
UK green lights Gilead Sciences, Galapagos ulcerative colitis drug
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has approved Jyseleca (filgotinib) 200mg tablets to treat ulcerative colitis (UC) in adults whose UC is […]

The UK Medicines and Healthcare products Regulatory Agency (MHRA) has approved Jyseleca (filgotinib) 200mg tablets to treat ulcerative colitis (UC) in adults whose UC is […]

US pharma giant Gilead Sciences has accused a network of unauthorised suppliers and distributors of selling more than $250 million of counterfeit versions of its […]

Jazz’ Sunosi (solriamfetol) Receives NICE Recommendation for the Treatment of Excessive Daytime Sleepiness Caused by Narcolepsy Published: Nov 19, 2021 | Tags: Jazz, Sunosi, solriamfetol, […]

Gilead Sciences has forged closer ties to its cancer drug partner Arcus Biosciences , although a licensing deal doesn’t go as far as the takeover […]

Shots: Gilead has exercised its options to 3 programs in Arcus’s portfolio, including both anti-TIGIT molecules, domvanalimab & AB308 along with etrumadenant & quemliclustat & […]

Lonza Collaborates with Codiak to Acquire its Exosomes Manufacturing Facility in Lexington, Massachusetts (US) Published: Nov 3, 2021 | Tags: Lonza, Codiak, Acquire, Exosomes Manufacturing […]

Shots: The ongoing BICSTaR study evaluates the antiviral effectiveness & safety of Biktarvy (bictegravir 50 mg/emtricitabine 200 mg/tenofovir alafenamide 25 mg tablets) in 1135 patients […]

LEO Reports 16-week Results of Tralokinumab in P-III ECZTRA 6 Trial for the Treatment of Moderate-to-Severe Atopic Dermatitis Published: 22 Oct, 2021 | Tags: LEO, […]

Shots: The approval is based on cohort 3 of a P-II/III study that evaluates Biktarvy (bictegravir 30mg/emtricitabine 120mg/tenofovir alafenamide 15mg) in 22 Pediatric patients with […]

This thought leadership series has been paid and developed by Kite, a Gilead Company. This Blood Cancer Awareness Month, Blood Cancer UK’s Kate Keightley explains […]

Everest’s SPR206 Receives the NMPA’s IND Approval for the Treatment of MDR Gram-Negative Bacterial Infections Published: Sept 24, 2021 | Tags: Everest, SPR206, NMPA, IND, […]

Shots: The P-III trial evaluates the efficacy & safety of a 3-day course of Veklury (IV) vs PBO in a ratio (1:1) in 562 non-hospitalized […]

Working in partnership will be key, says Alex Kalomparis, vice president, public affairs, international at Gilead Sciences. 2021 marks 40 years since the first cases […]

The COVID-19 pandemic has highlighted the importance of ensuring a reliable supply of treatments across the globe at pace and speed. Jirair Ratevosian, executive director of […]

Janssen Presents Results of Rybrevant (amivantamab-vmjw) in P-I CHRYSALIS Study for Advanced NSCLC with METex14 Mutations at WCLC Published: Aug 20, 2021 | Tags: Astellas, […]

Shots: The MAA is based on the ongoing P-II/III CAPELLA study evaluates the antiviral activity of lenacapavir (PO, SC, every 6mos.) in combination with other […]

The US FDA has approved 6 NDAs and 2 BLAs in 2021, leading to treatments for patients and advances in the health care industry The […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

Lineage Signs an Exclusive Option Agreement with Amasa Therapeutics to Supply and Use of Clinical-Grade Hystem Published: June 18, 2021 | Tags: Lineage, Option Agreement, […]

Shots: Jounce to receive $85M upfront and $35M in its common stock under the terms of Sep 2020 agreement. Additionally, it is eligible to receive […]

Vertex and CRISPR Therapeutics Present New Data of CTX001 from 22 Patients with TDT and SCD at EMA 2021 Published: June 11, 2021 | Tags: […]

Shots: The US FDA has approved the NDA of an oral pellet formulation of Epclusa (sofosbuvir 200 mg/velpatasvir 50 mg and sofosbuvir 150 mg/velpatasvir 37.5 […]

The UK has been a global leader in introducing cell and gene therapies that can potentially transform the lives of patients with debilitating diseases – […]

In 2018, the former leadership of team from Kite Pharma founded Allogene, on a mission to outdo the CAR-T cancer cell therapies they had just […]

BMS’ Opdivo + Yervoy and Onureg Receive EC’s CHMP Positive Opinion for MPM and AML Published: Apr 23, 2021 | Tags: BMS, Opdivo, Yervoy, Onureg, […]

Shots: The submission is based on P-IIb/III SELECTION study evaluating the efficacy and safety of filgotinib (200mg, qd) vs PBO in patients with mod. to […]

Dr. Anne Kerber, VP, Head of Clinical Development, Europe at Kite, a Gilead Company, explains how the company works with researchers and physicians across Europe […]

Gilead’s Trodelvy has another string to its bow after FDA approval in patients with advanced metastatic urothelial cancer. Trodelvy (sacituzumab govitecan) has been approved for […]

Evotec and Exscientia Initiate Human Clinical Trials of their Novel Immuno-Oncology Drug Published: Apr 9, 2021 | Tags: Evotec and Exscientia, Initiate, Human Clinical Trials, […]

Gilead’s $21 billion purchase of Immunomedics and its potential blockbuster Trodelvy continues to pay off, as the drug has now been approved by the FDA […]

Shots: AbCellera to receive upfront, milestone, and royalties based on the development and commercialization of Abs, emerge under the collaboration The expanded collaboration will utilize […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Shots: The companies plan to initiate a P-IIb study assessing NovoNordisk’s semaglutide and FD combination of Gilead’s cilofexor + firsocostat vs PBO in ~440 patients […]

Shots: The companies plan to initiate a P-IIb study assessing NovoNordisk’s semaglutide and FD combination of Gilead’s cilofexor + firsocostat vs PBO in ~440 patients […]

Shots: The companies will co-develop & co-commercialize the combination of Gilead’s Lenacapavir and Merck’s Islatravir in long-acting oral & injectable formulations for HIV. Gilead & […]

PhaseBio Signs a Supply Agreement with BioVectra to Support the Development and Commercialization of Bentracimab Published: Mar 12, 2021 | Tags: PhaseBio, Signs, Supply, Agreement, […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Shots: The P-II/III CAPELLA trial involves assessing lenacapavir (SC, every 6mos.) vs PBO in 36 participants in a ratio (2:1) with multi-drug resistant HIV-1 infection […]

Lymphoma Action is the only UK health charity dedicated to people affected by lymphoma. Chief Executive Ropinder Gill and Director of Operations and External Affairs […]

Shots: The two OLE P-III 1489 and 1490 studies assessing Biktarvy (bictegravir 50 mg/emtricitabine 200 mg/tenofovir alafenamide 25 mg) vs dolutegravir-containing triple for the treatment […]

The biopharma industry saw numerous deal terminations in 2020. Clinical and regulatory results, change in control limitations, and strategic reprioritizations were among the most common […]

Shots: The P-III program consists of two trials (ISABELA 1 & 2) assessing ziritaxesta (200/ 600mg, qd) + SOC vs PBO + SOC in ~1500 […]

Reistone Report Results for SHR0302 in P-II Study to Treat Ulcerative Colitis Published: Feb 5, 2020 | Tags: Reiston, Report, Result, SHR0302, P-II, Study, Treat, […]

Gilead’s COVID-19 drug Veklury (remdesivir) is helping the company through disruption caused by the pandemic and patent expiry on two HIV drugs, according to its […]

Shots: Gristone to receive $60M at closing, including $30M up front and a $30M as an equity investment at a premium and is eligible to […]

This year J.P. Morgan 39th Annual Healthcare Conference was conducted virtually and we witnessed multiple announcements from numerous Biopharma companies An analysis of events and […]

Patients with rheumatoid arthritis (RA) in England will be able to get treatment with Gilead Sciences and Galapagos’ JAK inhibitor Jyseleca, after it was backed […]

NHS patients in England will be among the first in the world to receive Gilead’s Tecartus cancer cell therapy for certain types of lymphoma, after […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

Shots: The companies plan to initiate a P-II study assessing selgantolimod + VIR-2218 in treatment-experienced and treatment-naïve people living with HBV. People in the trial […]

Biopharma merger and acquisition activity in 2020 was mainly filled with late-stage, bolt-on acquisitions, which were orders of magnitude smaller than the mega M&A deals […]

Analysts are continuing to back Inventiva’s lanifibranor as a potential “best-in-class” drug for the fatty liver disease NASH, as the company prepares for the launch […]

Louis van de Wiel, Vice President, Site Head EU Manufacturing, Kite, a Gilead Company, reveals the complexity that sits behind the process of individualised cell […]

“Lockdown’ declared Collins Dictionary word of the year. The year 2020, well known as COVID-19 year has been a busy year for global pharma and […]

2020’s M&A activity hasn’t quite reached the heights of last year’s, where two pharma mega-mergers – BMS’ buyout of Celegne and AbbVie’s acquisition of Allergan […]

Sandoz to Launch Hyrimoz (biosimilar, adalimumab) in Canada Published: Dec 17, 2020 | Tags: Authorization, Biosimilar, canada, health, Humira, Hyrimoz, Launch, receives, Sandoz Amgen’s Riabni […]

Shots: Following the FDA Type A meeting, Gilead will not pursue the approval of Jyseleca for RA in the US. Galapagos will be solely responsible […]

Hepatitis is an infection that causes inflammation of the liver. The condition can be self-limiting or can progress to fibrosis (scarring), cirrhosis, or liver cancer. […]

Roche Launches Elecsys SARS-CoV-2 Antigen Test to Support High-Volume COVID-19 Testing Published: Dec 11, 2020 | Tags: Antigen, COVID-19, High, Laboratory, Launch, patients, Roche, SARS-CoV-2, […]

Doctors in the US are to get another option to treat COVID-19 after the FDA granted an Emergency Use Authorization (EUA) for Eli Lilly’s baricitinib. […]

The US FDA has approved 4 NDAs and 1 BLA in Oct 2020, leading to treatments for patients and advances in the health care industry. […]

GlaxoSmithKline’s HIV joint venture ViiV Healthcare could mount a challenge to Gilead’s HIV pre-exposure prophylaxis (PrEP) medicines after the FDA granted Breakthrough Therapy Designation for […]

Gilead and Novo Nordisk have announced drug combinations for the fatty liver disease known as NASH involving the GLP-1 class drug semaglutide have checked out […]

The third quarter of 2020 was flooded with major acquisitions in the pharma and biotech industry along with multiple approvals. Starting with the latest acquisitions, […]

Akebia Presents Results of Vadadustat in P-lll INNO2VATE Program for Anemia Due to CKD at ASN Kidney Week Published: Oct 23, 2020 | Tags: Akebia, […]

Gilead’s Veklury (remdesivir) has become the first COVID-19 drug approved in the US following a nod from the FDA. As an antiviral drug, remdesivir works […]

Shots: The approval is based on three studies including P-lll ACTT-1 study assessing the efficacy and safety of a 10-day treatment course of Veklury vs […]

Novartis’s Luxturna (voretigene neparvovec) Receives Health Canada Approval as the First Gene Replacement Therapy for Inherited Retinal Disease Published: Oct 15, 2020 | Tags: approval, […]

Gilead’s Veklury (remdesivir) has failed to produce an effect on hospital stay and mortality in COVID-19 patients in a large global trial, casting doubt on […]
Eli Lilly’s Olumiant (baricitinib) has a significant effect on recovery from COVID-19 when combined with Gilead Veklury (remdesivir), according to a large trial backed by […]

1. Taysha Gene Therapies’ TSHA-101 Receives the US FDA’s Orphan Drug Designation and Rare Pediatric Disease Designation for GM2 Gangliosidosis Published: Aug 27, 2020 | […]

1. BioMarin Reports NDA Submission to the US FDA for Vosoritide to Treat Children with Achondroplasia Published: Aug 20, 2020 | Tags: BioMarin, Reports, NDA, […]

Shots: The US FDA has issued a complete response letter for the NDA of filgotinib to treat moderately to severely active RA. FDA has requested […]

The FDA has rejected Gilead and Galapagos’ rheumatoid arthritis (RA) pill filgotinib over concerns that it could damage male fertility, prompting speculation that the big […]
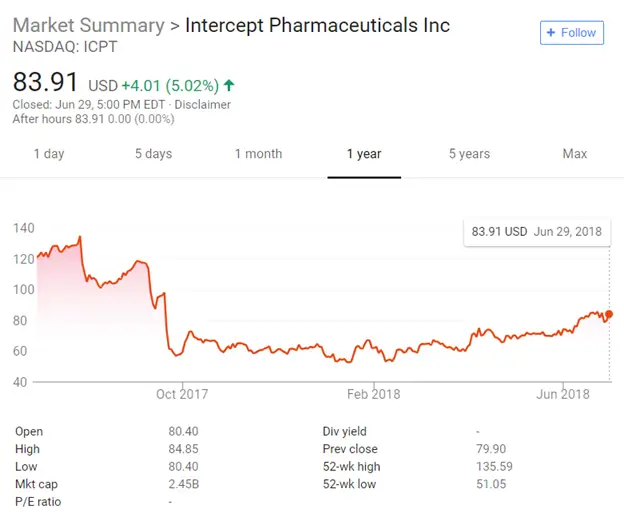
Shots: Tango to receive $125M up front, $20M as equity investment, up to $410M/ program as an opt-in, extension, and milestones along with royalties on […]

1. Roche’s Evrysdi (risdiplam) Receives the US FDA’s Approval for SMA in Adults and Children Published: Aug 10, 2020 | Tags: Roche, Evrysdi, risdiplam, Receives, US, […]
Shots: The NDA filing is based on P-III studies evaluating Veklury vs PBO, conducted by NIAID. The studies demonstrated that Veklury led to faster time […]

Pfizer has signed a multi-year agreement with Gilead to manufacture and supply the COVID-19 antiviral remdesivir, which is also being tested in combination with other […]

1. Oxford Biomedica Signs Three Year Clinical Supply Agreement with Axovant to Manufacture and Supply AXO-Lenti-PD for Parkinson’s Disease Published: Jul 31, 2020 | Tags: Oxford […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]
Gilead has blamed a $3.3 billion loss in Q2 on its acquisition of immune-oncology company Forty Seven – but revenues are declining as the pandemic […]

1. Gilead and Galapagos’ Jyseleca (filgotinib) Receive the CHMP’s Positive Opinion for Moderate to Severe Rheumatoid Arthritis Published: Jul 24, 2020 | Tags: Gilead, Galapagos, Jyseleca, […]

Shots: Then CHMP’s positive opinion is based on P-III FINCH and P-II DARWIN programs that included 4,544 RA patient-years of experience with filgotinib. All three […]

Shots: Gilead invests $300M to obtain 49.9% equity interest in Tizona and receives an exclusive option to acquire remaining stakes of Tizona for up to […]
1. Eli Lilly Reports Results of Mirikizumab in P-III OASIS-2 Study to Treat Moderate to Severe Plaque Psoriasis Published: Jul 17, 2020 | Tags: Eli Lilly, […]

Shots: Gilead to receive an up front in cash and convertible notes into Kronos equity, regulatory & commercial milestones along with royalties on sales of […]
Copyright © 2024 | WordPress Theme by MH Themes