
Cumulus’ gamified dry EEG system tops lab monitoring in trial
An at-home electroencephalogram (EEG) headset used alongside game-like cognitive tasks developed by UK company Cumulus Neuroscience has been shown to be more effective than lab-based […]

An at-home electroencephalogram (EEG) headset used alongside game-like cognitive tasks developed by UK company Cumulus Neuroscience has been shown to be more effective than lab-based […]

Healthcare technology firm Koneksa will carry out a study to evaluate whether digital biomarkers can be used to track the progression of Parkinson’s disease – […]

Beleaguered Biogen has been handed some encouraging news from the FDA, after the regulator agreed to complete its review of amyotrophic lateral sclerosis (ALS) therapy […]

Despite reporting proof-of-concept results in a mid-stage trial, Kyowa Kirin has decided to discontinue development of its Parkinson’s disease candidate KW-6356, a drug in the […]

Memory Health CEO Edward Shehab talked about taking a preventive approach to neurodegenerative conditions.

AbbVie is pulling out of its collaboration with Alector on the development of Alzheimer’s disease candidate AL003, which aims to tackle the neurodegenerative disease by […]

The FDA has started an accelerated review of Eisai and Biogen’s a new amyloid-targeting antibody lecanemab for Alzheimer’s disease, with a 6 January deadline that […]

Biogen has ducked out of a four-year-old collaboration with Karyopharm on a drug candidate for the neurodegenerative disease amyotrophic lateral sclerosis that could have been […]

Amylyx Pharmaceuticals’ efforts to bring its amyotrophic lateral sclerosis therapy AMX0035 to market in the US have run into some roadblocks, so an approval by […]

Biogen has added a preclinical-stage drug candidate for Parkinson’s disease via a $722.5 million licensing agreement with Alectos Therapeutic, as it continues to rebuild a […]

Cumulus Neuroscience has started a pair of clinical trials to gauge the value of a panel of physiological and digital biomarkers in tracking the course […]

AbbVie has filed for approval of its therapy for Parkinson’s disease, ABBV-951, which promises continuous control of symptoms and would be an alternative to current […]

Considerable scientific progress across neurological diseases and disorders was reported at the recent 74th American Academy of Neurology (AAN) Annual Meeting. What may be important […]

Biogen has licensed rights to a digital therapeutic (DTx) developed by MedRhythms which combines sensors, software, and music to help people with multiple sclerosis tackle […]

The US FDA has authorised a new test that can detect amyloid plaques in early-stage Alzheimer’s disease without the use of PET scans, which expose […]

Biogen has given up on seeking approval for its Alzheimer’s disease therapy Aduhelm in the EU, saying that discussions with EU regulators had made it […]

Diagnosing Parkinson’s in the earliest stages before symptoms become serious is a major challenge in medicine, and digital health company PreciseDx thinks it may have […]

One of the leading companies in the bid to bring a new class of alpha-synuclein targeting drugs to market for Parkinson’s disease – AbbVie – […]

Oligonucleotide Therapeutics and Delivery Conference 2022 Date: 21st – 22nd September 2022 Location: London, UK Website: www.oligonucleotide.co.uk/pharmaphorum Advancements in extra-hepatic delivery of oligonucleotides The Oligonucleotide […]

Patients with relapsing-remitting multiple sclerosis (RRMS) in England, Wales and Northern Ireland can now access treatment with Biogen’s oral therapy Vumerity, after NICE recommended the […]

GripAble has raised $11 million in first-round financing that will be used to advance its digital health approach to rehabilitation for people with neurological and […]

The largest-ever study of genetics in Alzheimer’s disease patients has identified 42 new genes that appear to be linked to the neurodegenerative disorder. The new […]

By a narrow margin, an FDA advisory committee has voted against approval of Amylyx’ experimental medicine for amyotrophic lateral sclerosis until the company can provide […]

After months of debate about the validity of its phase 3 clinical trials for Alzheimer’s therapy Aduhelm, Biogen has published the results in a peer-review […]

Japanese pharma Eisai has backed away from its 50:50 profit-sharing arrangement with Biogen for troubled Alzheimer’s therapy Aduhelm, handing over full responsibility to its US […]

With prospects of an early, accelerated approval for Roche’s Alzheimer’s disease candidate gantenerumab looking diminished, the drugmaker has launched a new four-year trial in the […]

Four years after spinning out from Belgium’s UCB, Syndesi Therapeutics and its SV2A modulator programme has been bought by AbbVie for $130 million upfront. The […]

Swiss digital health company MindMaze has raised $105 million to help take its digital therapeutics (DTx) for neurological diseases like stroke onto the global stage, […]

The financing round was led by Concord Health Partners, with AlbaCore Capital Group and Hambro Perks also contributing. The latest haul follows a $125 million […]

By JESSICA DaMASSA, WTF HEALTH You may know the term “digital therapeutics,” but how about the specialized category of “digital neuro-therapeutics”? MindMaze, which has developed […]

Japanese drugmaker Eisai has published comments filed with the US Centers for Medicare & Medicaid Services over its restrictive coverage proposal for beleaguered Alzheimer’s drug […]

Eli Lilly has backed away from plans to file Alzheimer’s drug donanemab in the first quarter of this year, saying the proposal to limit reimbursement […]

Biogen and Eisai have increased the earning potential of their Alzheimer’s drug Aduhelm by expanding the post-marketing trial of the drug required by the FDA […]

Biogen and Eisai head towards the end of the year with some much-needed good news in their Alzheimer’s programmes, as the FDA awards a fast-track […]

Biogen has cut the list price of its Alzheimer’s disease therapy in half from $56,000 to $28,200 a year, before the outcome of an ongoing […]

The EMA’s human medicines committee has said it cannot approve Biogen and Eisai’s Alzheimer’s disease therapy Aduhelm, saying it was unconvinced by the data submitted […]

Scientists in the US have found that people who use Viagra for erectile dysfunction seem to have a lower risk of developing Alzheimer’s disease – […]

Novartis has licensed a potential drug for Parkinson’s from Belgium’s UCB that it thinks could be the first oral, disease-modifying drug for the disease – […]

A year ago, Swiss biotech Santhera was slashing staff to cut costs after it abandoned lead Duchenne muscular dystrophy (DMD) therapy idebenone. Now, it’s ready […]

Biogen and Eisai have said that the EMA’s human medicines committee looks unlikely recommended approval of their Alzheimer’s drug Aduhelm when it comes up for […]

AbbVie has reported that a subcutaneous infusion of Parkinson’s disease candidate ABBV-951 was more effective than standard oral therapy in a phase 3 trial, paving […]

Eli Lilly has followed through on its plan to file for approval of its Alzheimer’s disease candidate donanemab, and is planning to take on Biogen […]

A hospital trust has become the first in the UK to deploy a gaming system developed by Swiss company MindMaze that is intended to help […]

A drug originally developed for high blood pressure could find a new use in the treatment of Huntington’s disease, thanks to an artificial intelligence-based drug […]

Pfizer’s tyrosine kinase inhibitor Inlyta is already a cornerstone of treatment for some forms of cancer, but could have potential as a therapy for Alzheimer’s […]

Biogen and Eisai’s first Alzheimer’s disease drug Aduhelm is facing an uphill battle in the US market, but the partners are forging ahead with their […]

Global Gene’s Craig Martin introduces a new patient identification initiative that strives to improve diagnosis and access to clinical trials for all patients with rare […]

Danish drugmaker Lundbeck has teamed up with Inscopix – the developer of a technology that can film neurons in the brain in real time – […]

Just a few months ago, Amylyx was talking about having to complete another clinical trial before it could file for FDA approval of its drug […]

The first patient has received a low dose of Beckley Psytech’s psychedelic medicine psilocybin in a clinical trial for short-lasting unilateral neuralgiform headache attacks (SUNHA), […]

A digital health company focusing on neurodegenerative disorders – Neuroglee Therapeutics – has raised $10 million in first-round funding to roll out a virtual neurology […]

Servier and Neurochlore have been forced to admit defeat in their bid to bring the first drug to market for people with autism spectrum disorder […]

Shares in AC Immune leaped today after the company said its tau-targeting Alzheimer’s disease candidate semorinemab hit one of its objectives in a phase 2 […]

UK psychedelic medicine specialist Beckley Psytech has raised an impressive £58 million (around $80 million) in an oversubscribed second-round financing that will help fund clinical […]

By MARTIN A. SAMUELS A 35 year old woman complains of weakness of the right side of her face and pain behind the right ear. […]

The FDA’s approval of Biogen’s Aduhelm for Alzheimer’s disease emboldened Eli Lilly to move forward plans to file its own candidate donanemab later this year, […]

AbbVie and Alphabet spin-out Calico Life Sciences have agreed to fund their seven-year-old partnership seeking treatments for diseases of ageing with another $500 million apiece. […]

AbbVie and Alphabet spin-out Calico Life Sciences have agreed to fund their seven-year-old partnership seeking treatments for diseases of ageing with another $500 million apiece. […]

Biogen has reported $2m in sales for newly-launched Alzheimer’s therapy Aduhelm that came in below its expectations, prompting the company to issue a rebuke to […]

Phase 3 results with Roche’s amyloid-targeting Alzheimer’s drug gantenerumab may not be due until the second half of last year, but the company is already […]

Bayer subsidiary BlueRock Therapeutics has been granted a fast-track review by the FDA for DA01, its stem cell-based therapy for Parkinson’s disease which is currently […]

French drugmaker Ipsen has made another foray into the Parkinson’s disease category, licensing rights to an oral dopamine D3 receptor antagonist from Sweden’s IRLAB for […]

Three FDA figures at the centre of the fallout over the approval Biogen and Eisai’s Alzheimer’s disease therapy Aduhelm have defended their actions, arguing that […]

Diagnosing Alzheimer’s disease is still a challenge, particularly in its earliest stages, but a new study suggests that subtle changes in behaviour whilst driving could […]

Biogen has moved to shore up its besieged multiple sclerosis franchise by licensing a BTK inhibitor from Chinese biotech InnoCare currently in mid-stage clinical testing. […]

The US government has started a review of how Biogen and Eisai’s pricey new Alzheimer’s disease therapy Aduhelm will be covered by Medicare, to see […]

One of the criticisms levelled at the FDA over its approval of Biogen and Eisai’s Alzheimer’s disease drug Aduhelm was its decision to clear use […]

The Institute for Clinical and Economic Review (ICER) was very damning of Biogen’s new Alzheimer’s disease therapy Aduhelm when it issued its first report on […]

The Institute for Clinical and Economic Review (ICER) was very damning of Biogen’s new Alzheimer’s disease therapy Aduhelm when it issued its first report on […]

Biogen has four other Alzheimer’s candidates in its clinical pipeline behind recently-approved Aduhelm, but one of them – anti-tau antibody gosuranemab – has just been […]

With the first patient set to receive Biogen’s new Alzheimer’s drug Aduhelm in Rhode Island today, US consumer advocacy organisation Public Citizen is deeply unhappy […]

The fallout over the FDA’s decision to approved Biogen’s Alzheimer’s therapy Aduhelm continues to be felt, with a third member of its advisory committee resigning […]

Two members of an FDA advisory committee have now resigned in protest over Monday’s approval of Biogen’s Alzheimer’s disease drug Aduhelm despite an overwhelming vote […]

The unexpected approval and lofty pricing of Biogen’s Alzheimer’s therapy Aduhelm is clearly the biopharma event of the year so far, but what are the […]

Bayer subsidiary BlueRock Therapeutics has dosed the first Parkinson’s disease patient in a phase 1 trial of its cell-based therapy DA01, which aims to replenish […]

The FDA “has failed in its responsibility to protect patients and families from unproven treatments with known harms” in approving Biogen’s Alzheimer’s disease drug Aduhelm. […]

Biogen and Eisai have defied the naysayers and secured an historic FDA approval for their amyloid-targeting antibody aducanumab as the first-ever disease-modifying treatment for Alzheimer’s […]

Digital health player Akili has raised $110 million fourth-round funding, backed by a number of pharma groups, as it works towards extending its product range […]

NICE has issued a final “no” to Bristol-Myers Squibb’s oral MS drug Zeposia (ozanimod), in a decision that prevents access for patients in England and […]

Exscientia has advanced a drug candidate for Alzheimer’s disease into human testing, the third clinical-stage project for the UK specialist in artificial intelligence (AI) based […]

Roche’s Enspryng has been recommended for approval in the EU for treating neuromyelitis optica spectrum disorder (NMOSD), extending the treatment options for people with the […]

Switzerland’s neurology specialist Newron must hold off starting a phase 3 study of its schizophrenia drug evenamide to complete a small study to show safety […]

The FDA got too close to Biogen during the development of the Alzheimer’s drug aducanumab, according to its own expert advisers who are urging the […]

Roche has stopped dosing in a phase 3 trial of tominersen, a potential Huntington’s disease (HD) drug developed with Ionis, after a pre-planned review by […]

COVID-19 has increased demands on informal carers and severely increased demands on their mental, physical and financial wellbeing, according to a global study. Embracing Carers […]

Injected neurotoxins can be incredibly potent in treating spasticity from neurological diseases, but the field doesn’t always get the attention it needs. Ipsen’s John Chaddock […]

Shares in Swiss biotech AC Immune have risen sharply after it said a vaccine in development for Alzheimer’s disease (AD) hit the mark in a […]

Scotland’s drugs cost-effectiveness body has recommended Bristol-Myers Squibb’s Zeposia multiple sclerosis pill, setting a potential north-south access divide following a draft rejection in England by […]

Adamas Pharmaceuticals’ Parkinson’s drug Gocovri has a new indication as an adjunctive treatment for levodopa/carbodopa in patients with Parkinson’s disease experiencing ‘off’ episodes. The new […]

The FDA has delayed its decision on aducanumab, the troubled Alzheimer’s drug from Biogen and Eisai by three months, raising hopes that it could be […]

New findings from a digital study have challenged the belief that stress is a significant factor in triggering migraine attacks. Results came from a study […]

Belgian drugmaker UCB has been steadily building its digital health expertise for several years, and that includes nurturing new projects. One of those is now […]

When Eli Lilly reported results for donanemab in Alzheimer’s disease earlier this week it was hailed as a rare win for the amyloid hypothesis, although […]

US biotech Atalanta Therapeutics has come out of stealth mode backed with $110m from Biogen and Roche, who have also signed separate partnerships to develop […]

By Boris Goldstein, Co-Founder & Executive Chairman at Brain Scientific COVID-19 is known for its impacts on a patient’s respiratory system, but it has also […]

There’s a new player in the neurology and mental health drugs market after Italy’s Angelini Pharma merged with Switzerland’s Arvelle Therapeutics in a deal worth […]

Biotech Clene Nanomedicine has gone public with a mission to use nanotherapeutics that will use gold to treat devastating neurological diseases including Parkinson’s disease. Over […]

A serotonin sensor designed using Artificial Intelligence (AI) could help scientists study sleep and mental health and potentially find new neurology drugs. The US National […]

Novartis is to buy neuroscience drugs firm Cadent Therapeutics in a deal worth up to $770 million. The big Swiss pharma already has a presence […]

The 9th Neurodegenerative Drug Development Summit is the industry’s definitive and unrivalled forum focused on revealing hot and promising pockets of innovation in drug discovery for neurodegenerative […]

Eli Lilly has acquired Prevail, a biotech focusing on gene therapies for neurodegenerative diseases including Parkinson’s, in a deal potentially worth more than $1 billion. […]

Swiss biotech Noema has raised 54 million Swiss francs ($59m) to develop four neurological disorder drugs licensed in from Roche. The company is developing four […]

Japanese drugmaker Eisai has teamed up with UK biotech Wren Therapeutics on an R&D programme seeking drugs for neurodegenerative disorders. The two companies will focus […]

With the prospects for its Alzheimer’s drug aducanumab still up in the air, Biogen has moved to shore up its late-stage pipeline with a $3 […]

The FDA may have been minded to approve Biogen and Eisai’s Alzheimer’s candidate aducanumab, but its clinical advisors have little doubt that the evidence for […]

As an influential panel of FDA-appointed experts prepares its verdict on Biogen’s Alzheimer’s drug, a little known China-based pharma is hoping to succeed where big […]

Novartis’ injectable migraine prevention antibody Aimovig has been shown to be more effective than topiramate – a go-to oral therapy for people with chronic migraine […]

An implantable neurostimulation migraine device from Salvia BioElectronics has gained Breakthrough Device Designation from the FDA. Traditional neurostimulation systems are not designed to be compatible […]

UK cost-effectiveness agency NICE has backed Novartis’ Mayzent for secondary progressive multiple sclerosis (SPMS), after turning it down earlier this year in draft guidance. The […]

Axovant has said it plans to continue developing its Parkinson’s Disease gene therapy after reporting supportive data from a small cohort of patients from a […]
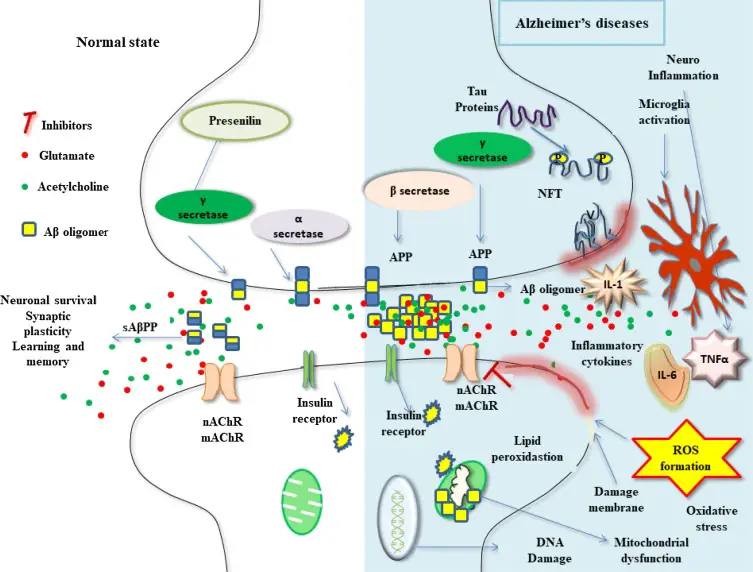
Recent Advances in Alzheimer’s Disease Prevention A prominent neurologist reviews what is known (and not known) about recent evidence-based advances in Alzheimer’s Disease prevention. The […]

Mr. James Evans suffered from serious back pain and he was taking pain injections to ease his pain. After the prolonged use of pain injections, […]

It’s mission accomplished for Novartis after the FDA approved ofatumumab for multiple sclerosis, completing a project where the former cancer drug has been repurposed. The […]

The FDA has approved Roche’s satralizumab for the rare autoimmune disorder Neuromyelitis Optica Spectrum Disorder (NMOSD), under the brand name Enspryng. Roche is taking on […]

MdDS Makes You Feel Like You’re Still on the Boat MdDS is an under recognized neurologic disorder where the rocking sensation you experience after getting […]

Bayer is to buy UK-based KaNDy Therapeutics in a deal worth more than $800 million, adding the biotech’s menopause drug to its women’s health pipeline. […]

Roche is hoping to undercut hugely expensive rivals after the FDA approved its oral spinal muscular atrophy (SMA) drug Evrysdi (risdiplam). Evrysdi is the third […]
Biogen has bought a licence to co-develop and co-market potential Parkinson’s disease drugs with US biotech Denali, in a deal worth more than $2.1 billion. […]

Roche already has one Alzheimer’s candidate in its pipeline targeting tau, but has added a second via a deal with Belgian drugmaker UCB worth up […]

Shots: The study involved 1402 patients across 3 cohorts: a) 81 patients in Arizona (34 with AD and 47 without), b) 699 in Sweden (301 […]

Shots: UCB to receive $120M and is eligible to receive $2B as cost reimbursement, development, and commercial milestones as well as royalties on sales of […]
The FDA has approved Jazz Pharmaceuticals’ sleep disorder drug Xywav, to treat daytime sleepiness in patients with narcolepsy, offering a regime with less sodium than […]

Roche has announced half-year results showing the COVID-19 pandemic affected sales of its key products, with currency headwinds also weighing on revenues. The Swiss pharma’s […]
Copyright © 2024 | WordPress Theme by MH Themes