For some years now, Amgen has been working on a technology called “BiTE“, which stands for “bispecific T-cell engager“. It fits into the general “bifunctional molecule” idea that’s so popular these days. It’s a big deal in protein localization and cellular localization, but there’s a longer history in trying to apply the idea to antibodies. What if each arm of the “Y” structure of a typical antibody bound to a different antigen? That’s not how the body grows them, but we could find uses for such things. There is, in fact, one of these on the market now: emicizumab, which brings together two proteins in the clotting factor cascade in a way that patients with hemophilia A can’t.
You can extend that idea to an even less natural-looking structure by moving to “nanobody” type proteins (single-chain antibodies) and linking them together. The first of these to make it through the clinic was blinatumomab, and its mechanism of action will tell you why Amgen has been sinking so much money into the idea after buying the protein’s original developer (Micromet) in 2012. Bilinatumomab has one small antibody domain that targets a surface protein called CD19, which is found specifically on B cells. There’s a short flexible linker between that one and another antibody domain that targets CD3, a surface protein on T cells. So blinatumomab should, in theory, force T cells over into proximity with B cells, while the binding to CD3 also activates the T cells to start exerting their cytotoxic effects. In a healthy individual this would be a spectacularly bad idea, but if you have B-cell leukemia, it could be just the thing.
If you’re into this sort of thing, you will already have noted the general similarity between such an approach and chimeric antigen receptor T cell therapy. CAR-T involves re-engineering a patient’s own T cells to recognize surface antigens such as CD19 to the same end: causing (in that case) a T-cell attack on the excess B cells in leukemia. Here’s a recent paper comparing the two approaches, but it needs to be noted that it’s an Amgen paper and thus could be more disposed towards the BiTE technology. Both approaches can be effective, with similar side effects (cytokine release syndrome being a major one), and both can show relapses over time. CRS is a real concern with all such approaches – in 2019, Regeneron reported two deaths from cytokine release in a clinical trial of one of their own bispecific antibody candidates (targeting CD3 again for the T cells and CD20 for follicular lymphoma). Interestingly, there’s at least one report of a patient with acute lymphoblastic leukemia who relapsed after CAR-T treatment who then went on blinatumomab and showed a complete remission. As with all immuno-oncology, we’re still finding our way on these things.
One of the ways that Amgen has been trying to improve their BiTE molecules is through improving their half-lives. They’ve been working on one of those (pavurutamab, AMG 701) that brings in T cells as before via CD3 and also targets the B-cell maturation antigen protein (BCMA) to treat multiple myeloma. But in their earnings release the other day, the company disclosed that they’re pausing trial enrollment due to some apparent toxicity events as they bring up the dosing levels in Phase I – that’s the only thing it can mean when you say you’re talking with the FDA about “optimizing safety monitoring and mitigation”. They also disclosed that they’re pausing another longer-half-life BiTE (AMG 673) that targets CD3/CD33 for AML – the company says that they’re going to gather more data in the clinic on the original bifunctional (AMG 330) they have in that combination before proceeding with this one. And yet another BiTE, this one (AMG 596) combining CD3 and an EGFR variant found in glioblastoma, has also been put on hold, and the only only reason given is “while we prioritize our portfolio”. As an aside, don’t you just love press release phrasing?
Now, they still have BiTE programs going – the release specifically mentions AMG 757, targeting delta-like ligand 3 (DLL3) for small cell lung cancer. But the path to extending clinical benefit with new immuno-oncology ideas has not been a smooth one, and it’s not going to be graded and paved any time soon. Amgen had some of the earlier Micromet-derived programs fail on them, such as AMG 110, a CD3/EpCAM (epithelial cell adhesion molecule) candidate, and there will absolutely be more failures to come.
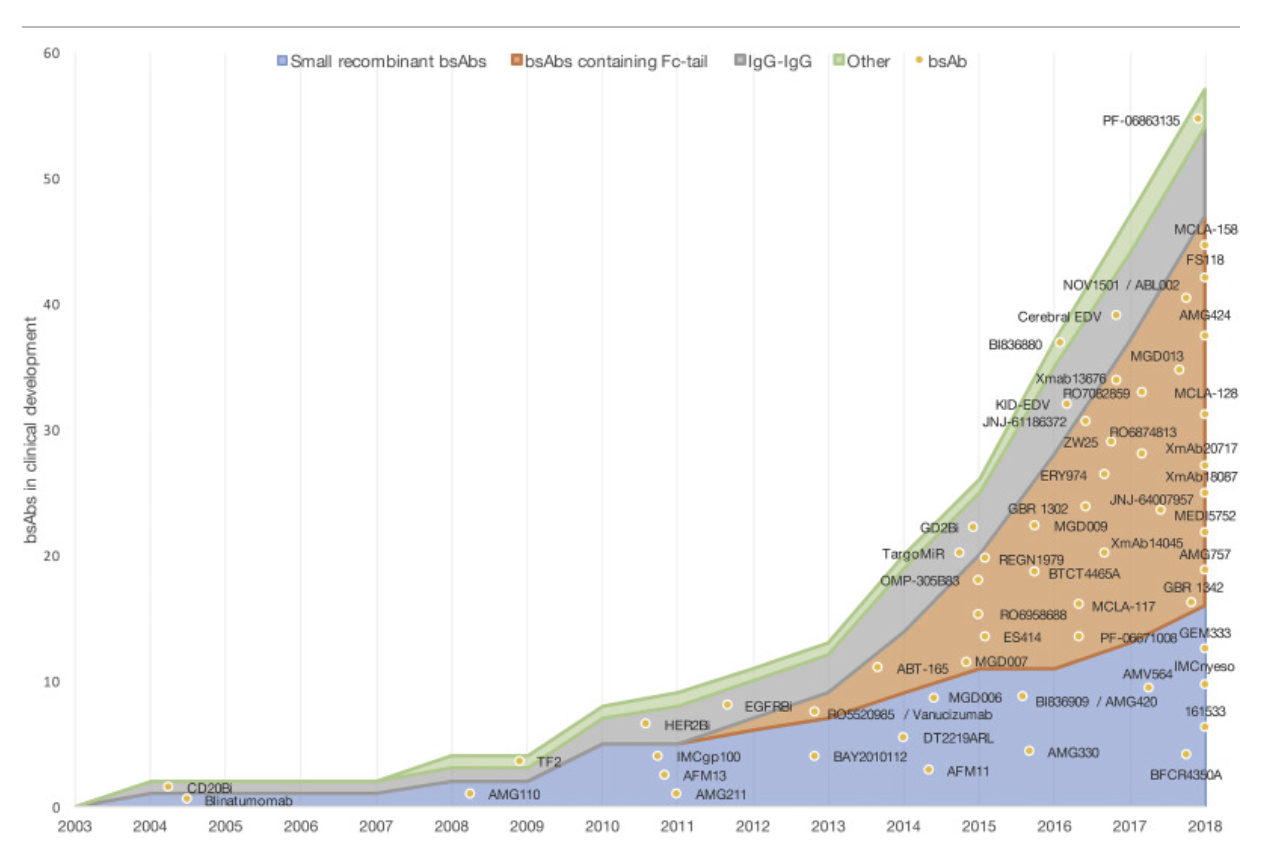
But not all of those will be Amgen’s, nor will all the successes. I haven’t even mentioned another parallel effort from MacroGenics, their “DART” (dual affinity re-targeting) platform and their “TRIDENT” one that brings in a third protein. They’ve gone after some similar targets (there are a number of obvious places to start in immuno-oncology, but the list is not as long as you might like). For example, here’s their CD3/CD19 program in an early iteration. There are applications outside oncology, too, of course, and they just signed a deal last month for one of those with J&J/Janssen (no details!) Another such platform is “CrossMab” from Roche, who have their own technology for making specific antibody light chain/heavy chain combinations. They’re all over the clinic as well, and there are plenty of others (see below!)
This 2019 review will give you an idea of the landscape in this area. It’s anyone’s guess what this will all end up looking like, but we’re going to learn a lot about immunology along the way, and the successes will very much be worth seeing. I’ll leave everyone with the graphic below, from that review paper – this is what an expanding field of research looks like and no mistake: