
MorphoSys slims down, narrowing its focus to cancer
MorphoSys seemingly inexorable shift into an oncology pure-play has continued with an out-licensing deal for two programmes in the kidney disease and immunology categories to […]

MorphoSys seemingly inexorable shift into an oncology pure-play has continued with an out-licensing deal for two programmes in the kidney disease and immunology categories to […]

MorphoSys is cutting the drug discovery and R&D work of Constellation Pharmaceuticals, a company it acquired lats year in a $1.7 billion deal. This consolidation […]

The European Commission has approved a BioMarin Pharmaceutical drug that treats achondroplasia, a rare genetic disorder that slows bone growth and is the most common […]

Ascendis’ Skytrofa (lonapegsomatropin-tcgd) Receives the US FDA’s Approval for Pediatric Growth Hormone Deficiency Published: Aug 27, 2021 | Tags: Ascendis, Skytrofa, lonapegsomatropin-tcgd, US, FDA, Approval, […]

Incyte and MorphoSys’ antibody therapy tafasitamab has been approved in Europe, promising to provide a more convenient alternative to expensive and cumbersome CAR-T therapies for […]

Shots: The conditional approval is based on the P-II L-MIND study evaluating the safety and efficacy of tafasitamab + lenalidomide in patients with r/r DLBCL […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

Tonix Pharmaceuticals Presents Results of TNX-102 SL in P-III RELIEF study for the Management of Fibromyalgia at ASCP 2021 Published: June 4, 2021 | Tags: […]

Shots: MorphoSys to acquire all of the outstanding shares of Constellation’s common stock for $34.00/ share in cash making a total equity value of $1.7B. […]

Shares in German biotech MorphoSys have gone on the slide after it announced a $1.7 billion deal to buy its US rival Constellation Pharma and […]

MorphoSys is acquiring Constellation Pharmaceuticals and its late-stage myelofibrosis drug in a deal that values the epigenetics biotech at $1.7 billion. Germany-based MorphoSys will finance […]

AzurRx BioPharma Reports Positive Interim P-II Data of MS1819 + PERT for Cystic Fibrosis Patients with Severe Exocrine Pancreatic Insufficiency Published: May 14, 2021 | […]

Shots: The first patient has been dosed in the P-III frontMIND study evaluating tafasitamab + lenalidomide + R-CHOP vs R-CHOP alone as a 1L treatment […]

Shots: The first patient has been dosed in the P-IIII inMIND study assessing the efficacy and safety of tafasitamab vs PBO in combination with lenalidomide […]
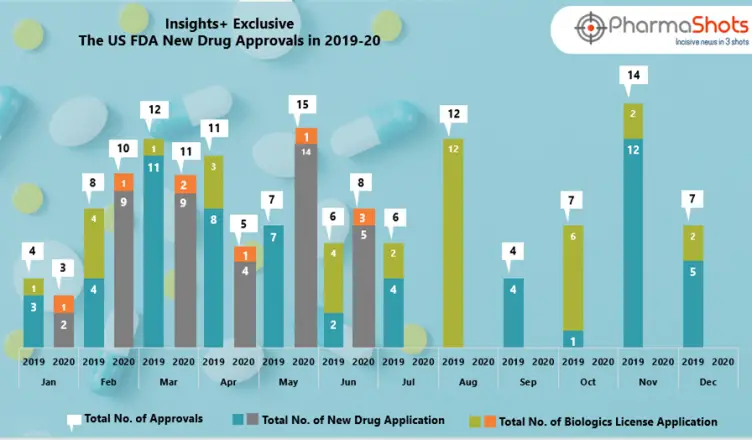
The US FDA has approved multiple NDAs and BLAs in July 2020, leading to treatments for patients and advances in the health care industry. The […]

Shots: Morphic will receive a $20M as license fee and is eligible to receive milestones and royalties on sales of any commercialized therapy while AbbVie […]
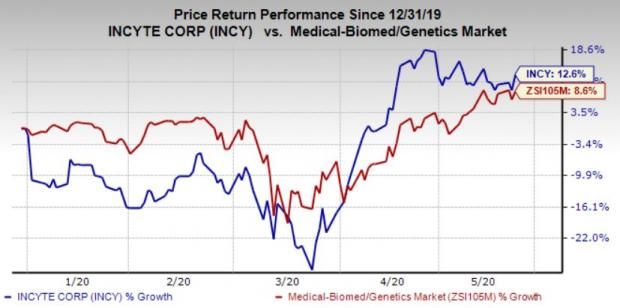
The FDA approved Monjuvi for second-line diffuse large B-cell lymphoma. Though the drug has the same antigen target as approved CAR-T cell therapies, it is […]

Incyte and Morphosys’ Monjuvi antibody therapy has been approved for certain patients with lymphoma, as the companies hope to provide a more convenient alternative to […]

Shots: The BLA submission is based on L-MIND P-II trial data results assessing Tafasitamab + lenalidomide in patients with r/r DLBCL and retrospective observational matched […]
Copyright © 2024 | WordPress Theme by MH Themes