
Bluebird trumpets long-term data from beta-thalassaemia gene therapy
bluebird bio has presented long-term data from its Zinteglo one-time gene therapy for the blood disorder beta-thalassaemia, as the company continues talks with payers in […]

bluebird bio has presented long-term data from its Zinteglo one-time gene therapy for the blood disorder beta-thalassaemia, as the company continues talks with payers in […]

Sanofi and Regeneron’s Dupixent has had the US market for the treatment of chronic rhinosinusitis with nasal polyps (CRSwNP) mainly to itself since June 2019, […]

A 90-year-old woman has become the first person given a COVID-19 vaccine as part of the UK’s mass vaccination programme. Margaret Keenan was given the […]

The Scottish Medicines Consortium (SMC) has rejected Daiichi Sankyo’s new cholesterol-lowering drug Nilemdo for use by NHS Scotland, saying the evidence backing the drug isn’t […]

Tuesday has been dubbed “V-day” in the UK, when the first doses of Pfizer/BioNTech’s coronavirus vaccine will be distributed to the public outside of a […]

Bureaucracy, political upheaval, and lack of regulations continue to make Latin America a difficult market for European and North American pharma to enter – and […]

In the midst of the jubilation about the UK’s emergency approval of Pfizer/BioNTech’s COVID-19 shot in the UK came the depressingly inevitable round of anti-vaccine […]

Few companies embody the term ‘pharma giant’ as much as Pfizer. Here we take a look at the colourful history of one of the biggest […]

Pfizer and BioNTech are preparing to deliver their COVID-19 vaccine to the UK after the country’s drugs regulator became the first authority in the world […]

AstraZeneca is to sell European rights to its cholesterol drug Crestor (rosuvastatin) to Germany’s Grunenthal. Crestor is a statin and at its peak generated annual […]

Moderna has said it will file for US, European and UK emergency approval of its coronavirus vaccine straight away, after reporting updated phase 3 results […]

The UK has appointed Nadhim Zahawi as vaccine rollout minister as the country prepares for a potential approval of Pfizer/BioNTech’s ground-breaking COVID-19 vaccine. According to […]

The European Union has fined Teva and its neurology unit Cephalon €60.5 million for an elaborate “pay for delay” scheme that prevented cheaper generic competition […]

A new EMA approval has expanded the use of Vertex Pharma’s exon-skipping cystic fibrosis therapy Symkevi to children as young as six if they have […]

UK diagnostics firm Salient Bio has launched a robotics-driven COVID-19 mass testing platform after spinning out from Imperial College. The company says the technology is […]

The UK’s pharma industry trade body has warned that Brexit could derail the government’s efforts to get coronavirus vaccines to the population, but welcomed other […]

COVID-19 is proving to be the perfect storm in terms of the supply of pharmaceutical products. But learning how to overcome the challenges of 2020 […]

The FDA has approved Alnylam’s gene silencing drug Oxlumo, the first treatment for primary hyperoxaluria type 1 (PH1), an ultra-rare and life-threatening genetic disorder. Oxlumo […]

Regeneron’s antibody cocktail is the latest COVID-19 drug to receive Emergency Use Authorization in the US, becoming the first therapy of this kind to become […]

Pfizer and BioNTech have filed for emergency use of their COVID-19 vaccine candidate in the US, paving the way for a launch before year-end. The […]

Doctors in the US are to get another option to treat COVID-19 after the FDA granted an Emergency Use Authorization (EUA) for Eli Lilly’s baricitinib. […]

The inaugural Gene Therapy Medical Affairs Summit is dedicated to exploring the pivotal internal and external role that Medical Affairs plays in delivering gene therapies to patients. Industry pioneers […]

Two months after rejecting Pierre Fabre’s Braftovi for a rare form of metastatic colorectal cancer (mCRC), NICE has changed its mind, and now says the […]

Novartis/Genentech’s eye drug Lucentis could be the next big blockbuster to face competition from cheaper biosimilars after its US patent expired this year – and […]

To mark World Antimicrobial Awareness Week NovaBiotics’s Deborah O’Neil tells us why 2021 might be the year anti-infective drugs finally demonstrate their economic value. COVID-19 […]

UK cost-effectiveness agency NICE has said that Eli Lilly’s Emgality can be made available through the NHS for migraine prevention, the second drug in the […]

Pfizer has begun a pilot delivery programme in the US for its experimental COVID-19 vaccine, as the company seeks to overcome logistical challenges caused by […]

As regional vice president for Northern Europe & Australia, Simon Lem led the digital launch of Vertex Pharma’s novel cystic fibrosis drug Kaftrio. He tells […]

EMA executive director Guido Rasi has ended his second term at the helm of the EU medicines regulator, with Emer Cooke taking the wheel and […]

Hungary is to order a consignment of Russia’s Sputnik V COVID-19 vaccine, as the country struggles to contain the infection rate. According to press reports, […]

Janssen has submitted a new combination regimen for its blockbuster multiple myeloma drug Darzalex for approval with the FDA and EMA, hoping to give the […]

The European Union has ordered 300 million doses of BioNTech/Pfizer’s COVID-19 vaccine following this week’s landmark announcement that it was effective in more than 90% […]

Previously treated patients with chronic lymphocytic leukaemia (CLL) in England will get a chemotherapy-free treatment option after NICE recommended NHS funding for a combination of […]

Eli Lilly has said it will start shipping supplies of its COVID-19 drug bamlanivimab immediately, after claiming emergency-use authorisation (EUA) for the antibody. The AbCellera-partnered […]

The UK’s NHS is ready to start rolling out a coronavirus vaccine, likely beginning in the new year, according to health secretary Matt Hancock. In […]

Unless president Donald Trump is successful with legal challenges, Democratic rival Joe Biden looks to have won the White House in last week’s election but […]

The Scottish Medicines Consortium (SMC) has ruled that there’s not enough evidence to support NHS funding of Vifor Pharma’s Veltassa for excess potassium in the […]

The FDA may have been minded to approve Biogen and Eisai’s Alzheimer’s candidate aducanumab, but its clinical advisors have little doubt that the evidence for […]

The UK’s drug cost-effectiveness body NICE has launched a public consultation, presenting the case for change about how it assesses medicines, medical devices and diagnostics. […]

COVID-19 has been a shock to the system for every industry, not least pharma, but the sector is already looking at new ways to build […]

Pharma may have avoided a worst case scenario amid the political upheavals in the US elections with the threat of tough drug price legislation lowered, […]

Regulatory reviews of Biogen’s Alzheimer’s drug aducanumab are now ongoing on both sides of the Atlantic, but debate is still ongoing about whether the data […]

Novo Nordisk’s Saxenda has been recommended by NICE as a treatment for obesity, ending a 10-year drought in new drug therapies for weight management. The […]

With exclusive data from Freedom of Information (FOI) requests sent to the National Institute for Health and are Excellence (NICE), Leela Barham takes a look […]

The US government said it will buy 300,000 doses of Eli Lilly’s COVID-19 therapy bamlanivimab for $375 million if the drug gets Emergency Use Authorisation […]

Pfizer could be just a few months away from getting FDA approval for its JAK1 inhibitor abrocitinib in atopic dermatitis, a drug that CEO Albert […]

Russia’s sovereign wealth fund has filed applications with the World Health Organization (WHO) that if approved could see the Sputnik V coronavirus vaccine backed for […]

US trials of AstraZeneca’s experimental COVID-19 vaccine AZD1222 have been cleared to restart by the FDA, several weeks after testing was suspended following a serious […]

Gilead’s Veklury (remdesivir) has become the first COVID-19 drug approved in the US following a nod from the FDA. As an antiviral drug, remdesivir works […]

Zosano Pharma has been hit by an FDA rejection of its marketing application for migraine drug Qtrypta, asking for new bioequivalence data. Qtrypta takes the […]

Pfizer now has hundreds of thousands of doses of BioNTech’s COVID-19 vaccine coming off the production line at a facility in Belgium, ready for delivery […]

Novartis’ near-$10 billion takeover of The Medicines Company last year was focused mainly on one asset – cholesterol-lowering drug inclisiran – and the Swiss pharma […]
Eli Lilly is to buy the private biotech Disarm Therapeutics, which is working on a new class of disease-modifying drugs for neurological diseases, in a […]

An antibody cocktail from Regeneron has been approved by the FDA as the first treatment for Ebola, offering hope that a similar approach could be […]

Scynexis has filed for FDA approval of its novel drug ibrexafungerp which – if approved – would be the first broad-spectrum antifungal to reach the […]

Regeneron has followed Eli Lilly in asking the FDA for emergency approval of its COVID-19 antibody therapy, shortly after the drug was thrust into the […]
The European Commission has taken its advance orders for potential COVID-19 vaccines to more than 1.1 billion, after signing a supply deal for up to […]

A few months into the pandemic, the world is now moving beyond managing the crisis, with more focus on addressing the collateral damages and shaping […]

The FDA may have been blocked from issuing stricter guidelines for emergency approval of COVID-19 vaccines by the Trump administration, but it has still managed […]

There are a host of new, previously unimaginable tools and techniques – from analytics to robotic process automation (RPA) and artificial intelligence (AI) – available […]

As European regulators begin a rolling review of AstraZeneca’s COVID-19 vaccine, the focus in the UK has shifted to the government’s plans to distribute any […]

Bluebird bio could be just a few months away from approval of its gene therapy for rare disease cerebral adrenoleukodystrophy (CALD) in the EU, after […]

Italian biotech Cassiopea has won US approval for Winlevi, a topical therapy it says is the first new approach to treating acne in nearly 40 […]

The US Department of Justice has charged Teva with conspiring with other pharma companies to fix prices for generic drugs. According to an indictment filed […]

The FDA has allowed a blood plasma treatment for COVID-19 to be administered under an Emergency Use license – but some have decried the move […]

It’s mission accomplished for Novartis after the FDA approved ofatumumab for multiple sclerosis, completing a project where the former cancer drug has been repurposed. The […]

NICE has recommended regular NHS funding for Roche’s Polivy in certain lymphoma patients in final draft guidance, overturning a previous rejection. The new guidance recommends […]

Catch up on the latest coronavirus vaccine and digital tech news in our roundup of this week’s biggest stories. Consumer advocacy group Public Citizen has […]
J+D Forecasting strike the balance between simplicity and complexity when modelling for the liquid tumour market The Challenge Identify a clear, transparent, and logical way […]

The FDA has started a speedy review of Regeneron’s ANGPTL3-targeting antibody evinacumab for a rare, inherited disorder that dramatically raises the risk of heart disease. […]

The FDA has approved a new therapy for the rare muscle wasting disease Duchenne muscular dystrophy (DMD) as Japan’s NS Pharma takes on Sarepta and […]

The US government has signed another big coronavirus vaccine supply deal, snapping up 100 million doses of Moderna’s mRNA jab for a little over $1.5 […]

The FDA has started its review of Israeli biotech Protalix BioTherapeutics and partner Chiesi’s Fabry disease therapy pegunigalsidase alfa, setting up a possible approval by […]

In our latest UK Leaders article Pinder Sahota, UK general manager at Novo Nordisk, tells us how he hopes to bring the NHS and pharma […]

Roche is hoping to undercut hugely expensive rivals after the FDA approved its oral spinal muscular atrophy (SMA) drug Evrysdi (risdiplam). Evrysdi is the third […]

Pharma’s reputation has soared due to the COVID-19 pandemic – but coronavirus is starting to bite companies’ sales. Find out more in our roundup of […]

DBV Technologies’ long and tortuous path to an FDA verdict on its Viaskin Peanut allergy shot has led to another dead end. The France-headquartered biotech […]
New research looks at the factors that speed up and slow down HTA appraisals for rare disease medicines across Europe. Rare diseases drugs have always […]
Cell and gene therapies offer some of the most groundbreaking advancements in patient care the pharma industry has ever seen. However, to fully realise the […]

NICE has said a combination of Merck KGaA’s Bavencio and Pfizer’s Inlyta should be available as a first line option for kidney cancer patients for […]

Vaccines, AI-driven drug discovery and llamas all hit the headlines this week as the search for a way to end the COVID-19 pandemic continues. Here […]
Roche’s checkpoint inhibitor Tecentriq has been cleared for another new use in the US – in melanoma – but could struggle to displace rival drugs […]

The UK government has signed its fourth coronavirus vaccine deal, snapping up 60 million doses of an experimental shot being developed by Sanofi and GlaxoSmithKline. […]
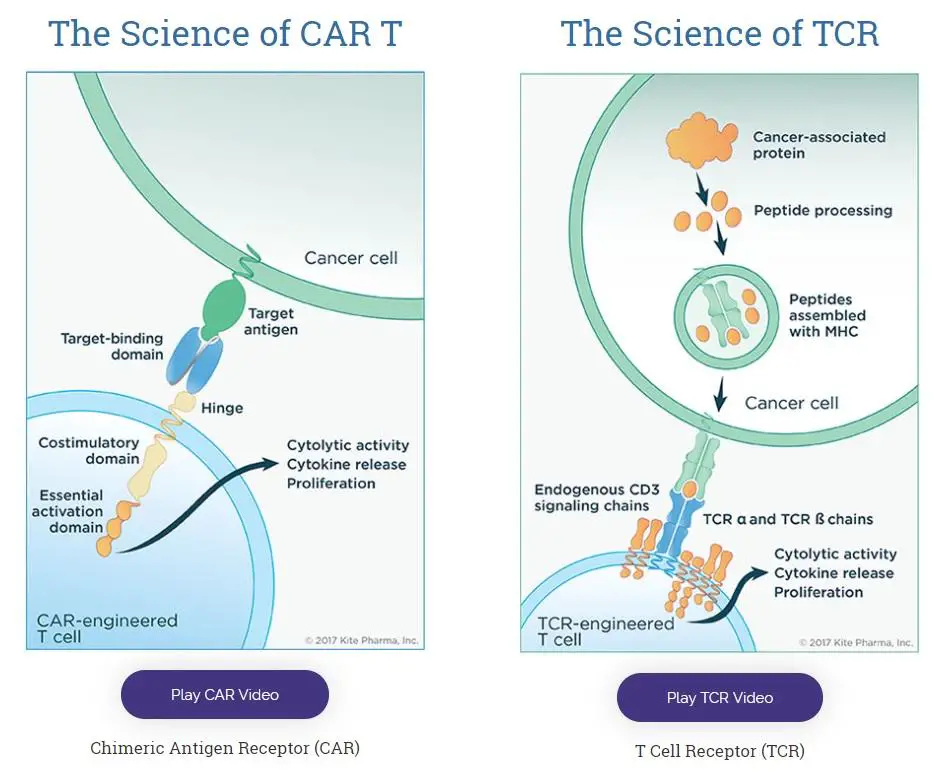
Gilead’s $11.9 billion purchase of Kite Pharma in 2017 didn’t follow the script, with two big write-downs in the value of the asset since then, […]

GlaxoSmithKline is closing in on EU approval for its first-in-class BCMA-targeting drug Blenrep, after getting a green light from the EMA’s human medicines committee. Blenrep, […]

After decades of turmoil, Iraq’s healthcare sector is growing fast – but the country still holds some unique challenges for companies looking to enter its […]

With coronavirus vaccine trials finally showing positive results, analysts are starting to wonder what access challenges might lie ahead. We highlight the top R&D, digital […]

The US government has agreed to pay Pfizer and BioNTech $1.95 billion to secure access to 100 million doses of their coronavirus vaccine on approval, […]

David Peacock has worked across the world for MSD and other companies, and has now taken the helm of MSD’s UK & Ireland operations. In […]
Concerns have emerged about access to some potential COVID-19 vaccines after pharma executives from three companies said they expect to make profits from their products […]

The COVID-19 pandemic has bit into sales at Novartis, the big Swiss pharma said in second quarter results, after orders fell back following a period […]

Coronavirus vaccine trials continue – but experts warn that it’s too early to get hopes up. Meanwhile, the rise in digital health looks like it’s […]
NICE has recommended regular NHS funding for Astellas’ Xospata (gilteritinib) monotherapy for certain adult patients with acute myeloid leukaemia. In final draft guidance NICE said that the drug is recommended […]

The UK’s regulator has granted early access to Alnylam’s ultra-rare disease drug lumasiran, allowing patients with primary hyperoxaluria type 1 to gain access ahead of […]

An FDA advisory committee has narrowly voted in favour of approval of Mallinckrodt’s terlipressin for hepatorenal syndrome type 1 (HRS-1), after the agency questioned the […]
Copyright © 2024 | WordPress Theme by MH Themes