
After initial failure, Intercept Pharma feels new data can get NASH drug approval
Two years after the stinging FDA rejection of its drug for the fatty liver disease NASH, Intercept Pharmaceuticals has more safety and efficacy data from […]

Two years after the stinging FDA rejection of its drug for the fatty liver disease NASH, Intercept Pharmaceuticals has more safety and efficacy data from […]

After a phase 3 trial win, Madrigal Pharmaceuticals reckons it could succeed where so many other companies have failed, and finally bring a drug to […]

Already scrambling to refile its obeticholic acid (OCA) drug for non-alcoholic steatohepatitis (NASH) after the FDA rejected it last year, Intercept Pharma was hoping for […]

US remote healthcare specialist ChronWell has raised $6 million from investors that will be used to accelerate the rollout of its patient management programmes for […]

GlaxoSmithKline has joined the ranks of drugmakers trying to develop new drugs for non-alcoholic steatohepatitis (NASH) – a condition with no approved therapies and a […]

The 5th Antifibrotic Drug Development Summit (AFDD) will provide you with a deeper understanding of how to progress early-stage candidates and avoid late-stage failures, know the […]

Shots: Boehringer Ingelheim has accepted a GalXC RNAi candidate (DCR-LIV2) for advancement under the existing agreement b/w the companies for the discovery and development of […]

Shares in NGM Biopharma have plummeted after the US biotech said it would abandon development of aldafermin in non-alcoholic steatohepatitis (NASH), adding to a lengthening […]

Shots: The companies plan to initiate a P-IIb study assessing NovoNordisk’s semaglutide and FD combination of Gilead’s cilofexor + firsocostat vs PBO in ~440 patients […]

Shots: The companies plan to initiate a P-IIb study assessing NovoNordisk’s semaglutide and FD combination of Gilead’s cilofexor + firsocostat vs PBO in ~440 patients […]

Analysts are continuing to back Inventiva’s lanifibranor as a potential “best-in-class” drug for the fatty liver disease NASH, as the company prepares for the launch […]

Two years after starting to work together, Novo Nordisk and Dicerna have selected the first candidate from a joint project to find new, gene-silencing drugs […]

The startup, which licensed three NASH candidates from Eli Lilly, raised $87 million from the drugmaker and Deerfield Management.

Gannex received US FDA fast track designation for its NASH drug, ASC42 an FXR Agonist Gannex Pharma has received Fast Track designation approval from FDA […]

Gilead and Novo Nordisk have announced drug combinations for the fatty liver disease known as NASH involving the GLP-1 class drug semaglutide have checked out […]

Merck & Co is to pay $10 million to Hanmi to repurpose an obesity drug discarded by Johnson & Johnson, into a therapy for the […]
Hanmi will retain options for the drug, efinopegdutide, in Korea under the deal, in which Merck is paying the Seoul-based company $10 million upfront and […]

Shots: Hanmi to receive $10M up front, $860M as development, regulatory approval, and commercialization of efinopegdutide along with royalties on sales of the approved product […]
The company said it was formally terminating the Phase III study of the drug elafibranor in NASH and refocus its development of the drug on […]
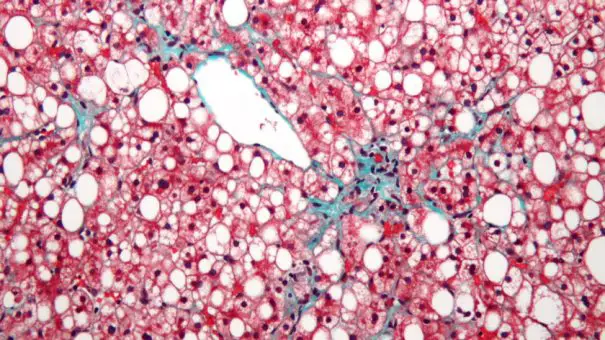
France’s Genfit has said its elafibranor drug as a treatment for non-alcoholic steatohepatitis (NASH) was unable to improve patient outcomes in a phase 3 study, […]
The company is developing biosensors, initially for NASH, that could be used in place of liver biopsy and have applicability in development of drugs for […]
Copyright © 2024 | WordPress Theme by MH Themes