
Gilead Reports Results of Veklury (remdesivir) in P-III Trial for the Treatment of COVID-19
Shots: The P-III trial evaluates the efficacy & safety of a 3-day course of Veklury (IV) vs PBO in a ratio (1:1) in 562 non-hospitalized […]

Shots: The P-III trial evaluates the efficacy & safety of a 3-day course of Veklury (IV) vs PBO in a ratio (1:1) in 562 non-hospitalized […]

The COVID-19 pandemic has highlighted the importance of ensuring a reliable supply of treatments across the globe at pace and speed. Jirair Ratevosian, executive director of […]

Gilead Sciences says its antiviral Veklury should be effective against the new, more contagious COVID-19 variants discovered in the UK and South Africa. According to […]

Shots: The EUA is based on ACCT-2 study assessing baricitinib (4mg, qd for 14 days or until hospital discharge) in combination with remdesivir vs PBO […]

Gilead Sciences is the first and so far only company to claim FDA approval for a COVID-19 treatment with its Veklury antiviral, and its latest […]

Gilead’s Veklury (remdesivir) has become the first COVID-19 drug approved in the US following a nod from the FDA. As an antiviral drug, remdesivir works […]

Shots: The approval is based on three studies including P-lll ACTT-1 study assessing the efficacy and safety of a 10-day treatment course of Veklury vs […]

Gilead’s Veklury (remdesivir) has failed to produce an effect on hospital stay and mortality in COVID-19 patients in a large global trial, casting doubt on […]
The European Commission has taken its advance orders for potential COVID-19 vaccines to more than 1.1 billion, after signing a supply deal for up to […]

While the White House and doctors at Walter Reed have sought to reassure the public that Trump’s condition is improving, he has received numerous drugs […]

The company said it was meeting real-time supply demands for Veklury (remdesivir) in the U.S. and anticipated meeting global demands for the drug worldwide. AmerisourceBergen […]

Date: October 19, 2020 Issue #: 1609 Summary: Remdesivir (Veklury – Gilead), an investigational antiviral drug administered by IV infusion, is now available through an […]
Darlene Dobry, Strategic Advisor, Medical Devices and Pharmaceuticals As the COVID-19 pandemic continues to bring the world’s economies, healthcare systems, and communities to their knees, […]

Shots: The company has launched Remdec at a price of $37.41 (Rs. 2800) for a 100mg lyophilized injection. The generic version is the most economical […]
In a commentary, physicians and pharmacists wrote that percentages of Black, Latino and Native American patients in two Phase III studies of the drug that […]
Shots: The NDA filing is based on P-III studies evaluating Veklury vs PBO, conducted by NIAID. The studies demonstrated that Veklury led to faster time […]
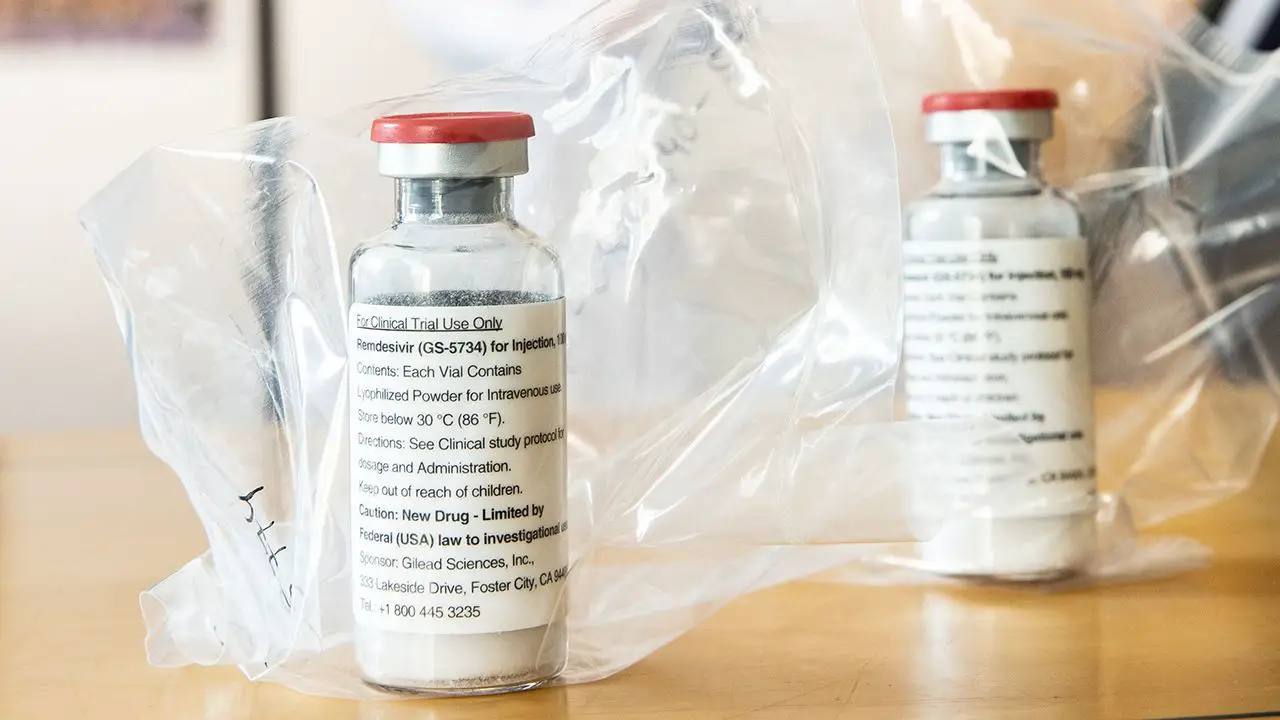
Gilead has filed its COVID-19 drug remdesivir with the FDA, to treat patients with severe disease, under the brand name Veklury. The drug is currently […]
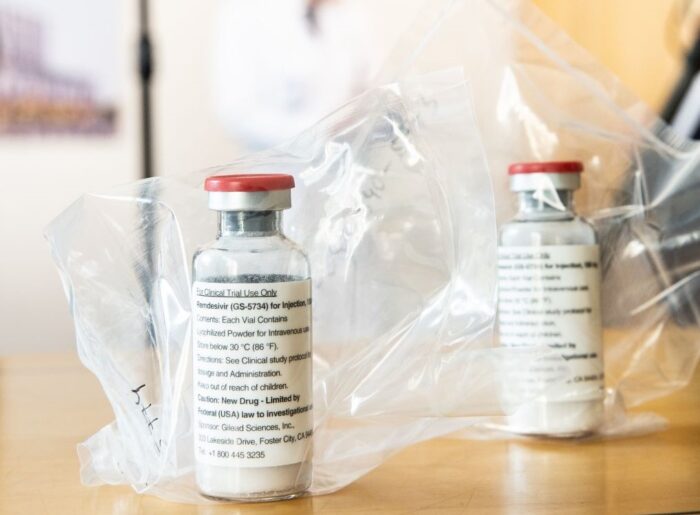
Gilead Sciences had said in its second-quarter earnings that it would partner with companies in North America, Europe and Asia to manufacture the antiviral drug, […]

Pfizer has signed a multi-year agreement with Gilead to manufacture and supply the COVID-19 antiviral remdesivir, which is also being tested in combination with other […]

A successful cocktail would also serve us in the long-run to treat the impacts of Covid-19 and keep those who are most vulnerable, the elderly […]
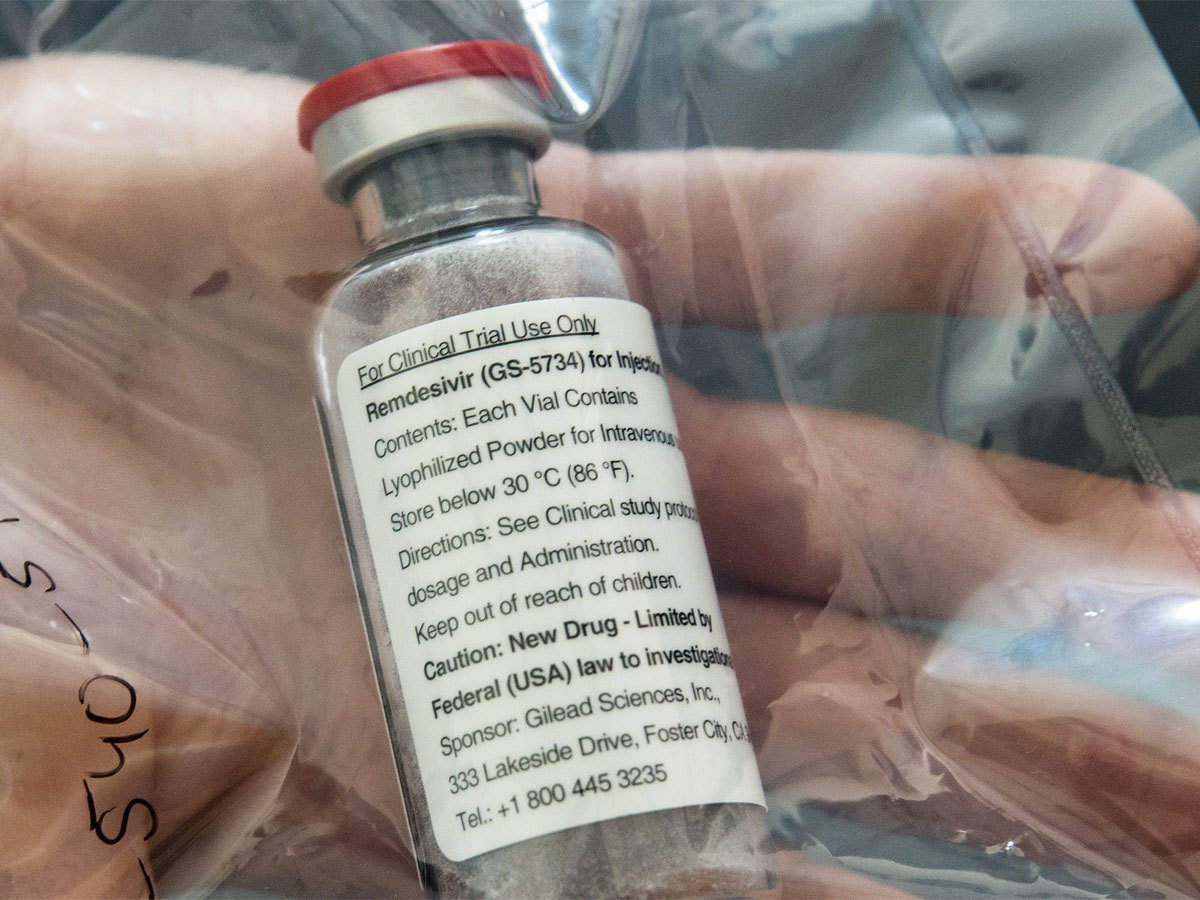
Shots: Mylan launches Desrem (remdesivir) addressing the unmet needs amid COVID-19. The drug is approved for the treatment of suspected or laboratory-confirmed incidences of COVID-19 […]

The study is enrolling 60 healthy volunteers aged 18-45, but the company hopes it will form the basis of trials of the drug as an […]
Copyright © 2024 | WordPress Theme by MH Themes