
FDA okays ViiV’s Apretude as first injectable for HIV PrEP
GlaxoSmithKline’ majority-owned HIV unit ViiV has scored a key FDA approval, getting approval for its long-acting drug cabotegravir as the first injectable for pre-exposure prophylaxis […]

GlaxoSmithKline’ majority-owned HIV unit ViiV has scored a key FDA approval, getting approval for its long-acting drug cabotegravir as the first injectable for pre-exposure prophylaxis […]

A digital pill that records when it is taken has been shown to be highly effective at helping patients adhere to pre-exposure prophylaxis (PrEP) treatment […]

Merck & Co has halted enrolment in a pair of phase 3 trials of its experimental HIV drug islatravir, raising another warning flag about the […]

Gilead Sciences has the data it was hoping for in a pivotal trial of lenacapavir, an HIV drug that only needs to be dosed by […]
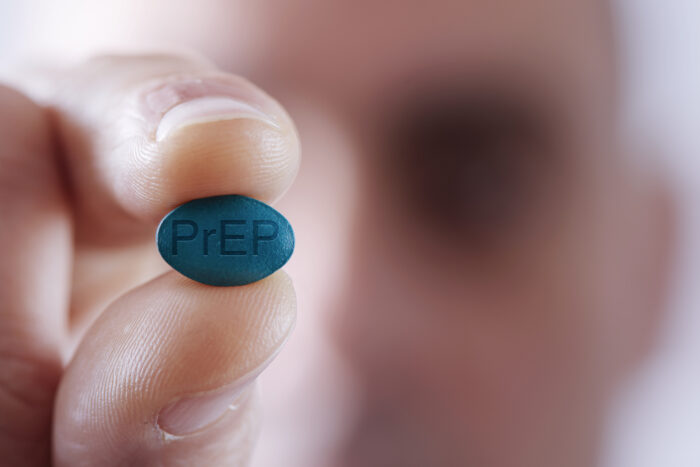
UnitedHealthcare sent a notice to plan members stating that it would no longer cover Descovy to prevent HIV, while Truvada – after it goes generic […]

The company said Tuesday that a final analysis of its Phase IIb/III study of cabotegravir showed greater efficacy in HIV prevention than Gilead’s Truvada. The […]
Copyright © 2024 | WordPress Theme by MH Themes