
Bayer bags swift OK for Nubeqa in metastatic prostate cancer
Bayer has won a key FDA approval to extend the use of its prostate cancer therapy Nubeqa, as it tries to claim market share from […]

Bayer has won a key FDA approval to extend the use of its prostate cancer therapy Nubeqa, as it tries to claim market share from […]

The largest pharma companies have all set themselves ambitious targets in recent years to improve representation for women and minority communities in their businesses, but […]

Patients with end-stage renal disease (ESRD) who are on dialysis often need to be treated with anticoagulants to prevent blood clots, but as a result, […]

Bayer’s venerable testosterone replacement product for men with hypogonadism – Nebido – is being sold to Grünenthal as the group continues a revamp of its […]

On the heels of approvals in the US, Europe and Japan, Bayer’s Kerendia for chronic kidney disease (CKD) associated with type 2 diabetes has been […]

Bayer has stepped up the digital capabilities of its radiology business with the launch of artificial intelligence-based apps for X-ray, MRI and CT imaging. The […]

In February, Atara Biotherapeutics paused enrolment into a trial of a CAR-T cell therapy for mesothelioma after a patient death. Now, its partner Bayer has […]

Aspen Neurosciences is developing a cell therapy that uses a patient’s own stem cells to develop a personalized treatment for Parkinson’s disease. The approach is […]

Bayer is committing another €1.3 billion to Leaps by Bayer, the company’s investment arm, to support additional investments in companies developing innovative technologies in healthcare […]

Bayer has filed for approval to extend the use of its prostate cancer therapy Nubeqa as it tries to achieve labelling parity with rival drugs […]

Astellas looks to be in pole position to bring a new non-hormonal therapy for symptoms of menopause to market, after its candidate fezolinetant cleared a […]

Bayer has claimed EU approval for Kerendia, its new drug for chronic kidney disease (CKD) in people with type 2 diabetes, as the company tries […]

Date: March 7, 2022 Issue #: 1645 Summary: View the Comparison Table: Some Nonopioid Analgesics for Pain

Bayer now says it expects to make €3 billion in sales for its prostate cancer treatment Nubeqa, up from an earlier estimate of €1 billion, […]

Belgian biotech Oxurion has revealed new data from a phase 2 trial of its plasma kallikrein inhibitor THR-149, in development for people with diabetic macular […]

The FDA has granted fast-track status to Bayer’s oral Factor Xia inhibitor asundexian, which is in phase 2 testing as an anticoagulant that could offer […]

Bayer has abandoned development of its investigational P2X3 receptor antagonist eliapixant, despite promising efficacy data in clinical trials, on what appears to be safety grounds. […]

Last year, Bayer launched a project to explore the use of artificial intelligence in clinical trials, working with Finland’s Aalto University to find ways to […]

The FDA has approved Roche’s much-anticipated faricimab for two major causes of blindness, setting up a market clash with Bayer and Regeneron’s market leading drug […]

The FDA has issued a complete response letter (CRL) to Merck & Co’s gefapixant candidate for refractory chronic cough, delaying the programme and giving Bayer […]

As the annual J.P. Morgan HealthCare Conference kicked off Monday, two pharma giants unveiled deals that expand give them access to in-vivo gene editing technologies. […]

Bayer has bolstered its cell and gene therapy platform by securing access to a CRISPR-based gene-editing platform developed by US biotech Mammoth Biosciences. The German […]

Bayer’s Nubeqa is making headway as a treatment for castration-resistant prostate cancer (CRPC), but could be on track to expand the eligible patient population for […]

The app lets people track their metrics through connected devices, and access health coaching and educational materials. It’s one of multiple that Bayer and One […]

Bayer’s two-year-old digital health collaboration with One Drop has borne its first fruit, an artificial intelligence-powered software module for preventing cardiovascular disease (CVD). The German […]

By JESSICA DaMASSA, WTF Health Bayer’s $98M co-development-plus-investment in One Drop from August 2020 has yielded its first new product: a highly-personalized, AI-powered digital program […]

Roche’s Genentech unit has won FDA approval for Susvimo, an implant for eyesight-robbing disease wet age-related macular degeneration (AMD) that gives patients an alternative to […]

A research project between Bayer and digital health company Huma will use artificial intelligence to detect lung cancer in CT scans – and determine which […]

Everest’s SPR206 Receives the NMPA’s IND Approval for the Treatment of MDR Gram-Negative Bacterial Infections Published: Sept 24, 2021 | Tags: Everest, SPR206, NMPA, IND, […]

Shots: Gubra to receive ~ $253.4M as up front, development, and commercialization milestones The collaboration will combine Gubra’s peptide drug discovery capabilities with Bayer’s expertise […]

Bayer has released the data in its phase 2b trial of eliapixant in refractory chronic cough, as it tries to close the gap with Merck […]

Shots: The P-IIb PAGANINI study involves assessing the efficacy, safety & tolerability of eliapixant (25, 75 or 150mg, bid) vs PBO in 310 patients with […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Shots: The company initiates P-III OASIS program to evaluate the efficacy and safety of elinzanetant (120mg, qd) in 1300 patients with vasomotor symptoms during menopause […]

The US FDA has approved 6 NDAs in 2021, leading to treatments for patients and advances in the health care industry The Center for Drug […]

There is plenty of evidence that Novartis’ ophthalmology drug Beovu can match its rivals when it comes to efficacy, with less frequent dosing, but the […]

There is plenty of evidence that Novartis’ ophthalmology drug Beovu can match its rivals when it comes to efficacy, with less frequent dosing, but the […]

Medtronic to Acquire Intersect ENT for ~$1.1B Published: Aug 6, 2021 | Tags: Medtronic, Intersect ENT, ~$1.1B Kite Signs a License Agreement with Appia to […]

Shots: Vividion to receive $1.5B up front & $500M milestone while Bayer to get full rights to Vividion’s discovery platform which consists of chemoproteomic screening […]

Bayer headlined its second-quarter results this morning by unveiling a deal to buy Vividion Therapeutics, saying it will boost its ability to develop medicines against […]

Bayer says its drug candidate for chronic cough – eliapixant – has hit the mark in a phase 2b trial, putting it in hot pursuit […]

Shots: The P-IIb clinical trial involves evaluating the efficacy and safety of eliapixant (25/75/150mg, PO, bid) vs PBO in 300 patients with refractory chronic cough […]

The FDA has started a priority review of Roche’s bispecific antibody faricimab for two major causes of blindness, preparing for a market showdown with Bayer […]

Bayer subsidiary BlueRock Therapeutics has been granted a fast-track review by the FDA for DA01, its stem cell-based therapy for Parkinson’s disease which is currently […]

Eli Lilly Signs an Agreement with Banner Alzheimer’s Institute to Evaluate Donanemab in P-III TRAILBLAZER-ALZ 3 Trial for Alzheimer’s Disease Published: July 16, 2021 | […]

Bayer’s big investment in time and money on the development of finerenone for chronic kidney disease (CKD) in diabetics has paid off with an FDA […]

Shots: The approval is based on a P-III FIDELIO-DKD study evaluating finerenone (10/20mg) + SoC vs PBO + SoC in ~ 5700 patients with CKD […]

The FDA has approved Bayer’s Kerendia to treat chronic kidney disease in patients with type 2 diabetes. The drug is the first in its class, […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

Vertex and CRISPR Therapeutics Present New Data of CTX001 from 22 Patients with TDT and SCD at EMA 2021 Published: June 11, 2021 | Tags: […]

Bayer subsidiary BlueRock Therapeutics has dosed the first Parkinson’s disease patient in a phase 1 trial of its cell-based therapy DA01, which aims to replenish […]

Bayer has agreed to buy Noria Therapeutics and its subsidiary PSMA Therapeutics, adding to its pipeline of radionuclide drugs for cancer as sales of its […]

Leaps by Bayer led a $90 million investment round into Ada, a German digital health startup with an AI-based symptom checker. As part of the […]

Prospects for Novartis’ ophthalmology drug Beovu have taken another dive after the drugmaker reported toxicity issues in a new phase 3 trial in wet age-related […]

Seelos’ SLS-005 (Trehalose) Receives EC’s Orphan Drug Designation to Treat Amyotrophic Lateral Sclerosis Published: May 28, 2021 | Tags: Seelos, SLS-005, Trehalose, EC, Orphan Drug […]

Ada Health has received another endorsement for its app to help patients diagnose conditions from symptoms, raising an impressive $90 million in funding from investors […]

Shots: The NDA submission is based on P-I study of adult patients, the P-II NAVIGATE trial in adult and adolescent patients and the P- I/II […]

BlueRock Therapeutics, a Bayer subsidiary, is teaming up with Opsis Therapeutics and Fujifilm Cellular Dynamics in an R&D pact focused on developing stem cell therapies […]

AzurRx BioPharma Reports Positive Interim P-II Data of MS1819 + PERT for Cystic Fibrosis Patients with Severe Exocrine Pancreatic Insufficiency Published: May 14, 2021 | […]

Shots: The P-III FIGARO-DKD study involves assessing the efficacy and safety of finerenone (10/20mg, PO, qd) + SoC vs PBO + SoC in ~7400 patients […]

ThermoFisher to Acquire PPD for ~$17.4B Published: Apr 16, 2021 | Tags: ThermoFisher, Acquire, PPD, ~$17.4B Roche’s Evrysdi (risdiplam) Receives Health Canada’s Approval for Spinal […]

Shots: The P-III CHRONOS-3 study involves assessing Aliqopa (IV) + rituximab vs PBO + rituximab in 458 patients with relapsed iNHL in extending PFS who […]

Pfizer and BioNTech Initiate COVID-19 Vaccine Trial in Children Under 12 Published: Mar 26, 2021 | Tags: Pfizer and BioNTech, Initiate, COVID-19 Vaccine Trial, Children, […]

Shots: The approval is based on the P-III NAVIGATE trial in adult and adolescent patients and P- I/II pediatric SCOUT trial involves assessing Vitrakvi for […]

The US FDA has approved 4 NDAs in Jan 2021, leading to treatments for patients and advances in the health care industry. The Center for […]

COVID-19 has already proved to be a digital accelerant for the pharmaceutical industry, sparking new ideas into life and lighting a fire underneath existing plans […]

NICE has recommended regular NHS funding for Novartis’ long-acting eye drug Beovu (brolucizumab) in England and Wales – but a patient group warned that access […]

Takeda’s Alunbrig (brigatinib) Receives MHLW’s Approval as a 1L and 2L Treatment for ALK+ Advanced or Recurrent NSCLC Published: Jan 22, 2020 | Tags: Takeda, […]

Bayer and Merck & Co’s heart failure drug vericiguat has been approved by the FDA under the brand name Verquvo, in an increasingly competitive market. […]

Shots: The approval is based on pivotal P-III VICTORIA trial involves assessing of Verquvo (2.5mg, 5mg & 10mg) vs PBO in 5,050 adult patients with […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

Almirall and Tyris Collaborate to Develop Next Generation Gene Therapies Published: Jan 7, 2020 | Tags: Almirall, Tyris, Collaborate, Develop, Next Generation, Gene Therapies Sarepta […]

Shots: Bayer to support the development, supply and key territory operations of CureVac´s COVID-19 vaccine candidate, CVnCoV and will contribute its expertise and established infrastructure […]

Biopharma merger and acquisition activity in 2020 was mainly filled with late-stage, bolt-on acquisitions, which were orders of magnitude smaller than the mega M&A deals […]

Shortly after launching large-scale trials of its COVID-19 vaccine candidate, CureVac has the partner it will need to roll the shot out at scale if […]

“Lockdown’ declared Collins Dictionary word of the year. The year 2020, well known as COVID-19 year has been a busy year for global pharma and […]

Roche Launches Elecsys SARS-CoV-2 Antigen Test to Support High-Volume COVID-19 Testing Published: Dec 11, 2020 | Tags: Antigen, COVID-19, High, Laboratory, Launch, patients, Roche, SARS-CoV-2, […]

Shots: Atara to receive $60M upfront and ~$610M as development, regulatory & commercialization milestones + tiered royalties on net sales. The collaboration focuses on off-the-shelf […]

KaliVir, Astellas Pharma forms a licensing deal for VET2-L2 oncolytic virus KaliVir Immunotherapeutics and Astellas Pharma entered into a worldwide exclusive licensing agreement for the […]

Eli Lilly and Incyte Receive FDA’s EUA for Baricitinib + Remdesivir to Treat Hospitalized Patients with COVID-19 Published: Nov 20,2020 | Tags: baricitinib, COVID-19, Eli […]

Shots: The funding focuses on fostering the development of a digital health ecosystem in the fields of cardiometabolic and renal diseases, oncology, and women’s health […]

Bayer has provided funding to five more digital health start-ups as part of its G4A partnering programme, which has already provided backing for more than […]

Shots: The P-III FIDELIO-DKD study in CKD and T2D enrolled over 13,000 patients with early kidney damage and more advanced stages of kidney disease. The […]

Qiagen Launches Portable Digital SARS-CoV-2 Antigen Test in the US Published: Nov 13,2020 | Tags: Antigen Test, Digital, Launches, Portable, Qiagen, SARS-CoV-2, US GSK and […]

Healthy animals contribute to the elimination of hunger and sustainable food production. The diseases that affect animal health also affect humans. Tackling them while they […]

Shots: The MAA submitted to the EMA and the NDA to the US is based on P-III FIDELIO-DKD study assessing finerenone (10/20mg, qd) + SOC […]

The European Commission has approved Roche’s Tecentriq immunotherapy, in combination with its established cancer drug Avastin, for patients with the most common form of liver […]

The deal, offering $2 billion up front and $2 billion in future milestone payments, includes a gene therapy manufacturing facility and a pipeline that includes […]

Shots: Bayer acquires AskBio for $2B as upfront and ~2B as milestones with 75% of the milestones to be paid during the next five years. […]

Bayer is making clear its ambitions in cell and gene therapy with a $4 billion acquisition of US biotech Asklepios BioPharmaceutical, also known as AskBio. […]

Bayer’s blockbuster ambitions for diabetic kidney disease (DKD) drug candidate finerenone look a lot firmer with the publication of data from the phase 3 FIDELIO-DKD […]

Akebia Presents Results of Vadadustat in P-lll INNO2VATE Program for Anemia Due to CKD at ASN Kidney Week Published: Oct 23, 2020 | Tags: Akebia, […]

Shots: The companies will collaborate on drug commercialization initiatives in China, including the commercialization of innovative drugs & healthcare products for chronic disease management, as […]

Novartis’s Luxturna (voretigene neparvovec) Receives Health Canada Approval as the First Gene Replacement Therapy for Inherited Retinal Disease Published: Oct 15, 2020 | Tags: approval, […]

Shots: The P-III CHRONOS-3 study involves assessing of Aliqopa + rituximab vs PBO + rituximab in 458 patients with relapsed indolent NHL who have received […]

Shares in Amgen were down nearly 7% after close of trading yesterday after the company’s heart failure drug omecamtiv mecarbil disappointed in a large phase […]
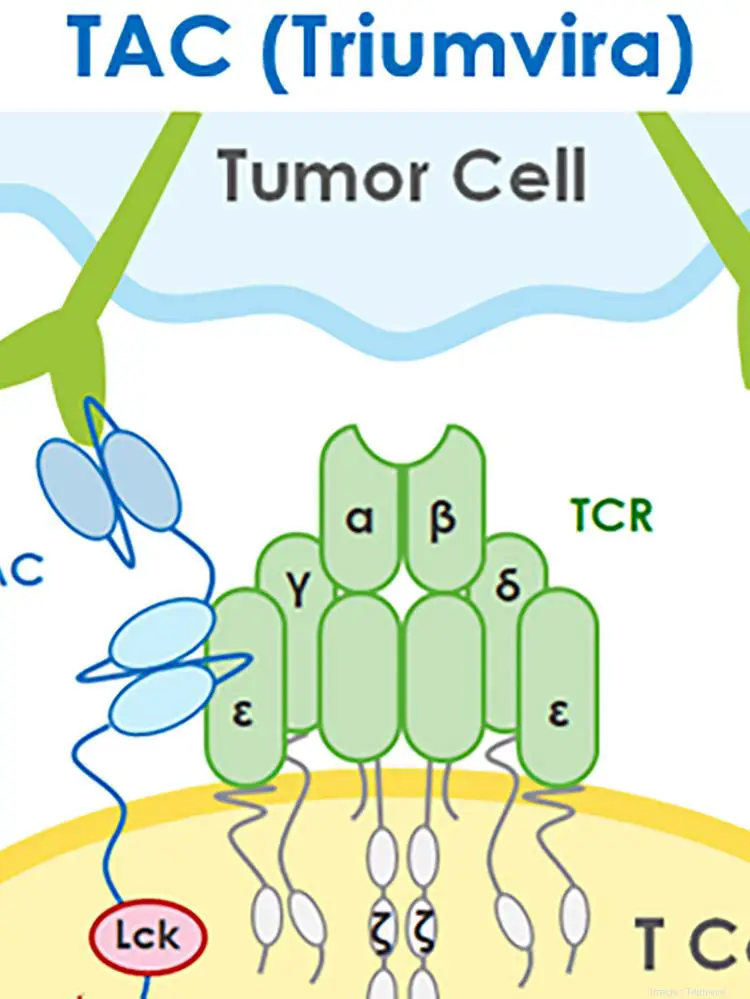
The company hopes to use the money to bring its T-cell antigen coupler cell therapy technology – currently in preclinical development – into human clinical […]

1. Taysha Gene Therapies’ TSHA-101 Receives the US FDA’s Orphan Drug Designation and Rare Pediatric Disease Designation for GM2 Gangliosidosis Published: Aug 27, 2020 | […]

Shots: The NDA submission was submitted to NMPA’s CDE and is based on P-III VICTORIA study, seeking regulatory approval of vericiguat in China In Oct’2014, […]

By JESSICA DaMASSA, WTF HEALTH One Drop just landed a $98.7M deal with Bayer — and we got the details from CEO Jeff Dachis. The […]

Bayer has taken a further stake in New York-based One Drop to support its plans to become a digital health business by developing new integrated […]

Shots: The two partners will jointly enhance the existing platform utilizing One Drop’s know-how and capabilities of the One Drop platform and will co-develop digital […]

KaNDy’s lead product candidate, NT-814, is a non-hormonal treatment for symptoms of menopause. The drug completed Phase IIb testing and is slated to start Phase […]

Shots: KaNDy to receive $425M upfront, ~$450M milestones until launch followed by additional commercial milestones. The transaction is expected to be completed in Sept’2020 The […]

Bayer is to buy UK-based KaNDy Therapeutics in a deal worth more than $800 million, adding the biotech’s menopause drug to its women’s health pipeline. […]

Bayer counted the cost of its expensive Roundup weedkiller settlement in the second quarter with a €9.5 billion net loss made worse by lower sales […]

Roche’s tumour-agnostic therapy Rozlytrek has been approved in the European Union for people with tumours with the NTRK mutation, meaning Bayer’s class rival faces competition […]

1. Oxford Biomedica Signs Three Year Clinical Supply Agreement with Axovant to Manufacture and Supply AXO-Lenti-PD for Parkinson’s Disease Published: Jul 31, 2020 | Tags: Oxford […]

1. Gilead and Galapagos’ Jyseleca (filgotinib) Receive the CHMP’s Positive Opinion for Moderate to Severe Rheumatoid Arthritis Published: Jul 24, 2020 | Tags: Gilead, Galapagos, Jyseleca, […]
1. Eli Lilly Reports Results of Mirikizumab in P-III OASIS-2 Study to Treat Moderate to Severe Plaque Psoriasis Published: Jul 17, 2020 | Tags: Eli Lilly, […]
The FDA has granted a faster Priority Review for Bayer and Merck & Co’s filing for vericiguat, which is being developed for chronic heart failure with reduced ejection […]

Shots: The NDA is based on P-III VICTORIA study assessing vericiguat (qd, (titrated up to 10mg) vs PBO when given in combination with available HF […]

I have to inject myself with needles just to stay alive. Still, Bayer will continue to sell Roundup, and refused to label it as carcinogenic […]
Copyright © 2024 | WordPress Theme by MH Themes