
3rd TIGIT Axis Therapies Summit
The 3rd TIGIT Axis Therapies Summit returns to unite large pharma, innovative biotechs and pioneering academics to discuss the full range of next-generation TIGIT axis targeted […]

The 3rd TIGIT Axis Therapies Summit returns to unite large pharma, innovative biotechs and pioneering academics to discuss the full range of next-generation TIGIT axis targeted […]

Bristol-Myers Squibb is on course to move its BCMA-targeting CAR-T therapy further up the treatment pathway in multiple myeloma, after reporting that it met its […]

Sanofi is making a €300 million investment in Chinese biotech Innovent as part of a collaboration to bring two new cancer therapies as quickly as […]

Two phase 3 trials of Merck & Co’s Keytruda in metastatic castration-resistant prostate cancer (CRPC) and advanced liver cancer have ended in failure, proving once […]

Bristol-Myers Squibb has revealed that its checkpoint inhibitor combination of Opdivo and Yervoy failed a phase 3 trial as adjuvant (post-surgery) therapy for renal cell […]

Bristol-Myers Squibb’s new immune checkpoint inhibitor combination Opdualag has only been on the US market for a few weeks, but is already off to a […]

The EMA’s human medicines committee has recommended approval of Johnson & Johnson’s Tecvayli as a fourth-line therapy for multiple myeloma, joining a growing group of […]

While immunotherapies have transformed treatment of many types of cancer, they do sometimes fail to have an impact – as Merck & Co has just […]

Roche’s Genentech unit has gone all in on its collaboration with UK biotech Bicycle Therapeutics, taking up an option on a second additional target in […]

The FDA has granted a priority review to Roche’s T-cell engager Lunsumio as a treatment for follicular lymphoma (FL), setting up a decision on the […]

AstraZeneca has signed a deal to buy US biotech TeneoTwo in a deal worth up to $1.27 billion that will boost its position therapies for […]

Novartis is still waiting for FDA approval of its PD-1/PD-L1 inhibitor latecomer tislelizumab in relapsed oesophageal cancer, but is already planning a move the drug […]

Just weeks after Bristol-Myers Squibb claimed an FDA green light for Opdivo as a neoadjuvant therapy for non-small cell lung cancer, AstraZeneca is looking to […]

Astellas has agreed a $1.36 billion partnership with US biotech Sutro Biopharma for a series of antibody-drug conjugates for cancer, focusing on applications in patients […]

Boehringer Ingelheim must like what it has seen in its two-year-old R&D collaboration with Trutino Biosciences to develop cytokine therapies – it has just taken […]

While Merck & Co’s Keytruda is the undisputed leader of the non-small cell lung cancer (NSCLC) category, there’s one category where it lags behind Roche’s […]

Roche has secured its first regulatory approval for mosunetuzumab, a first-in-class CD20xCD3 bispecific antibody, as a treatment for patients with follicular lymphoma (FL) in the […]

Astellas has become the latest big pharma company to sink some money into immuno-oncology start-up GO Therapeutics, forging an alliance to develop antibodies targeting two […]

Shares in Swedish biotech RhoVac were down 94% in mid-morning trading today after the company revealed a phase 2b trial of its prostate cancer vaccine […]

The FDA has cleared two combination drug regimens based on Bristol-Myers Squibb’s PD-1 inhibitor Opdivo for previously-untreated advanced oesophageal cancer, encroaching on territory held by […]

Johnson & Johnson and Legend Biotech will arrive at next week’s ASCO congress with a new EU approval for multiple myeloma CAR-T Carvykti. The European […]

In February, Atara Biotherapeutics paused enrolment into a trial of a CAR-T cell therapy for mesothelioma after a patient death. Now, its partner Bayer has […]

Merck & Co has quietly added another drug to its immuno-oncology pipeline via an agreement with China’s Sichuan Kelun Pharmaceutical, and is keeping the details […]

Bristol-Myers Squibb’s flurry of dealmaking has continued with en expansion of its partnership with BridgeBio on drugs targeting SHP2, in the hope of combining them […]

Roche’s anti-TIGIT antibody tiragolumab has failed a second phase 3 trial, dousing expectations for the programme and its hopes of finding a companion to its […]

Adding Nykode Therapeutics’ VB10.16 vaccine to treatment with Roche’s cancer immunotherapy Tecentriq resulted in disease control in two-thirds of patients with advanced cervical cancer. The […]

The FDA has kicked off a priority review of AstraZeneca’s checkpoint inhibitor Imfinzi for biliary tract cancer (BTC), setting up what could be approval for […]

Novartis’ CAR-T Kymriah has become the first therapy in the category to be approved for the treatment of relapsed/refractory follicular lymphoma, ahead of Gilead Sciences’ […]

With TCEs being studied in both hematological and solid tumors (39 and 34 trials respectively), there are now more than one hundred projects in the preclinical […]

Gilead Sciences has made another foray into the natural killer (NK) cell category, paying $300 million upfront to access Dragonfly Therapeutics’ TriNKET platform to find […]

The FDA has declined to approve two more cancer therapies developed by drugmakers based in China, in what may be further evidence of a tough […]

Nektar has said it will cut 70% of its workforce in the wake of the comprehensive failure in trials of its immuno-oncology candidate bempegaldesleukin, which […]

Compared to a ‘critical organ’ that had been largely ignored, companies working to develop microbiome-based therapies believe there is untapped potential in the area. Ben […]

Shares in Nkarta more than doubled yesterday on the results of early-stage clinical that suggested it could be on to a winner with its natural […]

CAR-T therapies have revolutionised the treatment of some blood cancers, but are costly as well as time- and resource-intensive. Now, US researchers think one solution […]

AstraZeneca’s anti-CTLA4 antibody tremelimumab is moving closer to what once looked like an extremely unlikely regulatory approval, after a string of negative trial results. The […]

Roche is on the brink of its first regulatory approval for what could be the first bispecific antibody targeting CD3 and CD20 – mosunetuzumab – […]

Regeneron has agreed an all-cash deal to acquire CheckMate Pharmaceuticals, adding immune activator vidutolimod to its cancer immunotherapy pipeline and ending a barren spell for […]

Four years after Bristol-Myers Squibb paid $1.85 billion upfront to make Nektar’s bempegaldesleukin a companion drug to its cancer immunotherapy Opdivo, the alliance is being […]

Two years after signing a $3.9 billion alliance with Genmab to find new cancer therapies, AbbVie has said it is ready to start talking to […]

Gilead Sciences can restart pivotal trials of its cancer antibody magrolimab – the centre piece of its $4.9 billion acquisition of Forty Seven in 2020 […]

Affimed says new data from a phase 1/2 trial of its natural killer (NK) cell engager therapy AFM13 provide further evidence of its efficacy in […]

Adding an mRNA vaccine to CAR-T cell therapy could be the key to unlocking activity in solid tumours, according to German biotech BioNTech. CAR-Ts have […]

Jazz Pharma has bolstered its pipeline of cancer drugs through a $1.3 billion licensing agreement with Werewolf Therapeutics that adds its first immuno-oncology candidate. The […]

The EU regulator has started a review of Novartis’ PD-1 inhibitor tislelizumab – licensed from Chinese biotech BeiGene – for oesophageal and lung cancers, ahead of […]

Immunocore now has approval on both side of the Atlantic for Kimmtrak – the first cancer therapeutic based on T cell receptor (TCR) technology – […]

Roche has suffered a setback with its much-anticipated pairing of anti-TIGIT antibody tiragolumab and PD-L1 inhibitor Tecentriq in small cell lung cancer (SCLC), in a […]

Johnson & Johnson is in course for approval of its multiple myeloma CAR-T therapy Carvykti in the EU the next few weeks, after getting a […]

AstraZeneca’s hopes of developing its PD-L1 inhibitor Imfinzi for cervical cancer have been hit by a failed phase 3 trial of the drug alongside chemotherapy […]

Israeli startup Nucleai has raised $33 million in a second-round financing that will be used to further its work on using artificial intelligence and spatial […]

Bayer’s venture capital unit has co-led a $175 million investment in Affini-T that will be used to take its T cell receptor (TCR) therapies for […]

Merck & Co’s KEYNOTE-158 trial of Keytruda has underpinned another FDA approval, this time for the PD-1 inhibitor as a single-agent therapy as a therapy […]

Merck & Co’s two powerhouse oncology drugs – Keytruda and Lynparza – have failed to show efficacy when used in combination for patients with previously […]

Four years ago, Bristol-Myers Squibb paid $1.85 billion upfront to claim rights to Nektar’s bempegaldesleukin, setting it up to be companion drug to its cancer […]

There isn’t enough evidence to support the use of Merck & Co/MSD’s Keytruda in combination with chemotherapy as a first-line treatment for advanced triple negative […]

Sanofi’s rash of dealmaking has continued with a $2.5 billion tie-up with Chinese cancer immunotherapy biotech Adagene, focused on developing a new generation of safer […]

FDA advisors have voted against approval of Eli Lilly and Innovent Biologics’ cancer immunotherapy sintilimab, undermining hopes of a new, lower-priced option in the PD-1/PD-L1 […]

Merck & Co has passed another milestone in its drive to move PD-1 inhibitor Keytruda earlier in the treatment pathway for cancer, with the European […]

Immunocore has secured a piece of biotech industry history, becoming the first company to get an FDA approval for a cancer therapeutic based on T […]

The FDA has placed a partial clinical fold on clinical trials of Gilead Sciences’ magrolimab, the centre piece of its $4.9 billion acquisition of Forty […]

Bristol-Myers Squibb chief executive Giovanni Caforio has promised to invest in the company’s pipeline at the virtual JP Morgan Healthcare Conference, and demonstrated that commitment […]

Johnson & Johnson’s much-touted crop of bispecific antibodies for cancer generated its first commercial product in May, and the drugmaker has now filed for FDA […]

Daiichi Sankyo has been granted breakthrough status by the FDA for patritumab deruxtecan, a HER3-targeted antibody-drug conjugate (ADC) in clinical trials for lung cancer. The […]

Sanofi has bolstered its immuno-oncology pipeline with a $1 billion agreement to acquire Amunix Pharma of the US – its fourth bolt-on deal of 2021. […]

Novartis has joined the ranks of big pharma companies developing TIGIT-targeted drugs for cancer, and found its candidate in an existing partner, Chinese biotech BeiGene. […]

Gilead Sciences has taken a big step towards positioning its CAR-T therapy Yescarta as an option for previously-untreated large B cell lymphoma (LBCL) with updated […]

A pair of trials have suggested that CAR-T therapies could offer an improved option for patients with large B cell lymphoma (LBCL) who have relapsed […]

Roche’s closely-watched combination of anti-TIGIT antibody tiragolumab and PD-L1 inhibitor Tecentriq has shown long-term efficacy in an ongoing trial in lung cancer, adding to the […]

China’s Shanghai Henlius Biotech says its experimental PD-1 inhibitor serplulimab has extended survival when added to chemotherapy in a phase 3 trial as first-line therapy […]

Atara Biotherapeutics’ tabelecleucel (tab-cel) has become the first ‘allogeneic’ cell therapy – in other words one which doesn’t involve harvesting cells from patients – to […]

Gilead Sciences has forged closer ties to its cancer drug partner Arcus Biosciences , although a licensing deal doesn’t go as far as the takeover […]

Merck & Co has chalked up another win in drive to move PD-1 inhibitor Keytruda earlier in the treatment pathway for cancer, after the FDA […]

Private equity firm Blackstone has agreed to provide up to $250 million in financing to UK biotech Autolus Therapeutics, to help it take acute lymphoblastic […]

Daiichi Sankyo has launched its oncolytic virus therapy Delytact in Japan – its first world market – as a treatment for malignant glioma, an aggressive […]

Take two recently-approved immunotherapies for triple negative breast cancer (TNBC), put them together, and what do you get? Hopefully a new standard of care for […]

Japanese drugmaker Takeda has said it will exercise its option to buy UK-based GammaDelta Therapeutics, a specialist in ‘off-the-shelf’ cell therapies for solid tumours and […]

The dominance of Merck & Co’s Keytruda in the treatment of previously-untreated non-small cell lung cancer (NSCLC) means it will be hard to dislodge – […]

AstraZeneca’s checkpoint inhibitor Imfinzi has become the first cancer immunotherapy to improve survival in previously-untreated patients with biliary tract cancer (BTC), a rare and aggressive […]

Merck & Co has claimed EU approval for Keytruda in triple-negative breast cancer (TNBC), a particularly hard-to-treat form of the disease. The European Commission has […]

Merck & Co’s Keytruda become the first cancer immunotherapy to be approved as a first-line oesophageal cancer treatment in the EU earlier this year, but […]

CAR-T therapies have revolutionised the treatment of some blood cancers, but so far haven’t been able to repeat that success in solid tumours. UK startup […]

Eli Lilly made a late entry into the checkpoint inhibitor market when it licensed ex-China rights to Innovent’s sintilimab last year, and a new trial […]

Roche has claimed an earlier-than-expected FDA approval for its checkpoint inhibitor Tecentriq in non-small cell lung cancer (NSCLC), giving the drug a lead in early-stage […]

SMi Group Proudly Presents… Medical Wearables for Biosensors USA Conference 2021 Conference: 25 – 26 October 2021 Virtual Conference: Online Access Only Website: www.biosensors-medical-wear.com/pharmaphorum Exploring […]

Patients in Scotland with newly-diagnosed advanced kidney cancer can now access a therapy combining Bristol-Myers Squibb’s immunotherapy Opdivo with Ipsen’s targeted drug Cabometyx, ahead of […]

A phase 1 trial of Allogene’s of-the-shelf CAR-T for lymphoma has been placed on hold by the FDA, after a “chromosomal abnormality” was seen in […]

Fresh from claiming its first bispecific antibody approval, Johnson & Johnson has delved deeper into the category with a deal to license a drug for […]

Discover the Intricacies of Inflammasome Regulation, Leverage Inflammasome Proteins as Biomarkers & Accelerate Novel Inflammasome Therapeutics into Proof-Of-Concept Clinical Trials & Beyond Built with Ventus […]

The FDA has approved Kite Pharma’s Tecartus as a treatment for adults with relapsed or refractory B-cell precursor acute lymphoblastic leukaemia (ALL), making CAR-T therapy […]

GlaxoSmithKline has ended its collaboration with Germany’s Merck KGaA’s cancer treatment bintrafusp alfa, after a trio of failed clinical trials. Given the disappointments the decision […]

NICE has recommended routine NHS access to Roche’s cancer immunotherapy Tecentriq for some bladder cancer patients, four years after it was first made available via […]

Merck & Co’s FDA approval for Keytruda in advanced liver cancer looks secure, after an Asian clinical trial found that the cancer immunotherapy improved survival […]

Boehringer Ingelheim says it has agreed to buy US biotech Abexxa Biologics, its second acquisition in the cancer immunotherapy area in the space of a […]

There’s no shortage of cancer immunotherapies trying to knock Merck & Co’s Keytruda off the top of the tree in previously-untreated non-small cell lung cancer […]

MSD’s Keytruda has become the first immunotherapy cleared by NICE for previously-untreated patients with advanced oesophageal cancer, making it an option for routine NHS treatment. […]

Novartis has filed for FDA approval of tislelizumab – a PD-1 inhibitor licensed from Chinese biotech BeiGene earlier this year – as a second-line treatment […]

Roche has largely been a spectator on the side lines as other drugmakers have embraced cell therapies for cancer, but it has made a big […]

Anna Biosciences’ Dr Anthony Rullo discusses the drug development company’s immunotherapy platform. Over the last 30 years, we have seen dramatic improvements in patient outcomes […]

Novartis’ CAR-T therapy Kymriah racked up back-to-back approvals in acute lymphoblastic leukaemia and large B-cell lymphoma a few years ago, but hasn’t added to its […]

GlaxoSmithKline’s big gamble on Merck KGaA’s bintrafusp alfa has gone from bad to worse, after the drug failed yet another mid-stage trial. The latest disappointment […]

Pfizer is the latest big pharma company to take a position on drugs that target CD47 – a ‘don’t eat me’ signal that protects cancer […]

The FDA has approved GlaxoSmithKline’s latecomer PD-1 inhibitor Jemperli for a second use that will significantly extend the patient population eligible for treatment with the […]

Merck & Co is looking at a decision from the FDA in December on its application to market Keytruda as a post-surgery treatment for people […]

Gilead Sciences’ Kite Pharma has mapped out another route to the development of allogeneic or ‘off-the-shelf’ cell therapies for cancer, using technology developed by Appia […]

Bristol-Myers Squibb is in a battle with Merck & Co for position in the upper gastrointestinal cancer immunotherapy market, and has just reinforced its position […]

Merck & Co’s Keytruda has just picked up its 30th approval from the FDA, and it is a good one – early-stage triple-negative breast cancer […]

Bristol-Myers Squibb has withdrawn its US marketing approval for PD-1 inhibitor Opdivo as a second-line, single-agent treatment for hepatocellular carcinoma, a common form of liver […]

Gilead Sciences just forged even closer ties with its cancer drug partner Arcus Biosciences, enticing the biotech’s chief medical officer Bill Grossman to a new […]

BioNTech has found a use for some of the windfall cash it has raked in from its COVID-19 vaccine, agreeing a deal to buy Kite […]

GlaxoSmithKline’s head of oncology Axel Hoos has resigned from the company, and will take up a new role as chief executive of US biotech Scorpion […]

A peptide vaccine developed by Australia’s Imugene has reduced tumour size in around half of patients with HER2-positive gastric or gastroesophageal junction (GEJ) cancer in […]

Merck & Co will voluntarily withdraw a US marketing approval for cancer immunotherapy blockbuster Keytruda in gastric cancer, after studies intended to confirm its efficacy […]

Gilead Sciences’ cell therapy Kite Pharma has bulked up its position in natural killer (NK) cell therapies, pledging up to $2.3 billion for a partnership […]

GlaxoSmithKline has staked a claim to the fast-moving category of cancer therapies targeting the TIGIT immune checkpoint, agreeing a $2 billion plus licensing agreement with […]

Daiichi Sankyo has made a little bit of pharma industry history, becoming the first drugmaker to win approval in Japan for a therapy for cancer […]

Belgian biotech Argenx has lost its development partner for acute myeloid leukaemia antibody cusatuzumab, after Johnson & Johnson backed away from the alliance after two […]

Genmab has enlisted the help of Bolt Biotherapeutics to develop a new class of immune-boosting antibody conjugates for cancer in a deal that could be […]

Johnson & Johnson has secured a six-month FDA review for its multiple myeloma CAR-T therapy ciltacabtagene autoleucel (cilta-cel), narrowing the lead for Bristol-Myers Squibb and […]

With the ink barely dry on the FDA’s approval of Johnson & Johnson’s Rybrevant, its first bispecific antibody for cancer, the drugmaker is already showcasing […]

Merck Serono’s Bavencio is the only checkpoint inhibitor to be approved for first-line maintenance treatment of bladder cancer in Europe, but UK cost-effectiveness agency NICE […]

Johnson & Johnson’s Janssen unit has filed for approval of its multiple myeloma CAR-T ciltacabtagene autoleucel in Europe, chasing after a rival therapy from Bristol-Myers […]

The FDA’s Oncologic Drugs Advisory Committee (ODAC) has voted to strip Merck & Co’s Keytruda of its accelerated approval in gastric and gastroesophageal junction (GEJ) […]

Merck & Co’s Keytruda and Roche’s Tecentriq should stay on the market as first-line treatments for bladder cancer, at least until new clinical data becomes […]

Shots: Pieris to receive $10M upfront and is eligible to receive $353M in development, regulatory, and commercial based milestones along with royalties on sales of […]

FDA advisors will scrutinise three cancer immunotherapies granted conditional approvals at a three-day meeting this week, to see if they should stay on the market. […]

Exscientia was the first company to start human trials of a new drug designed using artificial intelligence (AI) last year, and now says it has […]

UK biotech Achilles Therapeutics has completed its initial public offering (IPO) in the US, raising $175.5 million that will be used to take its personalised […]

Shots: In the P-I SPICE study, the companies will evaluate the clinical combination of BMS’ Opdivo with PsiOxus’ NG-641 and optimize the combination IV dosing […]

Shots: F-star to receive option exercise payment and will be eligible to get milestones and royalties on net sales of therapies resulting from the collaboration […]

Sanofi and Regeneron’s checkpoint inhibitor Libtayo has become the first immunotherapy to improve survival when used as a second-line therapy in cervical cancer, setting up […]

In an interview, BrightInsight Co-founder and CEO Kal Patel, MD, offered a preview of the report, The Role of Digital Health in Immuno-oncology Therapy Development […]

Nektar Therapeutics is waiting on multiple data readouts for its lead drug bempegaldesleukin paired with strategic partner Bristol-Myers Squibb’s checkpoint inhibitor Opdivo, but that hasn’t […]

Shots: Biomunex to receive up front, development, regulatory & sales milestones, equity investment along with royalties on sales of therapies Onward get the right to […]

New phase 3 data have shored up the position of Merck & Co’s cancer immunotherapy Keytruda in the increasingly competitive first-line kidney cancer market. The […]

The pairing of Heat Biologics’ lead drug HS-110 with Bristol-Myers Squibb’s blockbuster checkpoint inhibitor Opdivo has boosted survival in patients with non-small cell lung cancer […]

Dutch biotech InteRNA Technologies has raised €18.5 million ($22 million) in second-round financing as it prepares to start clinical trials of its lead drug candidate […]

Bristol-Myers Squibb finally has FDA approval for its CAR-T therapy liso-cel, which has been cleared by the US regulator as Breyanzi for certain forms of […]

Incyte has filed its PD-1 inhibitor retifanlimab with the FDA, seeking approval to treat a form of anal cancer associated with human papillomavirus (HPV) and […]

Pfizer and partner Merck KGaA now have approval on both sides of the Atlantic for their checkpoint inhibitor Bavencio as a first-line maintenance therapy for […]

Always a big player in oncology, Eli Lilly has fallen behind some of its rivals when it comes to cancer immunotherapy, but a new R&D […]

Gilead Sciences’ Kite unit has teamed up with the UK’s Oxford BioTherapeutics (OBT) to develop a new clutch of cell therapy products for solid tumours […]

Roche’s closely-watched combination of two checkpoint inhibitors – TIGIT-targeting tiragolumab and PD-L1 drug Tecentriq – has claimed breakthrough status from the FDA. Like PD-L1, TIGIT […]

Johnson & Johnson’s Janssen unit has filed a rolling submission for its multiple myeloma CAR-T ciltacabtagene autoleucel (cilta-cel) to the FDA, in hot pursuit of […]

In a deal that could top $1 billion, Merck & Co has teamed up with US biotech Janux Therapeutics to bring T-cell immunotherapies to cancer […]

Catamaran Bio has weighed anchor with a $42 million first-round financing that will be used to pull its off-the-shelf natural killer (NK) cell therapies for […]

Are you interested in finding the next breakthrough in immuno-oncology preclinical or clinical development? Recent scientific, clinical breakthroughs and high-profile industry deals have reignited the […]

Shares in US biotech Five Prime Therapeutics more than tripled after it reported promising trial results with stomach cancer drug candidate bemarituzumab, returning the stock […]

An experimental gene therapy developed by Texas biotech Genprex will be paired with AstraZeneca’s Tagrisso and Merck & Co’s Keytruda – both leading their respective […]

The FDA has started a priority review of Regeneron and Sanofi’s checkpoint inhibitor Libtayo in first-line non-small cell lung cancer (NSCLC), based on data they […]

As recently as June, NICE was minded not to back routine NHS of MSD’s Keytruda as a first-line treatment for advanced head and neck cancer, […]

Two years after quietly forming cancer start-up Nuvation Bio, ex-Axovant and Medivation CEO David Hung has agreed a big merger that will take it public […]

Bristol-Myers Squibb could be mere months away from claiming a second US approval for an Opdivo-based combination immunotherapy that will help defend its first-line kidney […]

With the ink barely dry on a deal to develop Dragonfly’s lead cancer immunotherapy, Bristol-Myers Squibb’s business development team has agreed to buy Canadian biotech […]

Novartis’ hopes of catching up with other companies in immuno-oncology have been dented after PD-1 inhibitor spartalizumab failed a phase 3 trial in skin cancer. […]

Eli Lilly has doubled down on its partnership with Chia’s Innovent with a licensing deal for ex-China rights to Tyvyt, their PD-1 inhibitor, which some […]
Bristol-Myers Squibb’s Opdivo has improved survival in a trial involving patients with mesothelioma, a form of lung cancer, getting one up over rival checkpoint inhibitor […]
Roche’s hopes of extending the use of its PD-L1 inhibitor Tecentriq in triple-negative breast cancer (TNBC) have been dashed by a late-stage trial failure looking […]
Regeneron and Sanofi’s PD-1 inhibitor Libtayo will be tested in tandem with a cancer vaccine developed by BioNTech in melanoma in people who have failed […]
Roche’s checkpoint inhibitor Tecentriq has been cleared for another new use in the US – in melanoma – but could struggle to displace rival drugs […]
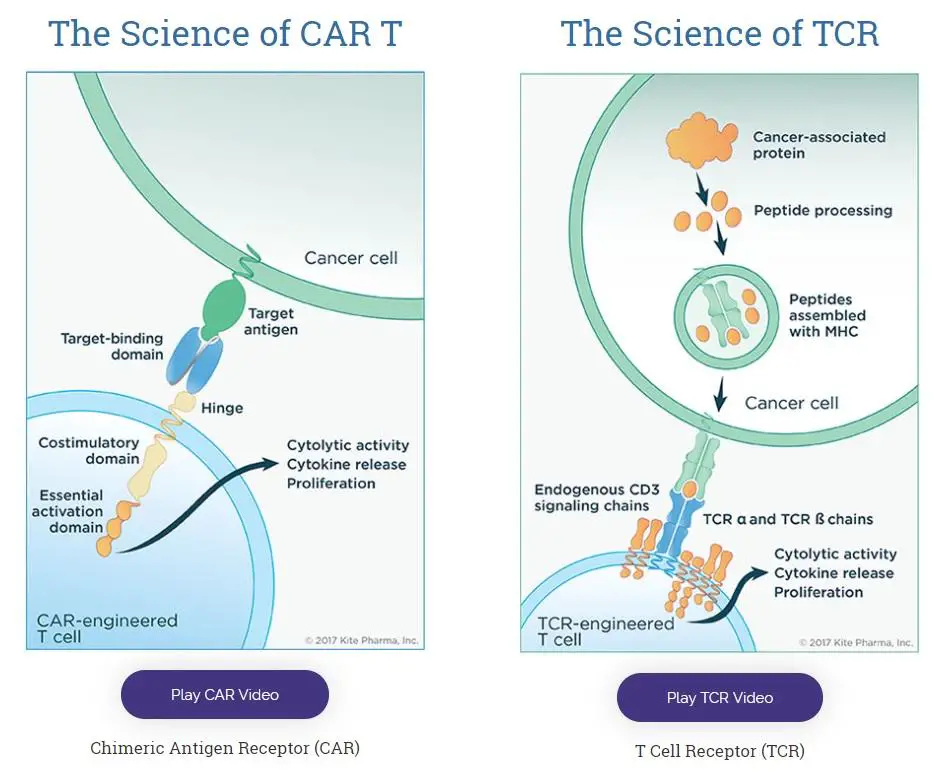
Gilead’s $11.9 billion purchase of Kite Pharma in 2017 didn’t follow the script, with two big write-downs in the value of the asset since then, […]

Gilead Sciences’ run of oncology-focused deals has continued with an option to buy cancer immunotherapy specialist Tizona – even though the biotech’s lead drug candidate […]
Shots: F-star to receive option exercise payment and will be eligible to get milestones and royalties on net sales of therapies resulting from the collaboration. […]

Shots: GenScript will be responsible for all pre-clinical pharmacy research and IND filling in Selecxine’s project which is based on cytokine-antibody complexes The company provided […]
Copyright © 2024 | WordPress Theme by MH Themes