
Record Shingrix sales help GSK raise 2022 forecasts
In its first set of financial figures since spinning off its consumer health division, GSK has reported a 19% increase in revenues, driven by burgeoning […]

In its first set of financial figures since spinning off its consumer health division, GSK has reported a 19% increase in revenues, driven by burgeoning […]

Gilead Sciences has refiled for FDA approval of its HIV capsid inhibitor lenacapavir – which only needs to be dosed every six months – after […]

The FDA has given ViiV healthcare and Johnson & Johnson a boost in the market for long-acting injectable HIV drugs, approving a new regimen for […]

A patent dispute between ViiV Healthcare and Gilead Sciences has been resolved, with Gilead agreeing to pay a $1.25 billion settlement as well as royalties […]

GlaxoSmithKline’ majority-owned HIV unit ViiV has scored a key FDA approval, getting approval for its long-acting drug cabotegravir as the first injectable for pre-exposure prophylaxis […]

Merck & Co has halted enrolment in a pair of phase 3 trials of its experimental HIV drug islatravir, raising another warning flag about the […]

People living with HIV in England and Wales now have an alternative to daily oral tablets, after NICE backed NHS use of two long-acting injectable […]

Shots: ViiV Healthcare has reported a regulatory submission to the US FDA for the approval of a new dispersible tablet formulation of the fixed-dose combination […]

BMS Reports EMA’s Validation of MAA for Relatlimab and Nivolumab as 1L Treatment for Unresectable or Metastatic Melanoma Published: Oct 1, 2021 | Tags: BMS, […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

Shots: The P-III TANGO study evaluating the antiviral efficacy and safety of dolutegravir/lamivudine switching to a 2DR vs TAF based regimens in HIV-infected adults who […]

Shots: Shionogi to receive $27.2M up front, $20.4M upon the achievement of a clinical milestone along with royalties on net sales of S-365598 (HIV integrase […]

In an interview with PharmaShots, Dr. Kimberly Smith, Head of Global Research and Medical Strategy at ViiV Healthcare shares insights on the clinical data of […]

In an interview with PharmaShots, Max Laitaillade, Vice President, Head of Global Research Strategy, ViiV Healthcare shared his views on the development of Rukobia and […]

PhaseBio Signs a Supply Agreement with BioVectra to Support the Development and Commercialization of Bentracimab Published: Mar 12, 2021 | Tags: PhaseBio, Signs, Supply, Agreement, […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Shots: The P-IIa POC study involves assessing GSK3640254 (GSK’254, qd) in 34 treatment-naïve adults with HIV. The study is divided into 2 parts- in part […]

Shots: The P- IIIb ATLAS-2M study involves assessing long-acting cabotegravir and rilpivirine regimen for the treatment of HIV The ATLAS-2M 96wks. data reinforce the 1EPs […]

In an interview with PharmaShots, Dr. Kimberly Smith, Head of Research & Development, ViiV Healthcare shed light on the FDA’s approval and the 3yrs. data […]

Visby’s POC COVID-19 Test Receives the US FDA’s EUA for Use in CLIA Waived Settings Published: Feb 12, 2020 | Tags: Visby, POC, COVID-19, Test, Receives, […]

The European Commission has given a green light to the use of ViiV Healthcare’s Rukobia as a treatment for people with HIV that has developed […]

Shots: The MAA is based on P-III BRIGHTE study assessing Rukobia (600mg, bid) + OBT vs PBO in 371 HTE adults living with multidrug-resistant HIV. […]

ViiV Healthcare and London’s Westminster Hospital NHS Foundation Trust have launched a digital triage service for HIV patients supported by a mobile app. ViiV, a […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

Shots: The approval is based on P1093 & ODYSSEY (Penta20) studies assessing safety, tolerability & dose-finding of Tivicay (5mg) in pediatric patients aged 4wks.-18yrs. while […]

In a recent interview with PharmaShots, Kimberly Smith, MD, MPH, Head of Research & Development at ViiV Healthcare shared information on the positive findings presented […]

Roche’s Phesgo (Perjeta + Herceptin) Receives EC’s Approval for the Treatment of HER2-Positive Breast Cancer Published: Dec 23, 2020 | Tags: (Perjeta + Herceptin), EC’s […]

Shots: The approval is based on P-III ATLAS, FLAIR and ATLAS-2M studies assessing Vocabria (cabotegravir) + Janssen’s Rekambys (rilpivirine inj.) or Edurant (rilpivirine tablets) in […]

Shots: In the PROgress study, 200 people living with HIV were provided with a pre-visit survey to guide their conversation with their HCP and assess […]

Shots: This BT designation is based on P-IIb/III HPTN 083 study assessing cabotegravir (q8w) vs. FTC/TDF tablets (qd PO, 200/300 mg) in a study population […]

Akebia Presents Results of Vadadustat in P-lll INNO2VATE Program for Anemia Due to CKD at ASN Kidney Week Published: Oct 23, 2020 | Tags: Akebia, […]

Shots: The company reported the positive findings of a pooled analysis of six ongoing clinical studies which includes P-IIb/IIIb LATTE-2, ATLAS, ATLAS-2M, FLAIR, POLAR, and […]
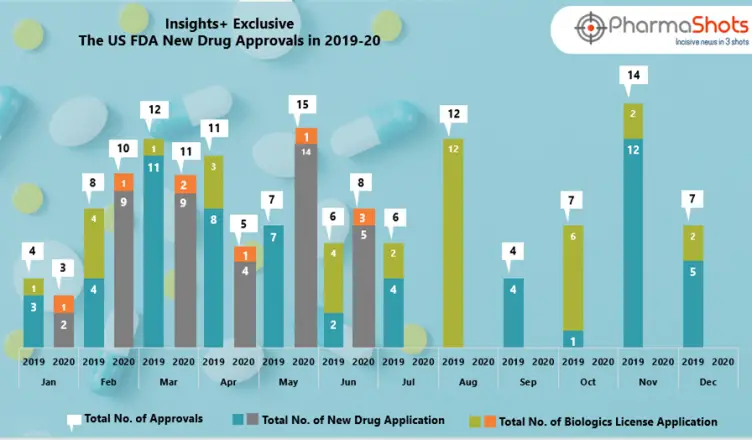
The US FDA has approved multiple NDAs and BLAs in July 2020, leading to treatments for patients and advances in the health care industry. The […]
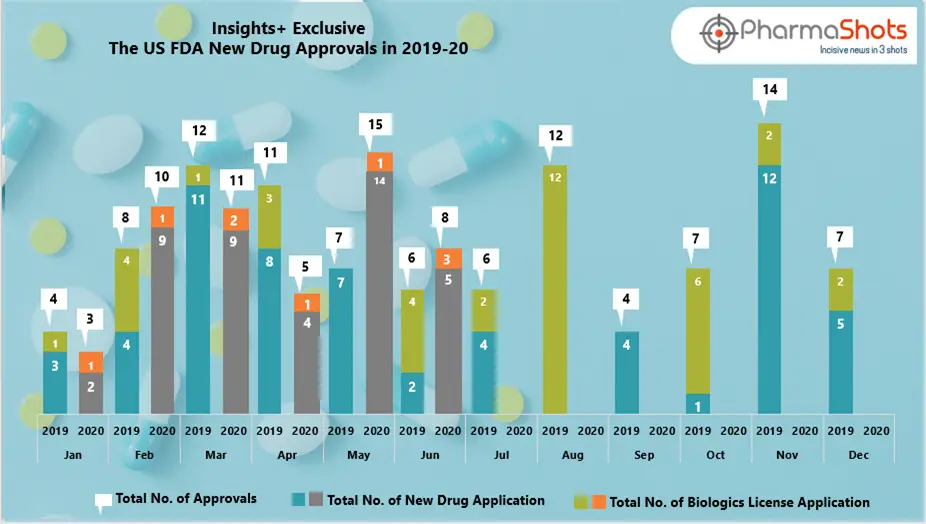
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]

The company said Tuesday that a final analysis of its Phase IIb/III study of cabotegravir showed greater efficacy in HIV prevention than Gilead’s Truvada. The […]
Copyright © 2024 | WordPress Theme by MH Themes