
Welcome to the 2nd Chronic HBV Drug Development Summit
Advance Combination Therapies, Outline Clinical Endpoints & Understand the Immunology Perspective to Achieve Functional Cures for Chronic Hepatitis B. With a tight lens on late […]

Advance Combination Therapies, Outline Clinical Endpoints & Understand the Immunology Perspective to Achieve Functional Cures for Chronic Hepatitis B. With a tight lens on late […]

After a phase 3 trial win, Madrigal Pharmaceuticals reckons it could succeed where so many other companies have failed, and finally bring a drug to […]

Bayer has bolstered its cell and gene therapy platform by securing access to a CRISPR-based gene-editing platform developed by US biotech Mammoth Biosciences. The German […]

Genfit was hit hard last year when it abandoned a phase 3 programme for elafibranor in non-alcoholic steatohepatitis (NASH), but pledged to press on with […]

Already scrambling to refile its obeticholic acid (OCA) drug for non-alcoholic steatohepatitis (NASH) after the FDA rejected it last year, Intercept Pharma was hoping for […]

US remote healthcare specialist ChronWell has raised $6 million from investors that will be used to accelerate the rollout of its patient management programmes for […]

Shares in NGM Biopharma have plummeted after the US biotech said it would abandon development of aldafermin in non-alcoholic steatohepatitis (NASH), adding to a lengthening […]

Two years after starting to work together, Novo Nordisk and Dicerna have selected the first candidate from a joint project to find new, gene-silencing drugs […]

Gilead and Novo Nordisk have announced drug combinations for the fatty liver disease known as NASH involving the GLP-1 class drug semaglutide have checked out […]
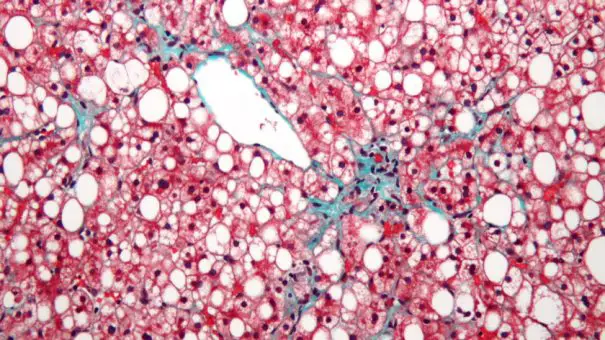
France’s Genfit has said its elafibranor drug as a treatment for non-alcoholic steatohepatitis (NASH) was unable to improve patient outcomes in a phase 3 study, […]
Copyright © 2024 | WordPress Theme by MH Themes