
On heels of new data, Gilead pays $455m for Asian Trodelvy rights
On the same day as it reported encouraging new data for its cancer drug Trodelvy, Gilead Sciences has agreed a deal to buy rights to […]

On the same day as it reported encouraging new data for its cancer drug Trodelvy, Gilead Sciences has agreed a deal to buy rights to […]
Gilead Sciences said it has statistically significant and clinically meaningful data from a pivotal test of its cancer drug, Trodelvy, in the most common type […]
In the space of a couple of months, Gilead Sciences’ Trodelvy impact on overall survival (OS) in the TROPICs-02 trial in advanced breast cancer has […]

The largest pharma companies have all set themselves ambitious targets in recent years to improve representation for women and minority communities in their businesses, but […]

Less than three years after being spun out of Oxford University, privately-held biotech MiroBio is heading for a takeover by Gilead Sciences in a $405 […]

Gilead Sciences’ investment in oncology has started to provide a solid return, with sales of cancer therapies breaching the $500 million threshold for the first […]

Cost-effectiveness watchdog NICE has recommended routine NHS use of two new therapies for women with advanced breast cancer – Gilead’s Trodelvy and Novartis’ Piqray – […]

AstraZeneca has signed a deal to buy US biotech TeneoTwo in a deal worth up to $1.27 billion that will boost its position therapies for […]

Gilead Sciences has refiled for FDA approval of its HIV capsid inhibitor lenacapavir – which only needs to be dosed every six months – after […]

The new FDA approval for Breyanzi moves the Bristol Myers Squibb cancer cell therapy into an earlier line of treatment for an aggressive type of […]

Breast cancer drugs and cell therapies were prominent in this year’s annual meeting of the American Society of Clinical Oncology. Here’s a recap of some […]

In March, Gilead Sciences reported positive results for its TROPiCS-02 study of Trodelvy, but the guarded tone of the announcement led to speculation that the […]

Gilead Sciences has more detail about breast cancer drug Trodelvy, a blockbuster hopeful it acquired two years ago. Additional data from a pivotal study were […]

Novartis’ CAR-T Kymriah has become the first therapy in the category to be approved for the treatment of relapsed/refractory follicular lymphoma, ahead of Gilead Sciences’ […]

Gilead Sciences has made another foray into the natural killer (NK) cell category, paying $300 million upfront to access Dragonfly Therapeutics’ TriNKET platform to find […]

Gilead has won a crucial court order in a wide-ranging lawsuit against a network of clinics, prescribers, medical labs and pharmacies in Florida that it […]

An FDA safety review has cleared Gilead Sciences to resume five clinical trials that were placed under a partial hold in January. The experimental Gilead […]

Gilead Sciences can restart pivotal trials of its cancer antibody magrolimab – the centre piece of its $4.9 billion acquisition of Forty Seven in 2020 […]

In draft guidance, NICE has said it cannot recommend Gilead Sciences’ Trodelvy in triple-negative breast cancer (TNBC), setting up a disparity in access to the […]

Gilead Sciences’ Kite Pharma has become the first drugmaker to get FDA approval to use a CAR-T therapy after just one earlier systemic therapy, moving […]

The FDA has given ViiV healthcare and Johnson & Johnson a boost in the market for long-acting injectable HIV drugs, approving a new regimen for […]

The results of Gilead’s much-anticipated TROPiCS-02 study of Trodelvy are in, and while the company says top-line data are positive, its guarded stance has rung […]

The FDA approved drugs from Johnson & Johnson and CTI BioPharma in the past week. Complete response letters were issued to Gilead Sciences, Amryt, and […]

Eli Lilly and Incyte’s JAK inhibitor Olumiant reduced the risk of death by 13% in hospitalised COVID-19 patients when added to other drugs, according to […]

A patent dispute between ViiV Healthcare and Gilead Sciences has been resolved, with Gilead agreeing to pay a $1.25 billion settlement as well as royalties […]

Gilead Sciences did not disclose details about the safety concern that led to an FDA partial clinical hold on five studies evaluating its cancer drug, […]

The FDA has placed a partial clinical fold on clinical trials of Gilead Sciences’ magrolimab, the centre piece of its $4.9 billion acquisition of Forty […]

Sierra Oncology myelofibrosis drug momelotinib achieved the main and secondary goals of a pivotal test and the biotech now plans to seek FDA approval. Sierra […]

The FDA has extended the emergency use authorisation for Gilead Sciences’ antiviral Veklury to include non-hospitalised patients with COVID-19, extending the uses of the drug. […]

GlaxoSmithKline’ majority-owned HIV unit ViiV has scored a key FDA approval, getting approval for its long-acting drug cabotegravir as the first injectable for pre-exposure prophylaxis […]

The insurers referred to deals Gilead struck with Janssen, Bristol Myers Squibb to prevent the generic version of its retroviral medications from being used as […]

Gilead Sciences has taken a big step towards positioning its CAR-T therapy Yescarta as an option for previously-untreated large B cell lymphoma (LBCL) with updated […]

A pair of trials have suggested that CAR-T therapies could offer an improved option for patients with large B cell lymphoma (LBCL) who have relapsed […]

A digital pill that records when it is taken has been shown to be highly effective at helping patients adhere to pre-exposure prophylaxis (PrEP) treatment […]

AstraZeneca and Daiichi Sankyo have reported new data with their antibody-drug conjugate for triple-negative breast cancer (TNBC), as they build a case to challenge Gilead’s […]

Merck & Co has halted enrolment in a pair of phase 3 trials of its experimental HIV drug islatravir, raising another warning flag about the […]

Gilead Sciences has submitted its marketing application for bulevirtide, its antiviral therapy for rare hepatitis delta virus (HDV) infections, to the FDA. If approved, bulevirtide […]

Gilead Sciences’ Trodelvy has met its objectives in a phase 2b trial in triple-negative breast cancer (TNBC) carried out in China by its partner Everest […]

Gilead Sciences and BeiGene, which started research alliances with Shoreline Biosciences earlier this year, are among the investors in the biotech’s latest financing. The preclinical-stage […]

Take two recently-approved immunotherapies for triple negative breast cancer (TNBC), put them together, and what do you get? Hopefully a new standard of care for […]

The FDA has approved Kite Pharma’s Tecartus as a treatment for adults with relapsed or refractory B-cell precursor acute lymphoblastic leukaemia (ALL), making CAR-T therapy […]

After 22 years at the helm of Belgian biotech Galapagos, chief executive Onno van de Stolpe has decided it is time to retire and hand […]

Gilead Sciences’ Kite Pharma has won a key victory on appeal in its long-running dispute with Bristol-Myers Squibb over patents relating to CAR-T therapies, overturning […]

Gilead Sciences’ Kite Pharma has mapped out another route to the development of allogeneic or ‘off-the-shelf’ cell therapies for cancer, using technology developed by Appia […]

Gilead Sciences just forged even closer ties with its cancer drug partner Arcus Biosciences, enticing the biotech’s chief medical officer Bill Grossman to a new […]

BioNTech has found a use for some of the windfall cash it has raked in from its COVID-19 vaccine, agreeing a deal to buy Kite […]

Galapagos has had to send its SIK inhibitor programme back to the drawing board after its lead compound GLPG3970 has failed two of three clinical […]

Gilead Sciences’ cell therapy Kite Pharma has bulked up its position in natural killer (NK) cell therapies, pledging up to $2.3 billion for a partnership […]

Novartis’ Kymriah has had the CAR-T therapy market for acute lymphoblastic leukaemia (ALL) to itself so far, but Gilead Sciences’ Kite subsidiary is now looming […]

Eli Lilly’s JAK inhibitor Olumiant has missed its main objective in a phase 3 trial in hospitalised COVID-19 patients, but had a significant effect on […]

Despite having the global pandemic, the biopharma companies were involved in multiple M&A, licensing deals. The major focus of the year remains the production and […]

Clinical tests of drugs from Gilead Sciences and Novo Nordisk have yielded encouraging results in nonalcoholic steatohepatitis (NASH), and the partners now want to see […]

FDA approval for Yescarta in follicular lymphoma makes it the first approved CAR-T treatment for this type of cancer. It also marks Gilead Sciences’ third […]

Gilead and Galapagos’ much-trumpeted R&D alliance has suffered another setback with the abandonment of ziritaxestat after a late-stage failure in idiopathic pulmonary fibrosis (IPF). Ziritaxestat […]

The decision to discontinue all clinical trials for ziritaxestat is the latest setback in a broad alliance between Gilead Sciences and Galapagos. Last year, the […]

The therapeutic vaccine will be based on Gritstone Oncology’s technology and will use antigens from Gilead Sciences. It’s the latest in a string of partnerships […]

Gilead Sciences says its antiviral Veklury should be effective against the new, more contagious COVID-19 variants discovered in the UK and South Africa. According to […]

Biopharma merger and acquisition activity in 2020 was mainly filled with late-stage, bolt-on acquisitions, which were orders of magnitude smaller than the mega M&A deals […]

Gilead Sciences’ Kite unit has teamed up with the UK’s Oxford BioTherapeutics (OBT) to develop a new clutch of cell therapy products for solid tumours […]

Shots: Gilead to acquire MYR for ~$1.4B in the all-cash transaction along with ~$364.73M as a milestone upon the US FDA’s approval of Hepcludex (bulevirtide) […]

A discussion on the different models healthcare stakeholders are developing to cover the cost of costly therapies will be just one of the compelling conversations […]

Gilead Sciences has the data it was hoping for in a pivotal trial of lenacapavir, an HIV drug that only needs to be dosed by […]

Gilead Sciences is the first and so far only company to claim FDA approval for a COVID-19 treatment with its Veklury antiviral, and its latest […]

The Food and Drug Administration approved Gilead’s antiviral drug as the first treatment for Covid-19 but just last week a WHO report showed that the […]

Gilead Sciences’ Kite Pharma unit is closing on approval of its second European approval for a CAR-T for cancer, after the CHMP backed its Tecartus […]
The European Commission has taken its advance orders for potential COVID-19 vaccines to more than 1.1 billion, after signing a supply deal for up to […]
The findings may make it possible to identify a subset of patients who experience poorer outcomes or more serious side effects, especially neurotoxicity.

While the White House and doctors at Walter Reed have sought to reassure the public that Trump’s condition is improving, he has received numerous drugs […]

The company said it was meeting real-time supply demands for Veklury (remdesivir) in the U.S. and anticipated meeting global demands for the drug worldwide. AmerisourceBergen […]

The company has raised $148 million so far and plans to raise the remaining $7 million by next month. Its lead candidate is entospletinib, a […]

While there had been some risks to filgotinib’s approval cited before, analysts expressed surprise at the decision, with one writing it would likely delay approval […]

The update to the partnership includes opt-in rights for Gilead on 15 drug targets, up from five under the original 2018 deal. Gilead will pay […]
UnitedHealthcare sent a notice to plan members stating that it would no longer cover Descovy to prevent HIV, while Truvada – after it goes generic […]
In a commentary, physicians and pharmacists wrote that percentages of Black, Latino and Native American patients in two Phase III studies of the drug that […]
Gilead Sciences had said in its second-quarter earnings that it would partner with companies in North America, Europe and Asia to manufacture the antiviral drug, […]
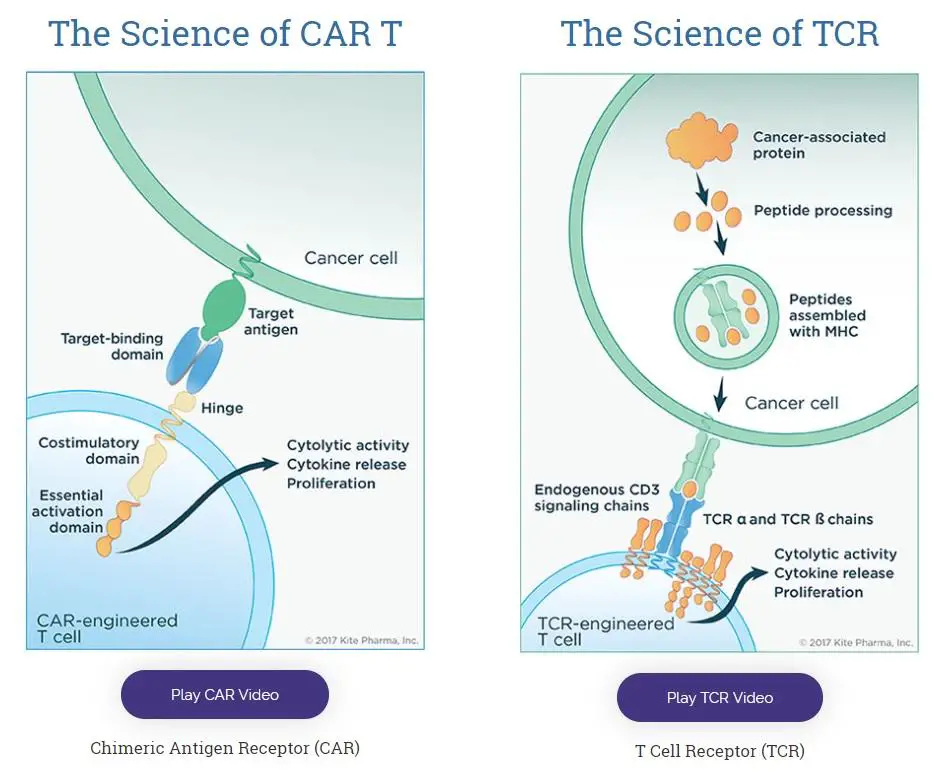
Gilead’s $11.9 billion purchase of Kite Pharma in 2017 didn’t follow the script, with two big write-downs in the value of the asset since then, […]
The agency approved Tecartus, previously developed under the name KTE-X19, as the first CAR-T therapy for mantle cell lymphoma. The company had previously won approval […]

Gilead Sciences’ run of oncology-focused deals has continued with an option to buy cancer immunotherapy specialist Tizona – even though the biotech’s lead drug candidate […]
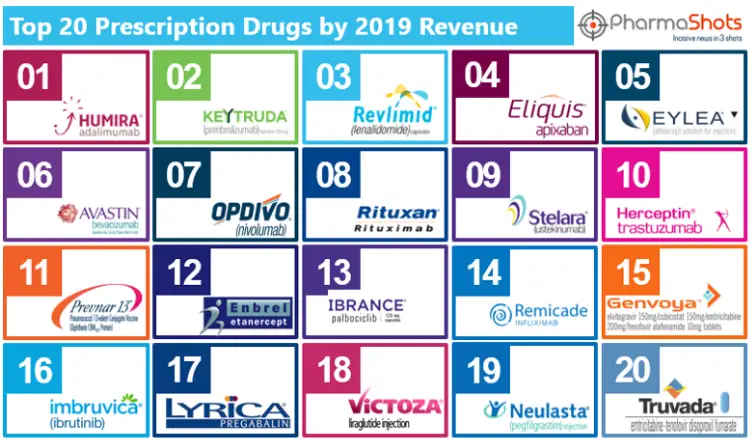
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
The company is developing biosensors, initially for NASH, that could be used in place of liver biopsy and have applicability in development of drugs for […]

The study is enrolling 60 healthy volunteers aged 18-45, but the company hopes it will form the basis of trials of the drug as an […]

Research on the drug’s molecular target dates back to the 1950s. A spokesperson for Gilead noted that the company anticipates potentially investing more than $1 […]
Copyright © 2024 | WordPress Theme by MH Themes