
BioMarin Reports FDA’s Hold on P-I/II Phearless Trial of BMN 307 in Adults with PKU
Shots: The FDA’s clinical hold is based on interim safety findings from a pre-clinical study which showed that 6/7 animals treated with BMN 307 (2e14 […]

Shots: The FDA’s clinical hold is based on interim safety findings from a pre-clinical study which showed that 6/7 animals treated with BMN 307 (2e14 […]

Shots: The P-III MELODY study involves evaluating Nirsevimab (50mg/100mg, IM) vs PBO to determine the incidence of medically attended LRTI due to RSV confirmed by […]

Shots: The submission is based on P-IIb/III SELECTION study evaluating the efficacy and safety of filgotinib (200mg, qd) vs PBO in patients with mod. to […]

Shots: The company has completed the enrollment in the P-IIB HOPE4MCI trial evaluating the efficacy of AGB101 vs PBO in 164 patients to treat amnestic […]

Shots: The first sBLA is based on the P-III EV-301 trial assessing Padcev vs CT in ~ 600 patients with LA or mUC prior treated […]

Shots: The company reported new post hoc data from the P- III ASCLEPIOS trials assessing Kesimpta (ofatumumab) vs teriflunomide in a subgroup of newly diagnosed, […]

Shots: The P-II KINETIC study involves assessing the efficacy, safety, and tolerability of SAGE-324 (60mg, qd) vs PBO in 69 patients in a ratio (1:1) […]

Shots: MD Anderson will sponsor and lead P-I/II study to assess DS-1594 as monothx. and in combination regimens for patients with r/r AML and ALL […]

Shots: The US FDA has accepted IND to assess the safety, tolerability, PK, and preliminary antitumor activity of XB002 in patients with advanced solid tumors. […]

Shots: The company has completed the enrollment in the first cohort of P- I/II trial involves assessing AMT-130 in 10 patients, out of which six […]

Shots: The first patient has been dosed in P-Ib trial evaluating SpringWorks’ Nirogacestat (GSI) + Janssen’s Teclistamab (Ab targeting BCMA and CD3) in patients with […]

Shots: The sBLA is based on data from the P- I/II ZUMA-3 trial in adult patients age ≥18 years old for the treatment of adult […]

Shots: The NDA submission is based on data from P-III PROTECTIVE-2 study evaluating Plinabulin + Neulasta vs Neulasta alone for the prevention of CIN The […]

Shots: The EUA submission is based on an interim analysis of the P-III COMET-ICE study assessing VIR-7831 vs PBO for the early treatment of COVID-19 […]

Shots: The submission is based on a P-III TIDES trial assessing TAK-003 (0.5ml, SC) vs PBO in 20,000+ healthy children & adolescents aged 4-16yrs. to […]

Shots: The P-III trial involves assessing REGEN-COV (casirivimab & imdevimab, 1200/2400mg, IV) vs PBO in non-hospitalized patients with COVID-19 The trial met its 1EPs i.e. […]

Shots: The NDA is based on P-II study-004 trial involves assessing Belzutifan (120 mg, PO, qd) in 61 patients with VHL disease who had at […]

Shots: Genentech reports the new 2yrs. data from part 2 of SUNFISH study assessing Evrysdi vs PBO in people aged 2-25 years with Type 2 […]

Shots: The submission is based on P-III HELP study evaluating lanadelumab (300mg, q2w & q4w) vs PBO in 125 patients with HAE attack for 26wks. […]

Shots: The submission is based on P-III HELP study evaluating lanadelumab (300mg, q2w & q4w) vs PBO in 125 patients with HAE attack for 26wks. […]

Shots: The submission is based on EV-301 and EV-201 trials in Japan. The P-III EV-301 trial assessing Enfortumab Vedotin vs CT in ~ 600 patients […]

Shots: The P-III BLAZE-1 study involves assessing Bamlanivimab (700 mg) + Etesevimab (1400 mg) vs PBO in 769 COVID-19 patients aged ≥12yrs. who are at […]

Shots: The P-III CANOPY-2 study involves assessing canakinumab (ACZ885) + CT (docetaxel) in 237 adults with LA/m-NSCLC, prior treatment with PD-L1 inhibitors and Pt-based CT. […]

Shots: Takeda has submitted an NDA to MHLW to import and distribute Moderna’s (mRNA-1273/ TAK-919) in Japan Takeda is conducting a P- I/II study assessing […]

Shots: The P-III SURPASS-2 trial involves assessing Tirzepatide (5/10/15mg) vs Semaglutide (1mg) in 1,879 patients in a ratio (1:1:1:1) with T2D inadequately controlled with ≥1500 […]

Shots: The P-III 20120215 study involves assessing of Blincyto vs SoC consolidation CT before alloHSCT in pediatric patients with high-risk first relapsed B-cell ALL. The […]

Shots: The NDA submission is based on two P-III studies (COUGH-1 & -2) involves assessing gefapixant (45/15mg, bid) vs PBO in 2,044 patients with RCC […]

Shots: Regeneron reported changes to the P-III study assessing REGEN-COV in non-hospitalized patients with COVID-19, following recommendations from the IDMC The IDMC found clear clinical […]

Shots: Otsuka and Click have reported the initiation of the Mirai study which is a fully remote clinical trial to investigate the effectiveness of digital […]

Shots: Following the completion of a P-I program, the company plans to initiate P- II studies to evaluate ITI-1284 ODT-SL for the treatment of behavioral […]

Shots: The US FDA has accepted the PR of BLA for TicoVac to prevent TBE in patients aged ≥1year with an anticipated PDUFA date in […]

Shots: The sNDA filing is based on P-III ADVANCE trial data, assessing Otezla (30 mg, bid) vs PBO in 595 patients in a ratio (1:1) […]

Shots: The sNDA submission is based on the P-III REACH3 study assessing ruxolitinib vs best available therapy in adult and pediatric patients ≥12yrs. with steroid-refractory […]

Shots: The NDA is based on two P-III TRuE-AD 1 & 2 assessing ruxolitinib cream (0.75%/1.5%, bid) vs vehicle (non-medicated cream) in 600 patients aged […]

Shots: The P-III studies SKYLIGHT 1 & 2 involves assessing fezolinetant (30 & 45mg, qd) vs PBO in women with mod. to sev. vasomotor symptoms […]

Shots: The first BLA submission is based on P-III EV-301 trial which involves assessing of Padcev vs CT in 600 patients with locally advanced or […]

Shots: The sDNA submission is based on a P-III APSEN study assessing Brukinsa vs ibrutinib in 351 patients with WM and includes a pivotal P-II […]

Shots: The P-lll SURPASS-3 study involves assessing Tirzepatide (5/10/15 mg) vs titrated insulin degludec in 1444 participants in a ratio (1:1:1:1) with T2D treated with […]

Shots: The P-III YOSEMITE & RHINE studies involve assessing faricimab (6mg at personalized dosing intervals of up to 4mos. and at fixed 2mos. intervals) vs […]

Shots: The EUA submission is based on P-III ENSEMBLE trial assessing the safety and efficacy of the COVID-19 vaccine in protecting adults aged ≥18yrs. against […]

Shots: The MAA submission is based on a P-Ib/II CARTITUDE-1 study assessing the safety and efficacy of cilta-cel in adults with r/r MM. The company […]

Shots: The P-I CHRYSALIS study involves assessing of amivantamab (RP2D of 1050 mg [1400 mg for a patient weight of ≥80 kg]) as a monothx. […]

Shots: The BLA submission is based on data from P-III clinical comparison study that demonstrated BAT1706 is highly similar to reference bevacizumab in clinical efficacy, […]

Shots: The P-II/III BLAZE-1 trial involves assessing of Bamlanivimab + Etesevimab (2800 mg) vs PBO in 1035 patients recently diagnosed with COVID-19 in the ambulatory […]

Shots: The BLA submission is based on P-II POD1UM-202 trial involves assessing of retifanlimab (500 mg, q4w) in 94 patients with locally advanced or metastatic […]

Shots: The sNDA submission is based on a P-II study that involves assessing Esbriet vs PBO in patients aged ≥18-85yrs. with progressive fibrosing UILD for […]

Shots: The US FDA has accepted the BLA and granted the PR with an anticipated PDUFA date as Jul 17, 2021 The BLA filing marks […]

Shots: The P-Ib/II study involves assessing of Onvansertib + Folfiri and bevacizumab in 44 patients with 2L treatment of KRAS-mutated mCRC. The company also provided […]

Shots: The ongoing P-III GENEr8-1 study involves assessing of valoctocogene roxaparvovec (6e13 vg/kg dose) in 134 patients with with severe hemophilia A. The patients received […]

Shots: The Study 102 involves assessing of SRP-9001 vs PBO in 41 patients in a ratio (1:1) with DMD aged 4-7yrs. Results: @12wks. post treatment, […]

Shots: The first patient has been dosed in P-III CIFFREO study assessing PF-06939926 vs PBO in 99 ambulatory male patients aged 4-7yrs. with DMD across […]

Shots: The P-III HELIOS-A study assessing vutrisiran (25mg, SC, once every 3mos.) vs patisiran (0.3 mg/kg, IV, q3w) in 164 patients in a ratio (3:1) […]

Shots: The US FDA has accepted the PR for NDA of mirabegron (oral suspension) and sNDA for Myrbetriq (mirabegron, tablets) for neurogenic detrusor overactivity (NDO) […]

Shots: The two P-III studies KEEPsAKE-1 & -2 involves assessing Skyrizi (150mg) vs PBO followed by risankizumab (150mg, @24wks.) in patients with active PsA who […]

Shots: The BLA submission is based on P-lll study assessing the safety and efficacy of Somatrogon (0.66 mg/kg, qw) vs Somatropin (0.034 mg/kg, qd) in […]

Shots: The BLA for teplizumab to delay or prevention of clinical T1D in at-risk individuals has been filed to the US FDA. The FDA has […]

Shots: The NDA is based on STORM and SADAL studies. The P-IIb STORM study involves assessing of ATG-010 + low-dose dexamethasone in patients with rrMM […]

Shots: The P-II US study involves assessing Opaganib + SOC vs PBO + SOC in 40 patients requiring oxygen support in a ratio (1:1) and […]

Shots: Health Canada expedite the review of AstraZeneca’s COVID-19 vaccine after the vaccine received the UK’s MHRA approval for emergency use Following an agreement to […]

Shots. The company has submitted MAA to COFEPRIS (the health regulatory authority for Mexico) for its COVI-STIX rapid diagnostic test to detect SARS-CoV-2 virus nucleocapsid […]

Shots: Vertex’ new drug submission for Trikafta has been accepted for Priority Review by Health Canada for the treatment of CF in people aged ≥12yrs. […]

Shots: The results of an interim analysis of P-III study showed that the safety of the new coronavirus inactivated vaccine after inoculation All patients produced […]

Shots: The company reported encouraging initial data from an ongoing P-I/II/III trial of its casirivimab + imdevimab (8,000/2,400mg) in hospitalized COVID-19 patients requiring low-flow oxygen. […]

Shots: The submission is based on SUSTAIN FORTE trial assessing Ozempic (2.0mg, qw) vs Ozempic (1.0mg) in 961 people with T2D in need of treatment […]

Shots: The last patient is out from the P-IIb part of its multinational P-IIb/III study of NP-120 for the COVID-19 has now completed treatment with […]

Shots: The sNDA submission is based on P-II/P-III Clinical trial assessing Dextenza(dexamethasone ophthalmic insert, 0.4mg) vs PBO for the treatment of Ocular Itching associated with […]

Shots: The P-III SOURCE study involves assessing Tezepelumab (210mg, q4w) vs PBO in 150 adult patients as add-on therapy with patients maintained on their currently […]

Shots: The P-III YOSEMITE & RHINE studies assessing faricimab vs aflibercept in 1891 people living with DME. The studies include 3 treatment arms: faricimab (6mg) […]

Shots: The NDA submission is based P-II CodeBreaK 100 study assessing Sotorasib in patients with KRAS G12C-mutated NSCLC prior treated with CT and immunotherapy The […]

Shots: The US FDA has accepted the NDA and granted PR for TAK-721 for the treatment of EoE. If approved, TAK-721 will be the first […]

Shots: The P-lll RUXCOVID study involves assessing Ruxolitinib (5mg, bid) + SoC therapy vs PBO + SoC therapy in 432 patients aged ≥12 yrs. in […]

Shots: Roche reports an exploratory analysis of P-III IMvigor010 study assessing efficacy and safety of adjuvant treatment with Tecentriq vs observation in 809 people with […]

Shots: The submission is based on P-lll EMERGE and ENGAGE study assessing aducanumab vs PBO in patients with AD. The studies evaluate the efficacy of […]

Shots: The P-III MONALEESA-7 trial involves assessing Kisqali + endocrine therapy (goserelin + either an aromatase inhibitor or tamoxifen) as initial treatment vs endocrine therapy […]

Shots: The P-lll SURPASS-1 study involves assessing Tirzepatide as monothx. (5/10/15 mg) vs PBO in 478 patients in the ratio of (1:1:1:1) with T2D inadequately […]

Shots: The results of an interim of P-III program demonstrated that AZD1222 is safe & effective at preventing symptomatic COVID-19 and hospitalization. The interim analysis […]

Shots: The P-lll ASCEMBL study involves assessing Asciminib (40mg, bid) vs Bosulif (bosutinib, 500mg, qd) in 223 patients with Ph+ CML-CP prior treated with two […]

Shots: AbbVie reported pooled analysis and long-term integrated analysis of the P-lll RESONATE-2 and ILLUMINATE study evaluating Imbruvica as 1L treatment of high-risk patients with […]

Shots: The P-lb/ll GO29365 study involves assessing of Polivy (polatuzumab vedotin) + BR vs BR as monthx. in 106 patients with r/r DLBCL who are […]

Shots: AstraZeneca reported long-term follow-up from the positive P-ll ACE-LY-004 study involves assessing Calquence (acalabrutinib) in adult patients with r/r MCL Result: @median follow up […]

Shots: Five-year data from P-III MURANO trial shows sustained PFS with Venclexta/Venclyxto + MabThera/Rituxan vs BR in patients with r/r CLL The data showed a […]

Shots: The P-II ZUMA-5 study involves assessing Yescarta (axicabtagene ciloleucel) in patients with r/r iNHL after at least 2L of systemic therapy. After a single […]

Shots: Janssen reported pooled analyses of long-term follow-up from multiple clinical trials evaluating the use of Imbruvica monothx. and in combination as a 1L treatment […]

Shots: AstraZeneca will divulge new developments across BC at the 2020 SABCS. The key abstract includes data of P-ll DESTINY-Breast01 study reinforcing the efficacy seen […]

Shots: The P-IIb study will evaluate three dose levels of ION449 (once a month, SC) vs PBO and will enroll ~108 patients aged 18-75yrs., who […]

Shots: Amgen, UCB, and Takeda reported that the first patient has been enrolled in the COMMUNITY trial. The study will test whether Amgen’s Otezla, Takeda’s […]

Shots: The MAA is based on a P-II VISION study assessing Tepotinib as monothx. in patients with advanced NSCLC of METex14 skipping alterations, prospectively assessed […]

Shots: The P-IIa study involves assessing vibegron (75mg,qd) in 222 women with abdominal pain due to IBS with IBS-D (diarrhea) and IBS-M (mixed IBS) across […]

Shots: The ongoing P-lll study involves assessing of Sputnik V vaccine vs PBO in 40,000 patients in a ratio (1:3) with COVID-19. The efficacy of […]

Shots: The exploratory P-l/ll study involves the assessment of ONCOS-102 + SoC CT (pemetrexed/cisplatin) as a 1L and 2L treatment in 31 patients with MPM […]

Shots: Interim analysis results of COV002 & COV003 study of AZD1222 in the UK & Brazil showed the vaccine is effective in preventing COVID-19 and […]

Shots: The P-lll ORIENT-32 study involves assessing of Tyvyt (sintilimab) + Byvasda vs sorafenib as a 1L treatment in 571 patients in a ratio (2:1) […]

Shots: In the PROgress study, 200 people living with HIV were provided with a pre-visit survey to guide their conversation with their HCP and assess […]

Shots: The P-lll COVE study involves the assessing of mRNA-1273 (100 µg dose level) in 30,000 patients with COVID-19 in the ratio of (1:1) aged […]

Shots: The P-l study involves assessing CVnCoV in patients with COVID-19. The interim data showed that CVnCoV (2-12µg) was well tolerated and induced strong binding […]

Shots: The P-III NAVIGATOR study involves assessing Tezepelumab + SOC vs pbo + SOC in adults (18–80yrs.) & adolescents (12–17yrs.) with severe, uncontrolled asthma, who […]

Shots: The advisory committee voted 1-8, with 2 members voting uncertain that EMERGE study (without regard for ENGAGE study) provides strong evidence, supporting the effectiveness […]

Shots: Formycon & Bioeq reported the BLA resubmission strategy for FYB201 (biosimilar referencing Lucentis) has been adjusted With the revised submission strategy, the companies expect […]

Shots: The P-IIIb ULTIMATE study involves assessing Cosentyx (300/150mg) vs PBO weekly for a mos. with treatment starting @4wks., followed by a once-a-month dose for […]

Shots: The company will not expand the P-II EMERGE study that assesses Vopratelimab + ipilimumab in PD-(L)1 inhibitor experienced NSCLC patient. Early evaluation of the […]

Shots: The acceptance marks the first marketing application accepted for achondroplasia in the US with an anticipated PDUFA date as of Aug 20, 2021. The […]

Shots: The P-III POETYK PSO-1 study involves assessing deucravacitinib (6mg, qd) vs PBO & Otezla (apremilast) in 666 patients with moderate to severe plaque psoriasis […]

Shots: The pivotal P-III MONALEESA-7 study assessing Kisqali + endocrine therapy vs PBO + endocrine therapy, in pre- & perimenopausal women with HR+/HER2- advanced or […]

Shots: The P-II study is a two-way crossover study assessing BPN14770 (25mg, bid) vs PBO in 30 adult male patients aged 18-45yrs. with FXS due […]

Shots: The sNDA submission is based on P-lll Route-6 study assessing PP6M vs PP3M in 702 patients with schizophrenia previously stabilized on corresponding doses of […]

Shots: The P-III BE SURE study involves assessing bimekizumab vs Humira (adalimumab) for 24wks. is followed until 56wks. in 478 adult patients with chronic PsO […]

Shots: The P-III LIMMitless study is designed to evaluate the long-term safety & efficacy assessing risankizumab (150 mg q12wks.) continuous risankizumab with a loading dose […]

Shots: The P-II BLAZE-1 study assesses bamlanivimab (700, 2800 & 7000 mg) vs PBO in the outpatient setting in patients with mild or moderate symptoms […]

Shots: The P-ll/lll trial involves assessing of REGN-COV2 (8gm/2.4gm) + SOC vs PBO + SOC in 524 patients with mild-to-moderate COVID-19 in an outpatient setting […]

Shots: The P-II/III ASCEND trial involves assessing olipudase alfa 3 mg/kg (IV, q2w) vs PBO in 36 adult patients with ASMD type B to evaluate […]

Shots: Part A of P-lll study involves assessing of Dupixent (qw, 300mg) vs PBO in 81 patients aged≥ 12 yrs. with EoE. The study resulted […]

Shots: The company reported results from two real-world studies of Renflexis (infliximab-abda) in patients with IBD registered in the US Veteran Affairs Healthcare System database. […]

Shots: The companies present the findings from a new exploratory sub-analysis of the P-III EMPEROR-Reduced study demonstrating that Jardiance (empagliflozin) reduced the risk of adverse […]

Shots: The additional insight from the P-IIb CaLIPSO study showed that SNF472 slowed the progression of CAC across key subgroups of patients on hemodialysis participating […]

Shots: GSK reported that its RSV vaccines for maternal immunization (GSK3888550A) and older adults (GSK3844766A) were well-tolerated and highly immunogenic in P-I/II clinical studies. Both […]

Shots: The company reported the positive findings of a pooled analysis of six ongoing clinical studies which includes P-IIb/IIIb LATTE-2, ATLAS, ATLAS-2M, FLAIR, POLAR, and […]

Shots: The P-III study involves assessing Xywav vs PBO in patients aged ≥ 7yrs. with cataplexy or EDS with narcolepsy Results: study met its 1EPs […]

Shots: The P-I clinical trial involves assessing of a single dose of either NeuLara, US- or EU-sourced Stelara in ~200 healthy volunteers. NeuLara is NeuClone’s […]

Shots: AstraZeneca’s Tagrisso has received sNDA’s acceptance and has been granted PR in the US for the adjuvant treatment of patients with early-stage (IB, II, […]

Shots: The P-III CHRONOS-3 study involves assessing of Aliqopa + rituximab vs PBO + rituximab in 458 patients with relapsed indolent NHL who have received […]

Shots: The company reports NDA submission and request for PR to the US FDA for hyperpolarised 129Xenon gas used to evaluate the pulmonary function and […]

Shots: The Rolling Submission is based on the preliminary results from pre-clinical and early clinical studies in adults, which shows that BNT162b2 triggers the production […]

Shots: The P-III STR1VE-EU study involves assessing Zolgensma (IV) in patients with SMA Type 1 aged <6mos. at the time of gene therapy, with 1 […]

Shots: The P-II COMPARE study assessing SEL-212 (once monthly) vs pegloticase (twice monthly) in 170 patients with chronic refractory gout SEL-212 showed a numerically higher […]

Shots: The P-III ILLUMINATE-B study involves assessing Lumasiran in 18 patients with PH1 under the age of 6 (3-72mos.), with an eGFR >45 mL/min/1.73 m2 or normal […]

Shots: The NDA submission is based on P-III UPTRAVI IV study he safety and tolerability of 20 patients with PAH temporarily switching from oral UPTRAVI […]

Shots: The NDA submission was submitted to NMPA’s CDE and is based on P-III VICTORIA study, seeking regulatory approval of vericiguat in China In Oct’2014, […]

Shots: The P-III ASCEMBL study involves assessing of Asciminib (ABL001) vs bosutinib in patients with Ph+ CML-CP, prior treated with two or more TKIs The […]

Shots: The first healthy volunteer has been dosed in a P-I study of GRAd-COV2 against COVID-19 which is conducted by the Lazzaro Spallanzani National Institute […]

Shots: BioMarin has submitted an NDA to the US FDA for vosoritide to treat children with achondroplasia The company also reported that EMA has begun […]

Shots: The NDA submission is based on three clinical studies in which Trilaciclib was administered prior to chemotherapy treatment in patients with SCLC and has […]

Shots: The MAA submission is based on dose-finding Part 1 and confirmatory Part 2 of the FIREFISH and SUNFISH studies evaluating the efficacy and safety […]

Shots: The US FDA has accepted the sBLA for a new self-administration option for Xolair across all approved indications in the US. The company anticipates […]

Shots: The BLA is based on P-III study evaluating the efficacy and safety of evinacumab (15 mg/kg, IV, q4w) in 65 patients aged ≥12yrs. with […]
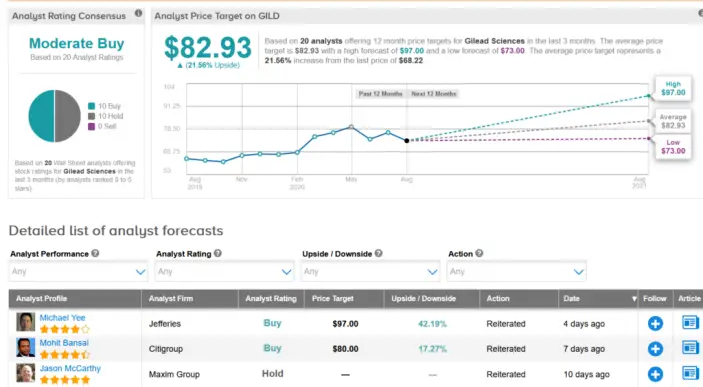
Shots: The NDA filing is based on P-III studies evaluating Veklury vs PBO, conducted by NIAID. The studies demonstrated that Veklury led to faster time […]

Shots: The two P-III studies, ASCLEPIOS I & II involves assessing of ofatumumab (20mg, monthly SC) vs teriflunomide (14mg) in 1882 patients aged 18-55yrs with […]

Shots: The P-II ELARA study involves assessing the efficacy and safety of Kymriah in adult patients with r/r FL across 12 countries in 30 sites […]

Shots: The P-III EMPEROR Trial involves assessing of Jardiance (10 mg) vs PBO in two studies EMPEROR-Reduced [NCT03057977, N=3,730] & EMPEROR-Preserved [NCT03057951, N=5,990] in patients […]

Shots: The P-III AD Up study involves assessing Rinvoq (15/30mg) + TCS vs PBO + TCS in patients (aged≥12) with the mod. to sev. AD […]

Shots: The P-III DAPA-CKD involve assessing of Farxiga ((dapagliflozin, 10mg, qd) + SOC vs PBO in 4,304 patients with CKD Stages 2–4 and elevated urinary […]

Shots: The MAA is based on P-III study assessing the efficacy and safety of vosoritide, further supported by the long-term safety and efficacy from the […]

Shots: The P-III Archway study involves assessing of PDS with ranibizumab, refilled @6mos. at fixed intervals, vs ranibizumab (0.5mg, monthly IVT) in 418 patients with […]

Shots: The P-III Measure Up 2 study involves assessing of Rinvoq (15/30mg) vs PBO in adults and adolescents aged 12-18yrs. or older with mod. to […]
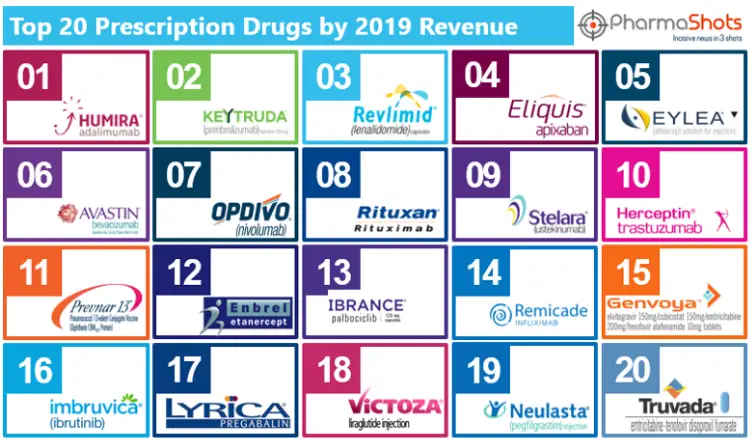
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]

Shots: The P-III OASIS-2 study involves assessing mirikizumab vs PBO & Cosentyx (secukinumab) in 1,465 patients with mod. to sev. PsO. The patients were randomized […]

Shots: The BLA submission is based on L-MIND P-II trial data results assessing Tafasitamab + lenalidomide in patients with r/r DLBCL and retrospective observational matched […]

Shots: The P-III study involves assessing of SCB-808 (SC) vs Enbrel (SC) in patients with ankylosing spondylitis (radiographic axial spondyloarthritis) to evaluate its safety, efficacy […]

Shots: The P-III study involves assessing of SCB-808 vs Enbrel in patients with AS (axSpA) via SC administration SCB-808 is being developed in the prefilled […]
Copyright © 2024 | WordPress Theme by MH Themes