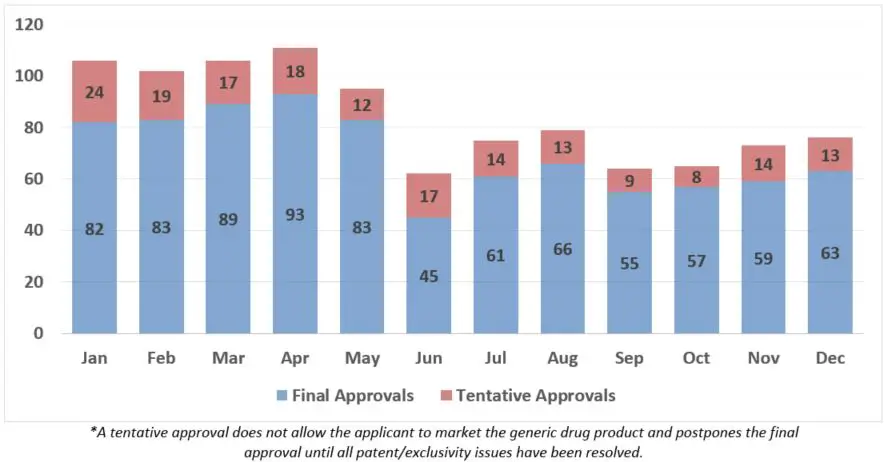
This report provides monthly activities metrics for the FDA Generic Drugs Program, outlining agency actions on abbreviated new drug applications. Data includes approvals, applications received, complete responses, and more.

Copyright © 2024 | WordPress Theme by MH Themes