
ICMRA seeks international alignment on COVID-19 product assessment
COVID-19 drugs, vaccines and diagnostics that are developed and tested within new international guidelines can expect rapid assessment and authorisation. The criteria, set out in […]

COVID-19 drugs, vaccines and diagnostics that are developed and tested within new international guidelines can expect rapid assessment and authorisation. The criteria, set out in […]
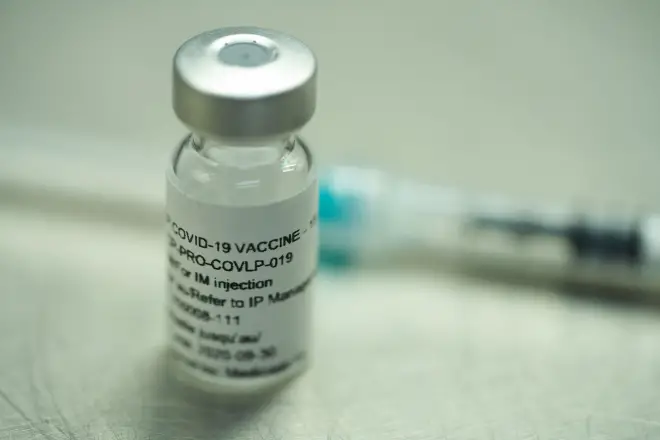
Medicago has started dosing healthy volunteers in the first human trial of its candidate vaccine for COVID-19, a few days after partnering with GlaxoSmithKline on […]

Click here to download printable PDF The Coronavirus Treatment Acceleration Program (CTAP) is a special emergency program instituted by the US Food and Drug Administration […]

Sponsored by Covance Register The PMSR regulations for drugs, devices, and biological products share many similarities, each set of regulations establishes distinct reporting requirements, including […]

Recently Mariah Baltezegar, MBA, Executive Director and Head of Peri- and Post-Approval Virtual Trials with Evidera, and Niklas Morton, MSc, Senior Vice President and Global […]

By making it easier for patients to participate in research today, Parexel’s Decentralized Clinical Trials are defining a better pathway to the medicines of tomorrow. […]

Download the white paper now https://bit.ly/evidera-digena Multiple strategies, each with their individual advantages and drawbacks, may be employed to answer a research question, but determining […]

eSource (defined as data initially recorded in an electronic format) formats have provided excellent opportunities to not only streamline clinical research securely, but to increase […]

Provided by: Elligo Health Research https://www.elligohealthresearch.com/ The ongoing COVID-19 pandemic continues to highlight the need for secure virtual capabilities and decentralized/hybrid approaches to clinical research […]

GVK Biosciences Walk In for Synthesis R and D /Analytical R and D 1st Feb 2020 GVK Biosciences Pvt. LtdRecruitment January 2020 M.Sc. Organic Chemistry for Synthesis […]

Aspiro Pharma Ltd – Walk-In Interview for AR and D on 6th Dec’ 2019 Greetings from Aspiro Pharma!!! Openings for R&D Professionals (Injectables) Department: AR&D […]

Gvk Bioscience walk in Drive for R and D on 7th Dec 2019 Visit above links for more information

Job Opening at CPL Biological apply now. Visit above links for more information
Copyright © 2024 | WordPress Theme by MH Themes