
What has Covid-19 taught us about virtual clinical trials?
At the upcoming INVEST PharmaTech virtual conference scheduled for July 26, the conversations will span myriad topics at the intersection of digital health and drug […]

At the upcoming INVEST PharmaTech virtual conference scheduled for July 26, the conversations will span myriad topics at the intersection of digital health and drug […]

Jazz’ Sunosi (solriamfetol) Receives NICE Recommendation for the Treatment of Excessive Daytime Sleepiness Caused by Narcolepsy Published: Nov 19, 2021 | Tags: Jazz, Sunosi, solriamfetol, […]

Shots: The P-II AXIOMATIC-TKR study evaluates the efficacy and safety of milvexian (qd/bid) vs enoxaparin (SC) in 1242 patients with elective TKR surgery The trial […]

Eisai Presents Results of Lecanemab in P-IIb Study 201 Study for the Treatment of Alzheimer’s Disease at CTAD 2021 Published: Nov 12, 2021 | Tags: […]

Shots: The companies collaborated to evaluate Inspirna’s RGX-104 + BMS’ Yervoy in P-Ib/II expansion study for patients with metastatic endometrial cancer whose genomes holds E2 […]

Episode 42 of the pharmaphorum podcast heard from Catherine Owen, who’s senior vice president of major markets at Bristol Myers Squibb, about the future of […]

Shots: The P-II LATTICE-UC study evaluates the safety and efficacy of deucravacitinib (PO) vs PBO in patients with mod. to sev. UC The trial did […]

BMS Reports EMA’s Validation of MAA for Relatlimab and Nivolumab as 1L Treatment for Unresectable or Metastatic Melanoma Published: Oct 1, 2021 | Tags: BMS, […]

Shots: The MAA was based on the efficacy & safety results from the P-II/III RELATIVITY-047 trial that evaluates the fixed-dose combination of relatlimab (160mg) + […]

The COVID-19 pandemic kickstarted a period of profound change in how the pharmaceutical industry approaches both communications and commercialisation. In some cases it was an […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

In an interview with PharmaShots, Dr. Ian Waxman, MD, Development Lead, Gastrointestinal Cancer at BMS shared his views on the EC’s approval of Opdivo (nivolumab) […]

Shots: The P-III CheckMate -214 trial evaluates Opdivo (3mg/kg) + Yervoy (1mg/kg, q3w for 4 doses) vs sunitinib in 1096 patients with previously untreated advanced/mRCC […]

Shots: The application is based on P-III CheckMate -648 study evaluating Opdivo (3mg/kg, q2w, IV) + Yervoy (1mg/kg, q6w up to 24mos.) or Opdivo + […]

Shots: The P-III CheckMate -743 trial evaluates Opdivo (3mg/kg, q2w) + Yervoy (1mg/kg, q6w) vs CT in 605 patients with previously untreated unresectable MPM. The […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Shots: Evotec receives $20M as an option fee & is eligible to receive ~$250M as milestones along with royalties. BMS will be responsible for further […]

Ascendis’ Skytrofa (lonapegsomatropin-tcgd) Receives the US FDA’s Approval for Pediatric Growth Hormone Deficiency Published: Aug 27, 2021 | Tags: Ascendis, Skytrofa, lonapegsomatropin-tcgd, US, FDA, Approval, […]

Janssen Presents Results of Rybrevant (amivantamab-vmjw) in P-I CHRYSALIS Study for Advanced NSCLC with METex14 Mutations at WCLC Published: Aug 20, 2021 | Tags: Astellas, […]

Shots: The conditional approval is based on KarMMa study evaluating Abecma (single infusion with a target dose of 420 x 106 CAR T cells) in […]

Shots: The applications are based on P-III CheckMate -648 trial evaluates Opdivo (3 mg/kg, q2w) + Yervoy (1 mg/kg, q6w up to 24mos.) or Opdivo […]

Immunology deals with physiological functioning of the immune system in states of both health and disease as well as malfunctions of the immune system in […]

Looking back over the past 18 months, the Joni Mitchell lyric ‘you don’t know what you’ve got til it’s gone’ comes to mind. It’s only now, […]

Roche Publishes the Results of Evrysdi (risdiplam) in FIREFISH Study to Treat of Type 1 SMA at NEJM Published: July 30, 2021 | Tags: Roche, […]

Shots: The companies will initiate P-I/II study evaluating the safety and preliminary efficacy of BBP-398 in combination with both Opdivo as dual therapy & Opdivo […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

Tonix Pharmaceuticals Presents Results of TNX-102 SL in P-III RELIEF study for the Management of Fibromyalgia at ASCP 2021 Published: June 4, 2021 | Tags: […]

Shots: The P-III CheckMate -648 trial evaluating Opdivo + CT or Opdivo (3 mg/kg, q2w) + Yervoy (1 mg/kg, q6w up to 24mos.) in patients […]

Shots: The conditional approval is based on the results of the P-II KarMMa study involves assessing Abecma in 122 patients with RRMM prior treated with […]

Seelos’ SLS-005 (Trehalose) Receives EC’s Orphan Drug Designation to Treat Amyotrophic Lateral Sclerosis Published: May 28, 2021 | Tags: Seelos, SLS-005, Trehalose, EC, Orphan Drug […]

Shots: The approval is based on a P-III True North trial assess Zeposia (0.92 mg) vs PBO in an adult with mod. to sev. active […]

Shots: The approval is based on P-III CheckMate -743 study assessing Opdivo + Yervoy vs SOC Pt. based CT in patients with previously untreated unresectable […]

Shots: BMS to get non-exclusive access to Xencor’s Xtend Fc technology to extend the half-life of a novel Ab combination therapy that is intended to […]

The UK has been a global leader in introducing cell and gene therapies that can potentially transform the lives of patients with debilitating diseases – […]

Shots: The opinion is based on P-II CheckMate -142 study assessing Opdivo + Yervoy in patients with mismatch repair deficient or microsatellite instability–high metastatic colorectal […]

BeiGene Reports Results of Tislelizumab + CT in P-III RATIONALE 309 Study as 1L Treatment for Recurrent or Metastatic Nasopharyngeal Cancer Published: May 21, 2021 […]

Shots: Exscientia to receive ~$50M as up front, ~$125M as milestones and additional clinical, regulatory, and commercial milestones of over $1.2B along with royalties on […]

Shots: Agenus to receive a $200M as up front & ~$1.36B as development, regulatory and commercial milestones plus royalties on net sales of the therapies. […]

Shots: The P-III EXPLORER-HCM study involves assessing Mavacamten vs PBO in 251 patients in a ratio (1:1) with symptomatic oHCM (LVOT gradient ≥50 mmHg and […]

AstraZeneca Reports Results of Imfinzi (durvalumab) and Imfinzi + Tremelimumab in P-III POSEIDON Study as 1L Treatment for Stage IV Non-Small Cell Lung Cancer Published: […]

Shots: DarwinHealth will receive an upfront and eligible to receive as a commercial milestone, as part of the NCTI collaboration The focus of the collaboration […]

Shots: The company presented new analysis data from the P-III EXPLORER-HCM trial assessing mavacamten in patients with oHCM that tested the impact of the therapy […]

Shots: The filing is based on results from the P-III CheckMate -274 trial evaluating Opdivo (240mg, q2w for up to 1 yrs.) vs PBO in […]

Shots: The two P-III POETYK PSO-1 and POETYK PSO-2 trials evaluating deucravacitinib (6mg, qd) vs PBO & Otezla (30mg, bid) @16wks. and vs Otezla @24wks. […]

BMS’ Opdivo + Yervoy and Onureg Receive EC’s CHMP Positive Opinion for MPM and AML Published: Apr 23, 2021 | Tags: BMS, Opdivo, Yervoy, Onureg, […]

Shots: The CHMP has recommended the approval of Opdivo (nivolumab) + Yervoy (ipilimumab) as 1L treatment for unresectable MPM and is based P-III CheckMate -743 […]

ThermoFisher to Acquire PPD for ~$17.4B Published: Apr 16, 2021 | Tags: ThermoFisher, Acquire, PPD, ~$17.4B Roche’s Evrysdi (risdiplam) Receives Health Canada’s Approval for Spinal […]

Shots: The approval is based on P-III CheckMate -9ER trial involves assessing Opdivo + Cabometyx vs sunitinib in 651 patients with previously untreated advanced or […]

Shots: The P-III CheckMate-816 study involves assessing Opdivo (360mg) + CT (q3w for 3 doses) vs CT, followed by surgery as a neoadjuvant treatment in […]

Evotec and Exscientia Initiate Human Clinical Trials of their Novel Immuno-Oncology Drug Published: Apr 9, 2021 | Tags: Evotec and Exscientia, Initiate, Human Clinical Trials, […]

Shots: In the P-I SPICE study, the companies will evaluate the clinical combination of BMS’ Opdivo with PsiOxus’ NG-641 and optimize the combination IV dosing […]

Pfizer and BioNTech Report Results of BNT162b2 to Prevent COVID-19 Published: Apr 2, 2021 | Tags: Pfizer and BioNTech, Report, Results, BNT162b2, Prevent, COVID-19 Acceleron […]

Shots: The approval is based on the P-II KarMMa trial that involves assessing Abecma in 127 patients with r/r MM prior treated with 3L+ therapy […]

Pfizer and BioNTech Initiate COVID-19 Vaccine Trial in Children Under 12 Published: Mar 26, 2021 | Tags: Pfizer and BioNTech, Initiate, COVID-19 Vaccine Trial, Children, […]

Cho discusses Bristol Myers Squibb’s research efforts in the field of protein degradation to advance potential treatments for a broad range of diseases. In a […]

Shots: The P-II/III RELATIVITY-047 (CA224-047) study involves assessing the FD combination of relatlimab (160 mg) + Opdivo (480mg) vs Opdivo (480mg, IV, q4w) alone in […]

Ho Sung Cho, senior vice president of Biodiscovery Therapeutics at Bristol Myers Squibb, talks about lessons learned from drug discovery, recent success tied to Covid-19, […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Eli Lilly and Incyte Report Results of Olumiant (baricitinib) in P-III BRAVE-AA2 Study for Severe Alopecia Areata Published: Mar 5, 2021 | Tags: Eli Lilly […]

Shots: The CHMP recommendation is based on P-III CheckMate -9ER trial involves assessing Opdivo + Cabometyx vs sunitinib in 651 patients with previously untreated advanced […]

Regeneron Reports IDMC Clearance of Efficacy for REGEN-COV (Casirivimab + Imdevimab) in P-III COVID-19 Outpatient Outcomes Trial Published: Feb 26, 2021 | Tags: Regeneron, Reports, […]

RedHill Signs a Manufacturing Agreement with Cosmo for Movantik and RHB-204 Published: Feb 19, 2021 | Tags: RedHill, Signs, Manufacturing, Agreement, Cosmo, Movantik, RHB-204 Astellas and […]

How is a Rare disease defined? Well, it depends on the geography in question, since there is no universal definition, even so, the definition revolves […]

Visby’s POC COVID-19 Test Receives the US FDA’s EUA for Use in CLIA Waived Settings Published: Feb 12, 2020 | Tags: Visby, POC, COVID-19, Test, Receives, […]

Shots: MTEM to receive $70M up front and is also eligible to receive ~$1.3B as development, regulatory and sales milestone along with royalties on future […]

The biopharma industry saw numerous deal terminations in 2020. Clinical and regulatory results, change in control limitations, and strategic reprioritizations were among the most common […]

Shots: The P-III CheckMate -9ER trial involves assessing Opdivo + Cabometyx vs sunitinib in 651 patients with previously untreated advanced or metastatic RCC Results: @median […]

Shots: The EC approval is based on JAKARTA and JAKARTA2 studies. The pivotal JAKARTA study involves assessing Inrebic (500mg) vs PBO in 289 patients with […]

Reistone Report Results for SHR0302 in P-II Study to Treat Ulcerative Colitis Published: Feb 5, 2020 | Tags: Reiston, Report, Result, SHR0302, P-II, Study, Treat, […]

Shots: The P-III POETYK PSO-2 study involves assessing deucravacitinib (6mg, qd) vs PBO & Otezla (30mg, bid) in 1,020 patients with moderate to severe plaque […]

In a recent interview with PharmaShots, Patricia Mae Doykos, Lead, Health Equity Initiative, and Director, BMS Foundation at Bristol Myers Squibb shared her views on […]

Life sciences companies are gearing up to enter new markets as they look to secure their positions after a spate of M&A, licensing, and research […]

In its report released last Tuesday, a nonprofit drug pricing research group claims that seven drugs have prices that do not align with any newly […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

The fourth quarter of 2020 contains multiple initiations of clinical trials, big approvals, and numerous deals. COVID-19 related news remains at the peak in this […]

Merck Collaborates with Siemens for the Digitalization of Production Published: Nov 26, 2020 | Tags: Agreement, Collaborative, Digitalization, Merck, Siemens, Sign CoSara Receives CDSCO’s Approval […]

Shots: Schrödinger to receive $55M upfront and is eligible to receive $2.7B as preclinical, development, regulatory & commercial milestones along with royalties on sales of […]

AstraZeneca’s Brilinta (ticagrelor) Receives the US FDA’s Approval to Reduce the Risk of Stroke in Patients with an Acute Ischemic Stroke or High-Risk Transient Ischemic […]

Shots: The P-III POETYK PSO-1 study involves assessing deucravacitinib (6mg, qd) vs PBO & Otezla (apremilast) in 666 patients with moderate to severe plaque psoriasis […]

Shots: Insitro to receive $50M as up front, $20M as near-term operational milestones and is eligible to receive ~$2B+ as discovery, development, regulatory and commercial […]

Immunology is an important branch of science which deals with the study of the immune system. The immune system is a highly regulated and balanced […]

The US FDA has approved 6 NDAs in Sept 2020, leading to treatments for patients and advances in the health care industry. The Center for […]

Shots: The approval is based on P-lll SUNBEAM & RADIANCE part B study which involves the assessing of Zeposia (0.92mg and 0.46 mg) vs Avonex […]
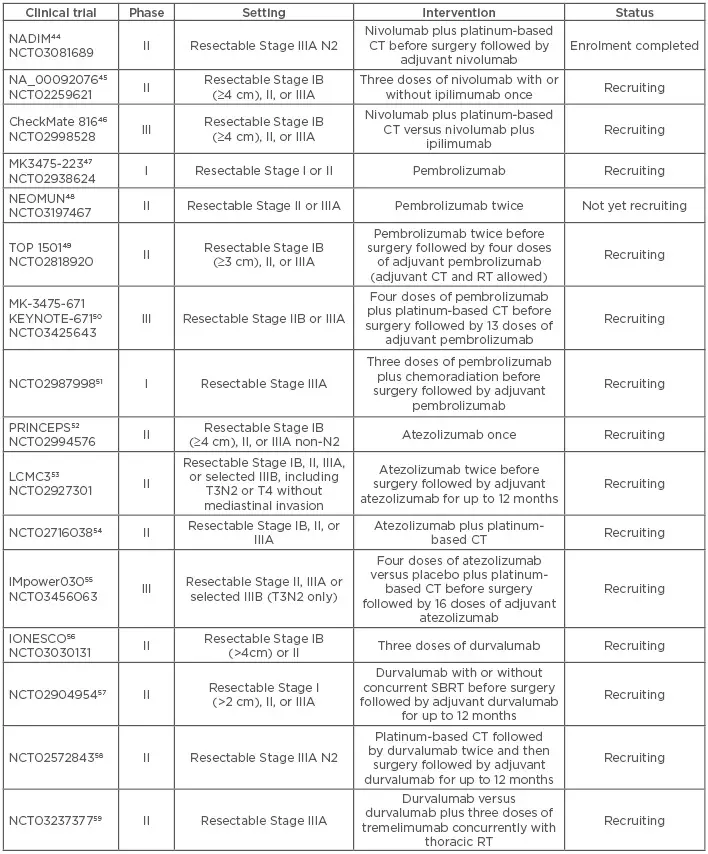
Shots: The P-III CheckMate-816 study involves assessing of Opdivo (360mg) + CT (q3w for 3 doses) vs CT (q3w for 3 doses), followed by surgery […]

Shots: BMS to acquire all the outstanding shares of MyoKardia’s common stock for $225.00/ share in cash, making a total deal value of ~$13.1B. The […]
Sobi & Selecta Report Results of SEL-212 in P-II COMPARE Study for Chronic Refractory Gout Published: Oct 1, 2020 | Tags: Chronic Refractory Gout, COMPARE […]

Shots: Forbius will receive up front and milestones while BMS will acquire Forbius’s TGF-beta program, including the lead investigational asset, AVID200. The transaction is expected […]

1. BioMarin Reports NDA Submission to the US FDA for Vosoritide to Treat Children with Achondroplasia Published: Aug 20, 2020 | Tags: BioMarin, Reports, NDA, […]

Shots: Dragonfly to receive $475M as up front and is eligible to receive development, regulatory and commercial milestones in addition to 24% royalties on global […]

Shots: The approval is based on P-III CheckMate -9LA study assessing Opdivo + Yervoy combined with two cycles of platinum-doublet CT vs CT (four cycles […]
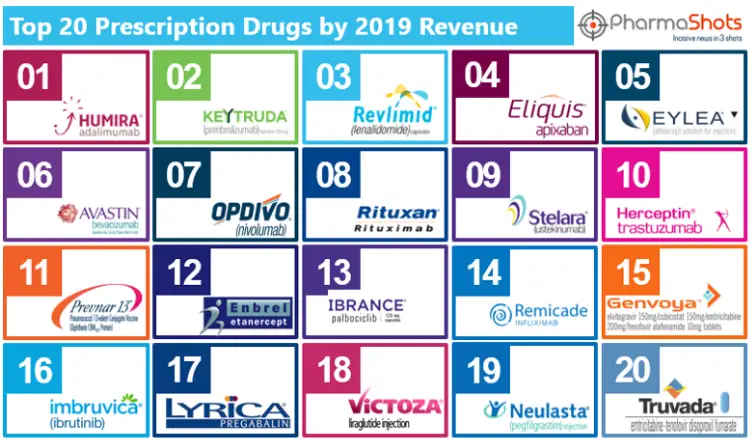
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
Copyright © 2024 | WordPress Theme by MH Themes