
Regeneron hands ex-US rights to HoFH drug Evkeeza to Ultragenyx
Regeneron has enlisted the aid of Ultragenyx to add momentum to the rollout of its recently-approved Evkeeza outside the US, which was approved for an […]

Regeneron has enlisted the aid of Ultragenyx to add momentum to the rollout of its recently-approved Evkeeza outside the US, which was approved for an […]

In an interview with PharmaShots, Dr. Stanley T. Crooke, Founder, and Chief Executive Officer and Chairman of the Board at n-Lorem Foundation share his views […]

Evotec and Exscientia Initiate Human Clinical Trials of their Novel Immuno-Oncology Drug Published: Apr 9, 2021 | Tags: Evotec and Exscientia, Initiate, Human Clinical Trials, […]

Shots: The companies collaborated to develop the personalized medicines for patients with ultra-rare diseases. The collaboration involves additional resources, financial contribution, and experience from both […]

Angelman syndrome (AS) is a complex genetic rare disorder that affects the nervous system. The first sign of a child suffering from Angelman syndrome is […]

Roche’s Phesgo (Perjeta + Herceptin) Receives EC’s Approval for the Treatment of HER2-Positive Breast Cancer Published: Dec 23, 2020 | Tags: (Perjeta + Herceptin), EC’s […]

Shots: Mereo to receive $50M upfront & is eligible for ~$254M as milestones along with royalties on sales of therapy outside the EU. Ultragenyx will […]
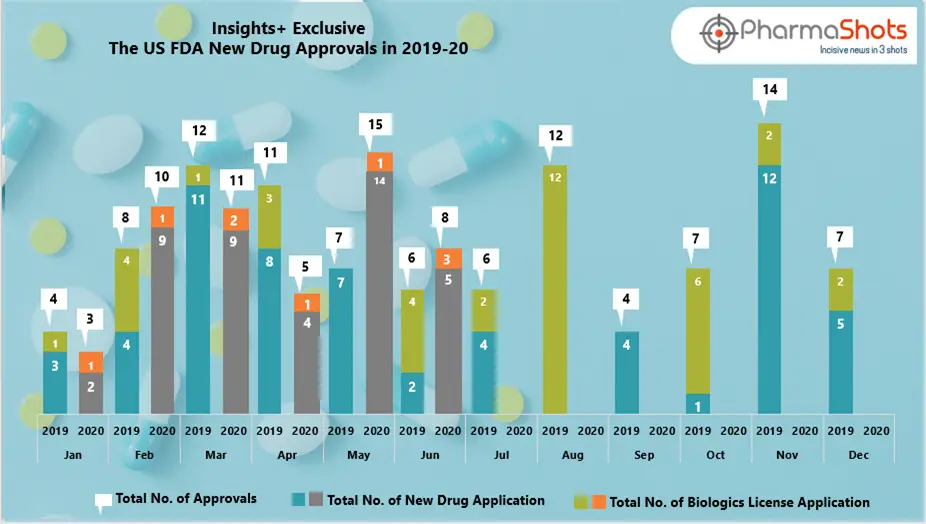
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]
Copyright © 2024 | WordPress Theme by MH Themes