
FDA okays Mylan insulin drug as first ‘interchangeable’ biosimilar
Generic drugmaker Mylan has become the first company to secure FDA approval for a biosimilar product that is considered completely interchangeable with the reference product […]

Generic drugmaker Mylan has become the first company to secure FDA approval for a biosimilar product that is considered completely interchangeable with the reference product […]

“Lockdown’ declared Collins Dictionary word of the year. The year 2020, well known as COVID-19 year has been a busy year for global pharma and […]

The global generics pharmaceutical market has become an intimidating segment of the pharmaceutical drug market which continuously growing and increasing with its accessibility & benefits […]

Mylan’s $12 billion takeover of Pfizer’s Upjohn unit has been cleared by the US authorities, but on the condition that the two companies divest various […]

1. Taysha Gene Therapies’ TSHA-101 Receives the US FDA’s Orphan Drug Designation and Rare Pediatric Disease Designation for GM2 Gangliosidosis Published: Aug 27, 2020 | […]

Shots: The companies launch Nepexto, a biosimilar referencing Amgen’s Enbrel, and is indicated for the mod. to sev. RA, JIA from the age of 2yrs., […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]
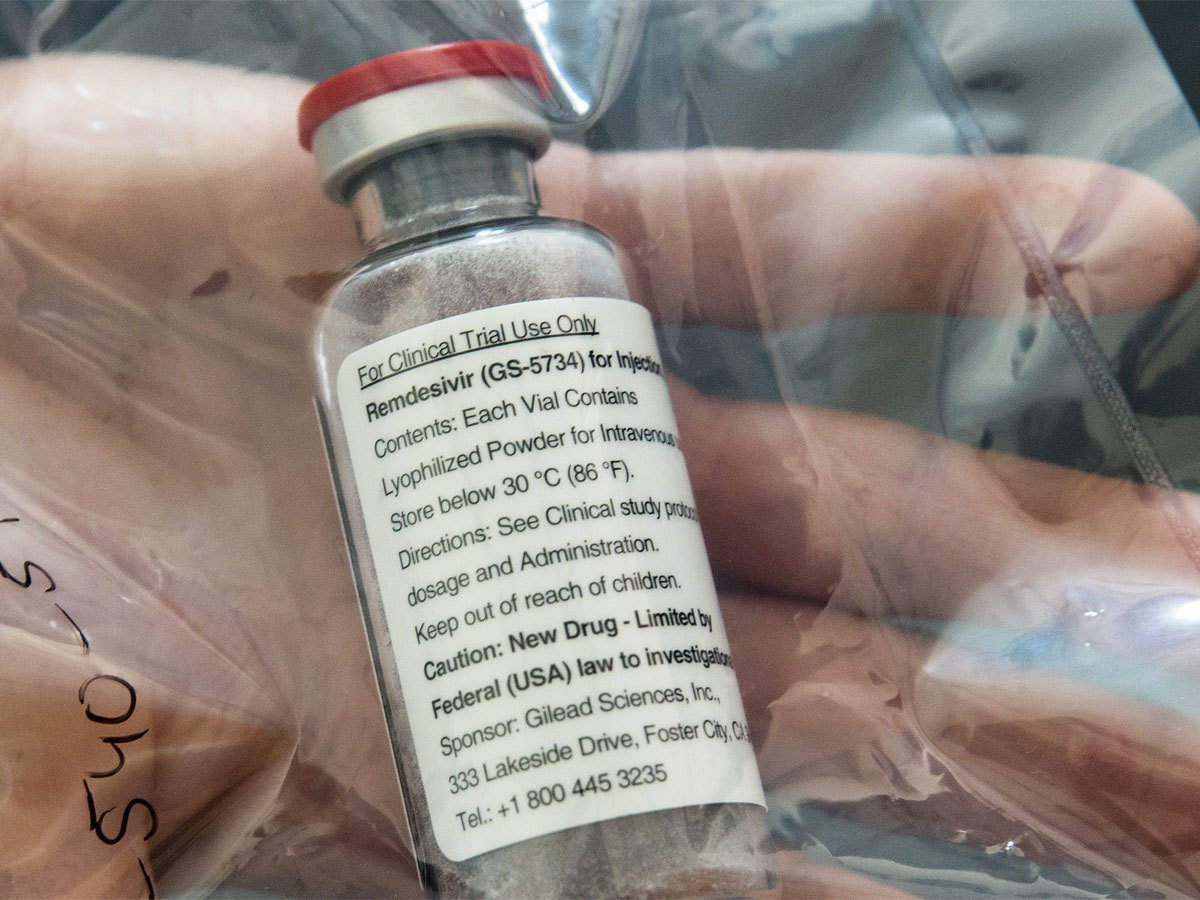
Shots: Mylan launches Desrem (remdesivir) addressing the unmet needs amid COVID-19. The drug is approved for the treatment of suspected or laboratory-confirmed incidences of COVID-19 […]
Copyright © 2024 | WordPress Theme by MH Themes