
Sanofi ingredients spin-out EuroAPI rises on market debut
Sanofi has completed the spin-out of its pharma ingredients business EuroAPI, with shares in the new company rising more than 3% on its starting price […]

Sanofi has completed the spin-out of its pharma ingredients business EuroAPI, with shares in the new company rising more than 3% on its starting price […]

Insights captured from KOL interactions represent a new way to identify and map the KOL landscape in which your brand must thrive. KOL identification and […]

Novartis has confirmed it is planning to reduce its headcount as part of a restructuring initiative announced last week, but has declined to put a […]

The US FDA has approved 4 NDAs and 2 BLAs in 2021, leading to treatments for patients and advances in the health care industry The […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The US FDA has approved 5 NDAs and 2 BLAs in 2021, leading to treatments for patients and advances in the health care industry The […]

In an interview with PharmaShots, Suela Sulo, Ph.D., M.S., Director of Global Health Economics, Outcomes Research and Policy at Abbott shared her views on how […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

The US FDA has approved 3 BLAs and 2 NDAs in 2021, leading to treatments for patients and advances in the health care industry The […]

Rapid creation of compliant marketing content is increasingly important and there are ways emerging pharma companies can stay ahead of the game – despite having […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The US FDA has approved 6 NDAs in 2021, leading to treatments for patients and advances in the health care industry The Center for Drug […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

Shots: PharmaShots has started its journey in July 2018 as a concise, real-time news delivery platform and is proud to announce the completion of three […]

The US FDA has approved 6 NDAs and 2 BLAs in 2021, leading to treatments for patients and advances in the health care industry The […]

In an interview with PharmaShots, Ramon Mohanlal, M.D., Ph.D., MBA, Chief Medical Officer and Executive Vice President of Research and Development at BeyondSpring share his […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The US FDA has approved 4 NDAs and 1 BLAs in 2021, leading to treatments for patients and advances in the health care industry. The […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

In an interview with PharmaShots, Neil Solomons, Co-founder and Chief Medical Officer of Aurinia shared his views on the data of Lupkynis (voclosporin) presented at the National […]

In an interview with PharmaShots, Anthony Costello, President, Patient Cloud at Medidata share his views on the launch of Sensor Cloud which is a transformative […]

The US FDA has approved 3 NDAs and 2 BLAs in April 2021, leading to treatments for patients and advances in the health care industry. […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

In an interview with PharmaShots, Dr. Drew Falconer, Neurologist and Director at Inova Parkinson’s and Movement Disorders Center in Fairfax, Va. shares insights on the […]

COVID-19 has massively accelerated the uptake of digital health technology across the whole ecosystem, with important implications for pharma, medical device and insurance tech companies. […]

In an interview with PharmaShots, Dr. Robert Koenekoop, MD, PhD, Professor of Pediatric Surgery, Human Genetics and Adult Ophthalmology at McGill University’s Montreal Children’s Hospital […]

In an interview with PharmaShots, Dr. Saoirse O’Sullivan, Ph.D., Scientific Advisor to Artelo Bio shares insights on new cannabinoids, their relevancy, and the other opportunities that […]

The first quarter of 2021 highlights new approvals, M&A, and the clinical data of COVID-19 vaccines. The companies expand their working in COVID-19 by targeting […]

The US FDA has approved 7 NDAs in Mar 2021, leading to treatments for patients and advances in the health care industry. The Center for […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The US FDA has approved 7 NDAs and 1 BLA in Feb 2021, leading to treatments for patients and advances in the health care industry. […]

The US FDA has approved 7 NDAs and 1 BLA in Feb 2021, leading to treatments for patients and advances in the health care industry. […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

The US FDA has approved 4 NDAs in Jan 2021, leading to treatments for patients and advances in the health care industry. The Center for […]

In an unusual move, 14 major providers — including Trinity Health, CommonSpirit Health and Tenet — have come together to launch a data analytics company. […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

In a recent interview with PharmaShots, Edouard Gasser, CEO of Tilak Healthcare shared his views on OdySight, which is the first medical game to monitor […]

The California-based healthcare organization will work with Accenture and Microsoft to grow and strengthen its cloud capabilities. The enhancements are expected to help Kaiser provide […]

In a recent interview with PharmaShots, Patricia Mae Doykos, Lead, Health Equity Initiative, and Director, BMS Foundation at Bristol Myers Squibb shared her views on […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]

This year J.P. Morgan 39th Annual Healthcare Conference was conducted virtually and we witnessed multiple announcements from numerous Biopharma companies An analysis of events and […]

Life sciences companies are gearing up to enter new markets as they look to secure their positions after a spate of M&A, licensing, and research […]

This article was first published by Elinevan Overbeeke, Sissel Michelsen, Mondher Toumi, Hilde Stevens, Mark Trusheim, Isabelle Huys, and StevenSimoens in Drug Discovery Today under […]

The US FDA has approved 7 NDAs and 2 BLA in Nov 2020, leading to treatments for patients and advances in the health care industry. The Center for Drug […]

The fourth quarter of 2020 contains multiple initiations of clinical trials, big approvals, and numerous deals. COVID-19 related news remains at the peak in this […]

In a recent interview with PharmaShots, Andrew Greenspan, MD, VP Medical Affairs of Janssen Immunology shared his views on Janssen’s commitment to advance research in […]

In a recent interview with PharmaShots, Dr. Riju Ray, MD, PhD, Senior Medical Lead at GSK, leading US medical affairs across asthma and COPD shared […]

This article was originally published by Abdrhman Mahmoud Gamil in World Journal of Advanced Research and Reviews, 2020, 08(03), 337–349 under a Creative Commons Attribution […]

This article was originally published by Andreas Bender and Isidro Cortes-Ciriano in Drug Discovery Today under a Creative Commons license. Although artificial intelligence (AI) has […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

“Lockdown’ declared Collins Dictionary word of the year. The year 2020, well known as COVID-19 year has been a busy year for global pharma and […]

Shots: Thank you for your overwhelming affection and support throughout the year. This year we achieved a massive increase in subscribers PharmaShots mobile app is […]

This article was originally published by Sajna T. in Management Insight, 16(1):38-40 under a creative commons license. Abstract Business enterprises are an important organ of […]

The US FDA has approved 5 NDAs and 1 BLA in Nov 2020, leading to treatments for patients and advances in the health care industry. […]

Mark Schultz and Jaci McDole recently used DrugPatentWatch to complete a report on recent trends in the latest editions of the world health organization essential medicines […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

Prescient hires three new senior members to expand its expertise and keep pace with the company’s growth LONDON, November 3, 2020 – Prescient, a biopharma […]

The US FDA has approved 4 NDAs and 1 BLA in Oct 2020, leading to treatments for patients and advances in the health care industry. […]

We prefer simple flat-rate unrestricted subscriptions — we’ll work on your terms, and with your favorite subscription contract template, to find a solution that fits. If […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]

In a recent interview with PharmaShots, Dr. Lotus Mallbris, the Vice President of Immunology Development at Eli Lilly shares her insights and highlights on Mirikizumab. Shots: @52wks. […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

The third quarter of 2020 was flooded with major acquisitions in the pharma and biotech industry along with multiple approvals. Starting with the latest acquisitions, […]

In a recent interview with PharmaShots, Andrea Stancati, Vice President, Head GI Global Medical Franchise shares his insights and highlights of Entyvio (vedolizumab) Shots: The […]

In a recent interview with PharmaShots, Kai te Kaat, the Vice President, Head of Franchise Oncology, MDx at QIAGEN shares his insights and highlights on QIAprep& Viral […]

The US FDA has approved 6 NDAs in Sept 2020, leading to treatments for patients and advances in the health care industry. The Center for […]

Here is a copy of the talk I gave at the recent Marcusevans 13th Portfolio Management and Pipeline Optimization for Generics. I cover: How to […]

This article not subject to U.S. Copyright. Published 2019 by the American Chemical Society. Noel T. Southall Abstract We report our experience working with the […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]
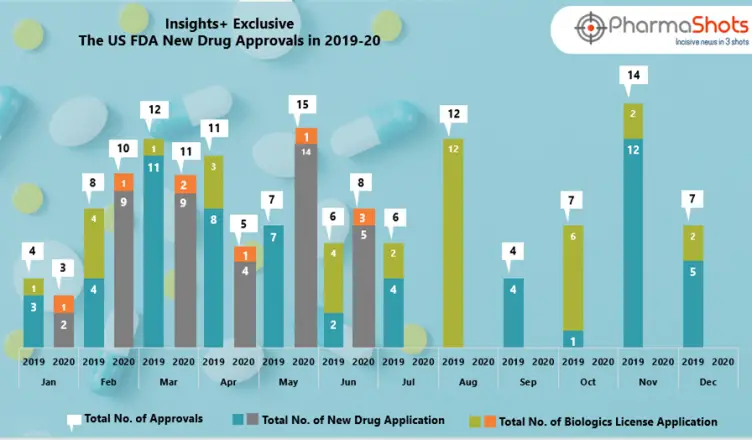
The US FDA has approved multiple NDAs and BLAs in July 2020, leading to treatments for patients and advances in the health care industry. The […]

When drug patents expire, billions of dollars in revenues can disappear overnight as competitors enter the market. Accordingly, many investors focus on drug patents when […]

Just because a drug has received FDA approval does not mean that it is available in the marketplace. But, why would a company go through […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]
Since the virus outbreak in China, all eyes are on the most awaited Covid-19 vaccine offering a glimmer of hope. Abide by social distancing, wearing […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

The USPTO Office of the Chief Economist has releases a comprehensive set of patent litigation data files for public use. The Patent Litigation Dataset has […]

Shots: PharmaShots started its journey in July 2018 as a concise news delivery service and is proud to announce the completion of two years, with […]
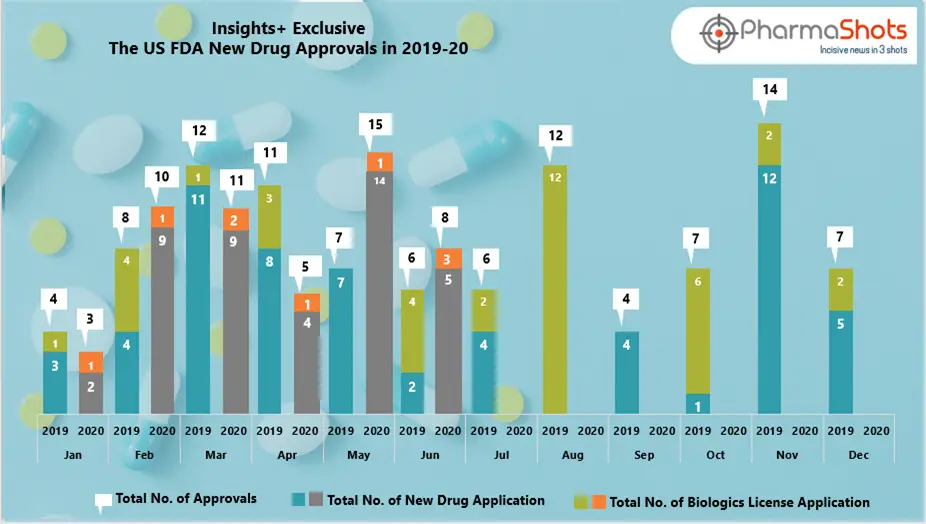
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]

The FDA Orange Book, or Approved Drug Products with Therapeutic Equivalence Evaluations, is the definitive source to link drugs with the patents that protect them. […]

Prescient hires two new senior members to support its fast-growing Advisory business LONDON, July 20, 2020—Prescient, a biopharma product and portfolio strategy firm, announces the […]
Paige is a global digital pathology founded in 2017 by Thomas Fuchs, Dr. Sc., and colleagues from Memorial Sloan Kettering Cancer Center. The company involves […]
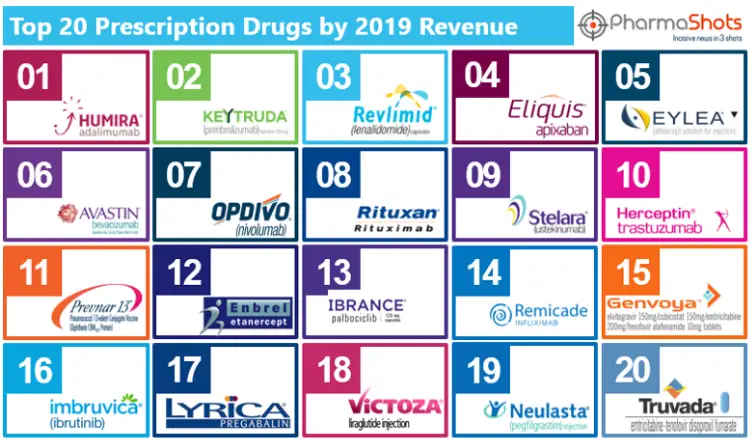
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]

This article was originally published by Syed Sauban Ghani in Eclética Química under a Creative Commons license. ABSTRACT As scientific community worldwide is publishing a […]

This article was originally published by Mohammed M. Alshehri and Mahmoud M. E. Mudawi in the Journal of Pharmaceutical Research International under the terms of […]

Check out this Bloomberg Law article where DrugPatentWatch shows how the number of patent challenges is increasing, while the number of challengers of each patent is […]
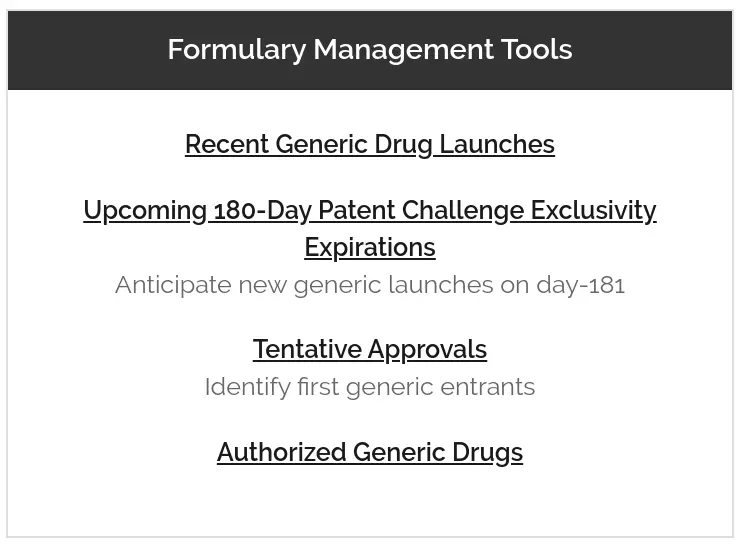
In response to customer requests, we’ve built some new tools for formulary management. You can preview these tools at: Recent Generic Drug Launches Upcoming 180-Day […]
Let DrugPatentWatch build custom dashboards and reports to take the load off of your team. We can ensure the production of accurate, objective, and timely […]

This article was originally published by Hermann Mucke & Peter Mucke in Future Drug Discovery under a Attribution-NonCommercial-NoDerivatives 4.0 Unported License. https://doi.org/10.4155/fdd-2020-0001. Drug developers require […]

In a recent court ruling, the U.S. Court of Appeals for the Federal Circuit (CAFC) denied a petition to rehear arguments regarding the invalidity of […]

One of our clients, the Belarus Republican Scientific and Technical Library recently posted an article on how they support innovation. They open with the statement […]

DrugPatentWatch was used in a recent research study on branded drug sales following generic entry. In this paper, authors Jeffrey Fujimoto, Daniel M Tien, Sophie […]

DrugPatentWatch serves companies across the drug development-delivery sector. A common request among drug manufacturers, payers, and suppliers alike is anticipation of which branded drugs will […]

Changes in drug markets can create growth opportunities for API manufacturers and CDMOs alike. Whether change comes from generic entry, from new formulations of drugs, […]

DrugPatentWatch has deep resources to support patent prosecution and patent litigation, but it is also used by law firms for business development. Find weak claims […]

The launch of a new product can be a daunting task. The main reason this happens is that no matter how many times your company […]

This article was originally published by Garth Boehm, Lixin Yao, Liang Hana, and Qiang Zheng in Acta Pharmaceutica Sinica under a creative commons license. Abstract […]

DrugPatentWatch can help identify generic entrants at the earliest stages Many clients come to DrugPatentWatch seeking to identify generic entrants well before they launch. CDMOs, […]

The latest Swiss Biotech annual report focuses on the theme of shaping change. Change is a theme DrugPatentWatch is very familiar with — whether it […]

This article was originally published by Christopher M. Holman* in Intellectual Property Watch under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. Despite the important role […]

This paper was originally published by Maya Tannoury and Zouhair Attieh in Current Therapeutic Research under a creative commons license. Abstract Emerging markets represent an […]
Copyright © 2024 | WordPress Theme by MH Themes