
Chasing rivals, AbbVie files migraine prevention drug atogepant in EU
AbbVie has submitted its oral CGRP inhibitor atogepant for prevention of both episodic and chronic migraine in the EU as it tries to catch up […]

AbbVie has submitted its oral CGRP inhibitor atogepant for prevention of both episodic and chronic migraine in the EU as it tries to catch up […]

AbbVie is pulling out of its collaboration with Alector on the development of Alzheimer’s disease candidate AL003, which aims to tackle the neurodegenerative disease by […]

AbbVie paid Alector $205 million up front five years ago to begin an R&D partnership on two drugs intended to bring an immunotherapy approach to […]

A report by the Senate Finance Committee has concluded that AbbVie exploited a financial loophole introduced by former President Donald Trump in 2017 to avoid […]

Pharmaceutical giant AbbVie generates almost all of its sales in the United States but has allegedly exploited former President Trump’s tax law to shield much […]

AbbVie has filed for approval of its oral CGRP inhibitor Qulipta for prevention of chronic migraine, seeking to leap ahead of its main rival in […]

AbbVie’s IL-23 inhibitor Skyrizi trailed some of its rivals in its first indication psoriasis, but has leapfrogged into first place in Crohn’s disease after getting […]

AbbVie drug Skyrizi is now approved for Crohn’s disease, making it the first treatment for the disorder that addresses a particular protein associated with inflammation. […]

AbbVie has reported new phase 3 data for its schizophrenia and bipolar disorder therapy Vraylar in treatment-resistant depression, as it waits for a decision by […]

AbbVie has filed for approval of its therapy for Parkinson’s disease, ABBV-951, which promises continuous control of symptoms and would be an alternative to current […]

AbbVie and Cugene have teamed up to research autoimmune diseases and cancer in an exclusive worldwide agreement. The Chicago-based pharma firm, AbbVie, received the option […]

AbbVie’s would-be blockbuster Rinvoq has notched up another positive trial in Crohn’s disease, showing that it can achieve clinical remission over a full year when […]

AbbVie’s Allergan subsidiary and Teva are reportedly considering making a $5 billion-plus offer to try to bring opioid liability lawsuits to a conclusion. According to […]

Pfizer’s $1.2 billion licensing deal with Biohaven for rights to oral migraine drug rimegepant is approaching a payoff after the European Commission approved the drug. […]

One of the leading companies in the bid to bring a new class of alpha-synuclein targeting drugs to market for Parkinson’s disease – AbbVie – […]

Two years after signing a $3.9 billion alliance with Genmab to find new cancer therapies, AbbVie has said it is ready to start talking to […]

AbbVie’s BCL-2 inhibitor navitoclax has reported phase 2 results in a trial involving rare and hard-to-treat blood cancer myelofibrosis, eyeing an indication not addressed with […]

AbbVie’s Qulipta has become the first oral drug in the CGRP inhibitor class to show efficacy in chronic migraine prevention in a pivotal trial, setting […]

Four years after spinning out from Belgium’s UCB, Syndesi Therapeutics and its SV2A modulator programme has been bought by AbbVie for $130 million upfront. The […]

AbbVie has acquired Syndesi Therapeutics, an early clinical neuroscience startup developing drugs that have potential applications in a range of cognitive disorders. Syndesi is based […]

AbbVie has filed with the FDA to extend the uses of its bipolar disorder therapy Vraylar to include adjunctive therapy for major depressive disorder, part […]

Prospects for Aldeyra’s dry eye disease candidate reproxalap were hit last month after the drug failed to hit its primary objective in a phase 3 […]

The FDA thinks an antibody-drug conjugate (ADC) developed by AbbVie could set new standards in treatment for certain patients with lung cancer, awarding the drug […]

Sanofi and Regeneron have another challenger to their big-selling drug Dupixent for atopic dermatitis, now that the FDA has approved Leo Pharma’s rival antibody tralokinumab. […]

AbbVie has ended speculation about what it will charge for its newly-approved Vuity therapy for presbyopia, a sight condition characterised by difficulty in focusing on […]

Jazz’ Sunosi (solriamfetol) Receives NICE Recommendation for the Treatment of Excessive Daytime Sleepiness Caused by Narcolepsy Published: Nov 19, 2021 | Tags: Jazz, Sunosi, solriamfetol, […]

With drug pricing controls back on the legislative table in the US, the influential Institute for Clinical and Economic Review (ICER) has published its third […]

Eisai Presents Results of Lecanemab in P-IIb Study 201 Study for the Treatment of Alzheimer’s Disease at CTAD 2021 Published: Nov 12, 2021 | Tags: […]

The development of digital medicine requires valid endpoints that can be used in clinical trials, and a consortium of pharma companies has just identified a […]

The FDA has approved the first and so far only pharmacological treatment for presbyopia, a sight condition that typically starts in middle age and involves […]

ADC Reports EMA’s Validation of MAA for Zynlonta to Treat R/R Diffuse Large B-Cell Lymphoma Published: 29 Oct, 2021 | Tags: ADC, EMA, MAA, Zynlonta, […]

AbbVie has reported that a subcutaneous infusion of Parkinson’s disease candidate ABBV-951 was more effective than standard oral therapy in a phase 3 trial, paving […]

Shots: The P-III M15-736 Study evaluates the efficacy, safety & tolerability of ABBV-951 to levodopa/carbidopa in 130 adult patients with advanced PD across 80 sites […]

LEO Reports 16-week Results of Tralokinumab in P-III ECZTRA 6 Trial for the Treatment of Moderate-to-Severe Atopic Dermatitis Published: 22 Oct, 2021 | Tags: LEO, […]

Boehringer Ingelheim has shown biosimilar manufacturers the way to win interchangeability with the world’s highest-grossing drug — but the company’s Cyltezo won’t come out until […]

Biosimilar competition to AbbVie’s big-selling Humira drug in the US has just been ratcheted up by the FDA approval of Boehringer Ingelheim’s Cyltezo – which […]

Shots: The opinion was based on the P-III KEEPsAKE-1 & 2 studies evaluate Skyrizi (150mg, SC) vs PBO in adults with active PsA who had […]

Shots: The approval is based on the P-III clinical trial program including Measure Up 1/2/AD Up that evaluates the efficacy & safety of Rinvoq (15/30 […]

Shots: Study 2 of the P-III SELECT-AXIS 2 clinical trial evaluates the efficacy & safety of upadacitinib (qd, 15mg) vs PBO in 314 adults with […]

BMS Reports EMA’s Validation of MAA for Relatlimab and Nivolumab as 1L Treatment for Unresectable or Metastatic Melanoma Published: Oct 1, 2021 | Tags: BMS, […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

AbbVie drug Quilpta, developed specifically for migraine prevention, is now FDA approved, the latest product in a new class of migraine medicines. The AbbVie drug […]

AbbVie has added a third drug to its arsenal in the crowded migraine market, getting FDA approval for Qulipta – its once-daily oral preventive therapy […]

Shots: The approval was based on a clinical program including P-III ADVANCE study, P-IIb/III study & P-III LTS study evaluated the safety, efficacy & tolerability […]

Swiss biopharma company ObsEva has filed its oral GnRH antagonist linzagolix for the treatment of uterine fibroids in the US, hoping to expand the number […]

AbbVie will take the lead on commercializing Regenxbio’s gene therapy in wet AMD and diabetic retinopathy. It’s AbbVie’s second major gene therapy deal this year, […]

AbbVie has expanded its ophthalmology pipeline via a licensing deal with Regenxbio, paying $370 million upfront to get its hands on a gene therapy for […]

Shots: Regenxbio to receive $370M up front in addition to ~$1.38B as development, regulatory and commercial milestones along with royalties on net sales of RGX-314 […]

The FDA has concluded its safety review of Pfizer’s JAK inhibitor Xeljanz and Xeljanz XR, requiring revised warnings for the drugs as well as others […]

Ascendis’ Skytrofa (lonapegsomatropin-tcgd) Receives the US FDA’s Approval for Pediatric Growth Hormone Deficiency Published: Aug 27, 2021 | Tags: Ascendis, Skytrofa, lonapegsomatropin-tcgd, US, FDA, Approval, […]

Pharmaceutical companies play a vital part in our lives and in helping us to live healthier lives. The pharmaceutical industry finds, develops, manufactures, and promotes […]

The FDA may be reluctant to trust JAK inhibitors, but the European Commission is more willing, and has just handed a first-in-class approval in atopic […]

Shots: The approval is based on 3 P-III studies i.e., Measure Up 1/2/AD Up evaluating the efficacy & safety of Rinvoq (15/30 mg, qd) as […]

Janssen Presents Results of Rybrevant (amivantamab-vmjw) in P-I CHRYSALIS Study for Advanced NSCLC with METex14 Mutations at WCLC Published: Aug 20, 2021 | Tags: Astellas, […]

Shots: The P-III ADVANCE trial evaluates efficacy, safety & tolerability of atogepant (PO, 10/30/60 mg, qd) vs PBO in 910 patients with EM who experienced […]

Junshi and Coherus’s Toripalimab Receive the US FDA’s Breakthrough Therapy Designation as 1L Treatment of Nasopharyngeal Carcinoma Published: Aug 13, 2021 | Tags: Junshi, Coherus, […]

Immunology deals with physiological functioning of the immune system in states of both health and disease as well as malfunctions of the immune system in […]

Shots: AbbVie selects two DUB targets to advance it to the next stage of drug discovery based on the results from in vitro & in […]

Shots: AbbVie has terminated the license agreement for abicipar pegol to treat nAMD and DME. Additionally, Molecular Partners will reacquire the development and commercial rights […]

AbbVie will not be pursued any further by the US government over allegations that it used ‘sham’ litigation to keep competition to its AndroGel testosterone […]

Roche Publishes the Results of Evrysdi (risdiplam) in FIREFISH Study to Treat of Type 1 SMA at NEJM Published: July 30, 2021 | Tags: Roche, […]

AbbVie and Alphabet spin-out Calico Life Sciences have agreed to fund their seven-year-old partnership seeking treatments for diseases of ageing with another $500 million apiece. […]

AbbVie and Alphabet spin-out Calico Life Sciences have agreed to fund their seven-year-old partnership seeking treatments for diseases of ageing with another $500 million apiece. […]

If you are in middle aged and find you have to hold your phone at arm’s length to read, you may have presbyopia – a […]

Shots: The P-III GEMINI 1 study involves assessing AGN-190584 (pilocarpine 1.25%, qd) ophthalmic solution in 323 patients in a ratio (1:1) with presbyopia for 30days. […]

Lineage Signs an Exclusive Option Agreement with Amasa Therapeutics to Supply and Use of Clinical-Grade Hystem Published: June 18, 2021 | Tags: Lineage, Option Agreement, […]

Shots: The P-III GLOW study evaluates Imbruvica + Venclexta/Venclyxto (I+V) vs chlorambucil + obinutuzumab (C+O) in 211 patients in a ratio (1:1) aged ≥65yrs. with […]

Vertex and CRISPR Therapeutics Present New Data of CTX001 from 22 Patients with TDT and SCD at EMA 2021 Published: June 11, 2021 | Tags: […]

Shots: The P-III CLL14 trial assessing the efficacy & safety of a combined regimen of Venclyxto/Venclexta + obinutuzumab vs obinutuzumab and chlorambucil in 432 patients […]

Shots: Caraway to receive ~$267M including $17M up front in cash and remaining as option payment & milestones and is eligible for additional regulatory and […]

Shots: The approval is based on data from two P-III studies i.e SELECT-PsA 1 & 2 evaluating the safety & efficacy of Rinvoq vs PBO […]

AstraZeneca has thrown down the gauntlet to AbbVie and Johnson & Johnson with new data for Calquence in chronic lymphocytic leukaemia (CLL) that it says […]

Tonix Pharmaceuticals Presents Results of TNX-102 SL in P-III RELIEF study for the Management of Fibromyalgia at ASCP 2021 Published: June 4, 2021 | Tags: […]

Shots: The P-III SELECT-COMPARE study evaluates the safety and efficacy of Rinvoq (15 mg, qd) vs PBO and Humira (SC, 40mg, every other week) in […]

Seelos’ SLS-005 (Trehalose) Receives EC’s Orphan Drug Designation to Treat Amyotrophic Lateral Sclerosis Published: May 28, 2021 | Tags: Seelos, SLS-005, Trehalose, EC, Orphan Drug […]

Myovant has chalked up a second approval for its flagship drug relugolix from the FDA, becoming the first once-daily oral drug in the US to […]

Shots: The approval is based on P-III VIALE-A and P-Ib M14-358 studies. The P-III study involves assessing Venclyxto + azacitidine in patients with newly diagnosed […]

Biosimilars firm Alvotech has filed a lawsuit that challenges AbbVie’s legal defences surrounding the inflammatory disease drug Humira in the US. Alvotech is challenging the […]

Shots: Soliton to receive $22.60 /share in cash for its all outstanding share making the total deal value $550M The acquisition adds Soliton’s Rapid Acoustic […]

Capsida Biotherapeutics’ technology can engineer viral vectors that deliver gene therapies to central nervous system cells. With that capability gave the startup was able to […]

BMS’ Opdivo + Yervoy and Onureg Receive EC’s CHMP Positive Opinion for MPM and AML Published: Apr 23, 2021 | Tags: BMS, Opdivo, Yervoy, Onureg, […]

Shots: The approval is based on P-III ENVISION I study that involves assessing the efficacy, safety, and PK of Humira (SC) in pediatric patients aged […]

The first quarter of 2021 highlights new approvals, M&A, and the clinical data of COVID-19 vaccines. The companies expand their working in COVID-19 by targeting […]

Evotec and Exscientia Initiate Human Clinical Trials of their Novel Immuno-Oncology Drug Published: Apr 9, 2021 | Tags: Evotec and Exscientia, Initiate, Human Clinical Trials, […]

Despite having the global pandemic, the biopharma companies were involved in multiple M&A, licensing deals. The major focus of the year remains the production and […]

Shots: The submission is based on two P-III studies KEEPsAKE-1 & -2 studies involves assessing Skyrizi (150mg) vs PBO in adult patients with active PsA […]

Pfizer and BioNTech Report Results of BNT162b2 to Prevent COVID-19 Published: Apr 2, 2021 | Tags: Pfizer and BioNTech, Report, Results, BNT162b2, Prevent, COVID-19 Acceleron […]

Eli Lilly and Incyte Report Results of Olumiant (baricitinib) in P-III BRAVE-AA2 Study for Severe Alopecia Areata Published: Mar 5, 2021 | Tags: Eli Lilly […]

Shots: Mitokinin to receive an up front and will continue to develop its PINK1 activator program through completion of IND enabling studies for its lead […]

Regeneron Reports IDMC Clearance of Efficacy for REGEN-COV (Casirivimab + Imdevimab) in P-III COVID-19 Outpatient Outcomes Trial Published: Feb 26, 2021 | Tags: Regeneron, Reports, […]

Shots: The approval is based on P-III ENVISION I study that involves assessing Humira (SC) vs PBO in pediatric patients aged 4-17yrs. with mod. to […]

Shots: The P-III U-ACCOMPLISH induction study involves assessing upadacitinib (45 mg, qd) vs PBO in patients with mod. to sev. UC The study met its […]

Visby’s POC COVID-19 Test Receives the US FDA’s EUA for Use in CLIA Waived Settings Published: Feb 12, 2020 | Tags: Visby, POC, COVID-19, Test, Receives, […]

The biopharma industry saw numerous deal terminations in 2020. Clinical and regulatory results, change in control limitations, and strategic reprioritizations were among the most common […]

Shots: Caribou to receive $40M up front in cash & equity investment, ~$300M in development, regulatory, and launch milestones along with additional commercial milestones and […]

The FDA has said it will wait for additional results from last week’s trial showing an increased risk of cardiac side effects with Pfizer’s arthritis […]

Roche Collaborate with Cambridge Quantum Computing to Develop Quantum Algorithm for Drug Discovery and Development Published: Jan 29, 2020 | Tags: Roche, Collaborate, Cambridge Quantum Computing, […]

JAK inhibitor Xeljanz is one of Pfizer’s top-selling drugs, despite a ‘black box’ warning for blood clots and cancers added to its label in 2019. […]

JAK inhibitor Xeljanz is one of Pfizer’s top-selling drugs, despite a ‘black box’ warning for blood clots and cancers added to its label in 2019. […]

Shots: The approval is based on data from three pivotal clinical trials P-II SELECT-PsA 1& 2 and P-II/III SELECT-AXIS 1 involves assessing of Rinvoq in […]

AstraZeneca’s fast-growing BTK inhibitor Calquence is already challenging class leader Imbruvica from Johnson & Johnson/AbbVie in the treatment of chronic lymphocytic leukaemia (CLL). Now, AZ […]

Life sciences companies are gearing up to enter new markets as they look to secure their positions after a spate of M&A, licensing, and research […]

In its report released last Tuesday, a nonprofit drug pricing research group claims that seven drugs have prices that do not align with any newly […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

The fourth quarter of 2020 contains multiple initiations of clinical trials, big approvals, and numerous deals. COVID-19 related news remains at the peak in this […]

US cost effectiveness watchdog ICER found 10 examples of substantial price rises for top-selling medicines in 2019, and concluded that seven of those were not […]

Shots: The P-III ADVANCE & MOTIVATE studies involves assessing of Risankizumab (600/1200mg) vs PBO in adult patients with mod. to sev. CD who had an […]

Almirall and Tyris Collaborate to Develop Next Generation Gene Therapies Published: Jan 7, 2020 | Tags: Almirall, Tyris, Collaborate, Develop, Next Generation, Gene Therapies Sarepta […]

AbbVie is in a race against time as US patent expiry of its Humira inflammatory diseases blockbuster looms in 2023 – but it just scored […]

Shots: The two P-III studies KEEPsAKE-1 & -2 involves assessing Skyrizi (150mg) vs PBO followed by risankizumab (150mg, @24wks.) in patients with active PsA who […]

AbbVie is to begin clinical development of an antibody designed to neutralise the SARS-CoV-2 coronavirus after licensing the therapy in from Harbour BioMed and Utrecht […]

Some of the biggest legal stories of the year include patients attempting to mount a class action lawsuit against Mayo Clinic after an employee improperly […]

Shots: AbbVie reported pooled analysis and long-term integrated analysis of the P-lll RESONATE-2 and ILLUMINATE study evaluating Imbruvica as 1L treatment of high-risk patients with […]

Eli Lilly’s buyout of Loxo Oncology last year has already yielded one approved drug, and it now has a path to market for a second […]

Richter Acquires Janssen’s Evra Transdermal Contraceptive Patch Assets for $263.5M Published: Dec 3, 2020 | Tags: Asset Purchase Agreement, Evra, Gedeon Richter, Janssen Pharmaceutical, Signs, […]

Shots: Frontier to receive ~$100M including $55M upfront and remaining as milestones within the first 12mos. of collaboration and is eligible for development & commercial […]

Shots: The approval of additional indication is based on a P-lll study involve the assessing the efficacy and safety of Humira targeting the patients with […]

Previously treated patients with chronic lymphocytic leukaemia (CLL) in England will get a chemotherapy-free treatment option after NICE recommended NHS funding for a combination of […]

Shots: The P-III LIMMitless study is designed to evaluate the long-term safety & efficacy assessing risankizumab (150 mg q12wks.) continuous risankizumab with a loading dose […]

The third quarter of 2020 was flooded with major acquisitions in the pharma and biotech industry along with multiple approvals. Starting with the latest acquisitions, […]

Akebia Presents Results of Vadadustat in P-lll INNO2VATE Program for Anemia Due to CKD at ASN Kidney Week Published: Oct 23, 2020 | Tags: Akebia, […]

Immunology is an important branch of science which deals with the study of the immune system. The immune system is a highly regulated and balanced […]

Shots: The company submitted applications to the US FDA & EMA seeking approval for Rinvoq (upadacitinib) for adults (15mg/30mg, qd) and adolescents (15mg, qd) with […]

Combination therapies involving Roche and AbbVie’s cancer drug Venclexta have been formally approved by the FDA in acute myeloid leukaemia, following supportive data from late-stage […]

Shots: The approval is based on P-lll VIALE-A & VIALE-C studies and updated data from the P-Ib M14-358 and the P-I/II M14-387 studies. The P-III […]
Sobi & Selecta Report Results of SEL-212 in P-II COMPARE Study for Chronic Refractory Gout Published: Oct 1, 2020 | Tags: Chronic Refractory Gout, COMPARE […]
Shots: AbbVie and Harvard University enter a $30M research alliance to study & develop novel therapies against emergent viral infections with an initial focus on […]

Shots: Morphic will receive a $20M as license fee and is eligible to receive milestones and royalties on sales of any commercialized therapy while AbbVie […]

Shots: Voyager retains full rights to the vectorization technology and novel vectorized Abs emerges from the collaboration and has the right to pursue vectorized Ab […]

Three of the companies making drugs used in the Phase II I-SPY COVID-19 study – Amgen, AbbVie and Takeda – announced the patient enrollments Monday. […]

Shots: The P-III AD Up study involves assessing Rinvoq (15/30mg) + TCS vs PBO + TCS in patients (aged≥12) with the mod. to sev. AD […]

Shots: The P-III Measure Up 2 study involves assessing of Rinvoq (15/30mg) vs PBO in adults and adolescents aged 12-18yrs. or older with mod. to […]

Gilead Sciences’ run of oncology-focused deals has continued with an option to buy cancer immunotherapy specialist Tizona – even though the biotech’s lead drug candidate […]

Shots: Gilead invests $300M to obtain 49.9% equity interest in Tizona and receives an exclusive option to acquire remaining stakes of Tizona for up to […]
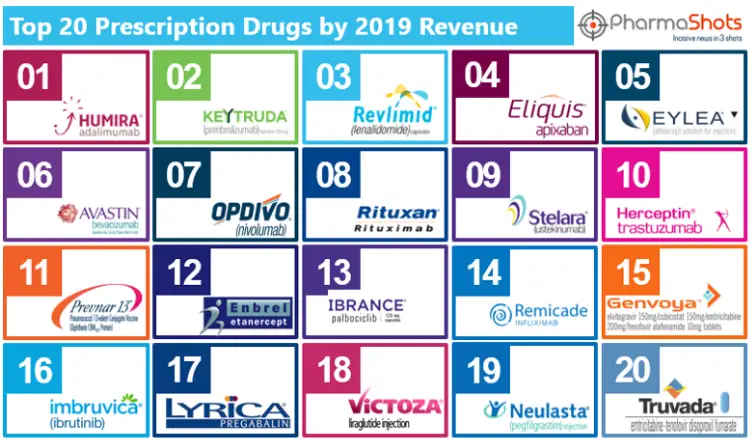
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
Copyright © 2024 | WordPress Theme by MH Themes