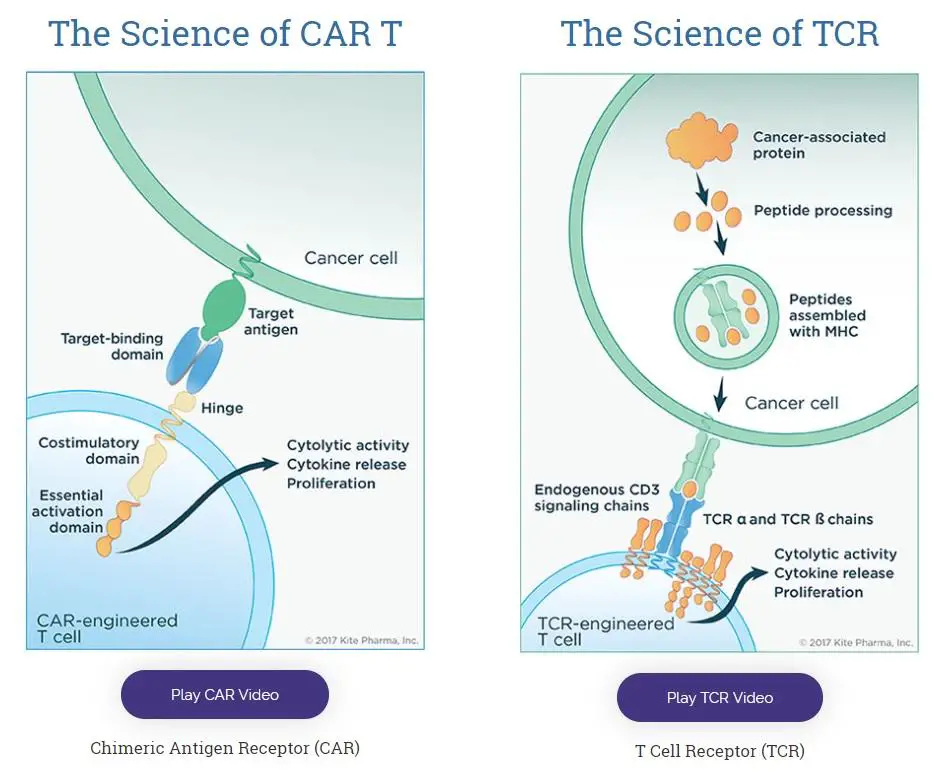
Gilead’s $11.9 billion purchase of Kite Pharma in 2017 didn’t follow the script, with two big write-downs in the value of the asset since then, […]
The agency approved Tecartus, previously developed under the name KTE-X19, as the first CAR-T therapy for mantle cell lymphoma. The company had previously won approval […]

The patient, who had been enrolled in the Phase I study at the higher dose level of UCARTCS1A, suffered a cardiac arrest, the causes of […]
Copyright © 2024 | WordPress Theme by MH Themes