Insights+: The US FDA New Drug Approvals in July 2020
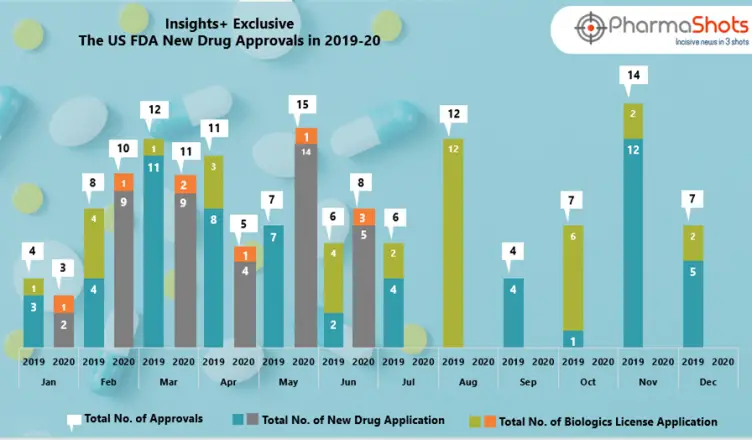
The US FDA has approved multiple NDAs and BLAs in July 2020, leading to treatments for patients and advances in the health care industry. The […]

The US FDA has approved multiple NDAs and BLAs in July 2020, leading to treatments for patients and advances in the health care industry. The […]

Shots: The approval was based on P-III trial from the ASCERTAIN study supporting P-I and P-II clinical studies which evaluated the safety and efficacy including […]

The approval is based on P-III trial assessing Wynzora Cream vs Taclonex Topical Suspension in 794 patients for the treatment of plaque psoriasis in adults […]

Shots: The BT designation is based on P-III ADAURA trial which involves assessing Tagrisso (80 mg) vs PBO in 682 patients with Stage IB, II, […]
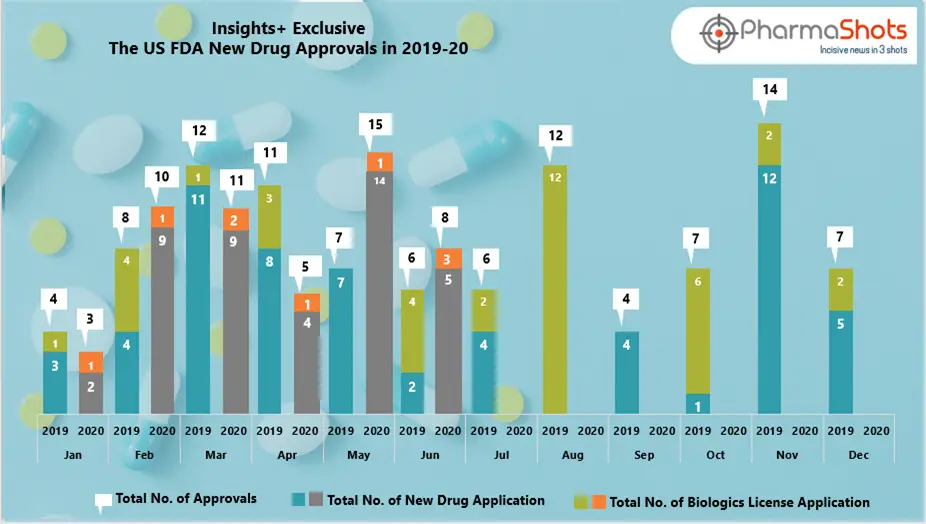
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]
Shots: The approval follows the US FDA Oncologic drugs Advisory Committee (ODAC) on 17 Dec based on P-III POLO trial, which involves assessing of Lynparza […]
Copyright © 2024 | WordPress Theme by MH Themes