
Bayer tops pharma rankings for diversity, inclusivity
The largest pharma companies have all set themselves ambitious targets in recent years to improve representation for women and minority communities in their businesses, but […]

The largest pharma companies have all set themselves ambitious targets in recent years to improve representation for women and minority communities in their businesses, but […]

Less than three years after being spun out of Oxford University, privately-held biotech MiroBio is heading for a takeover by Gilead Sciences in a $405 […]

Beijing biotech Sironax has closed a whopping $200 million second-round financing that shows investors still have a desire to back companies working on RIPK1 inhibitors […]

Roche has been trumpeting the rapid uptake of its new eye disease drug Vabysmo as it takes on Regeneron and Bayer’s mighty Eylea in the […]

The conference highlights the convergence of digital health and drug development. Among the panel discussions are a focus on the state of AI in drug […]

Thousands more people with breast cancer in England look set to be eligible for routine treatment with Eli Lilly’s Verzenios, after new guidance from NICE […]

A personalised, treatment plan for patients with breast cancer is the first digital health product to emerge from a newly-announced partnership between digital health specialist […]

Eli Lilly and Incyte’s Olumiant has secured the first FDA approval for a systemic drug to treat alopecia areata, an autoimmune condition causing patchy hair […]

It has been eight years since Sanofi licensed rights to an over-the-counter version of erectile dysfunction (ED) drug Cialis from Eli Lilly, and the FDA […]

Eli Lilly’s home state of Indiana is set to benefit from a massive $2.1 billion investment by the pharma group, earmarked for two brand new […]

Eli Lilly has the results it was looking for in a second, confirmatory trial of its anti-IL-23p19 antibody mirikizumab in ulcerative colitis as it waits […]

Sanofi’s Genzyme unit has moved a step closer to securing EU approval for Xenpozyme – its enzyme replacement therapy for rare disease acid sphingomyelinase deficiency […]

The FDA approved Eli Lily drug Mounjaro as a treatment for type 2 diabetes. The decision gives the pharmaceutical giant a new competitor to a […]

Arkansas has become the latest US state to issue a legal challenge to the big three insulin producers – Eli Lilly, Sanofi and Novo Nordisk […]

AstraZeneca’s SGLT2 inhibitor Farxiga has hit the mark in a phase 3 heart failure trial that brings it back into contention with its main rival […]

Eli Lilly in a statement to investors is saying that its new experimental anti-obesity drug has caused patients in a clinical trail to lose up […]

Denmark’s Novo Nordisk has been almost single-handedly rebuilding the market for drugs to treat obesity over the last few years with its GLP-1 agonist drugs […]

Nektar has said it will cut 70% of its workforce in the wake of the comprehensive failure in trials of its immuno-oncology candidate bempegaldesleukin, which […]

A pharmaceutical giant reported a Phase 3 success for a potential blockbuster drug, while four other companies reported disappointing data from their respective clinical trials. […]

A group of pharma companies and academic groups have joined forces with the Digital Medicine Society (DiMe) to develop standardised, digital clinical measures that can […]

Boehringer Ingelheim and Eli Lilly’s SGLT2 inhibitor Jardiance has become the first drug in the class to be approved in the EU for all adults […]

A judge ruled the lawsuit against Eli Lilly for their Medicaid rebate payments will proceed to trial; Ely Lilly is accused of underpaying for Medicaid […]

With prospects of an early, accelerated approval for Roche’s Alzheimer’s disease candidate gantenerumab looking diminished, the drugmaker has launched a new four-year trial in the […]

Eli Lilly and Incyte’s JAK inhibitor Olumiant reduced the risk of death by 13% in hospitalised COVID-19 patients when added to other drugs, according to […]

Non-profit drugmaker Civica Rx has said it will launch biosimilars of three big-selling insulin products in the US by 2024 to help diabetic patients struggling […]

Boehringer Ingelheim and Eli Lilly have published data for their SGLT2 inhibitor Jardiance in acute heart failure, setting them on course to position the drug […]

The racial reckoning of the past couple of years has inspired many industries to take a look at their histories and practices.

Eli Lilly and Boehringer Ingelheim’s blockbuster diabetes drug Jardiance (empagliflozin) has been granted approval by the US Food and Drug Administration (FDA) for a wider […]

The Lilly Institute for Genetic Medicine is a new facility in Boston that will focus on the R&D of RNA and DNA-based therapies. Eli Lilly […]

Eli Lilly has reported phase 3 results with its ulcerative colitis challenger mirikizumab, as it waits for the readout of a second trial in the […]

ImmunoGenAlmost four years after backing out of an antibody-drug conjugate (ADC) alliance with ImmunoGen, Eli Lilly has come back to the table with a new, […]

Eli Lilly has been granted an emergency use authorisation from the FDA for bebtelovimab, its second COVID-19 antibody drug and a rival to GlaxoSmithKline and […]

FDA advisors have voted against approval of Eli Lilly and Innovent Biologics’ cancer immunotherapy sintilimab, undermining hopes of a new, lower-priced option in the PD-1/PD-L1 […]

The FDA has dropped a bomb on Eli Lilly’s marketing application for cancer immunotherapy sintilimab ahead of an advisory committee meeting due to take place […]

Eli Lilly has backed away from plans to file Alzheimer’s drug donanemab in the first quarter of this year, saying the proposal to limit reimbursement […]

Eli Lilly has said it is seeking permission to build a new biopharmaceutical manufacturing facility in Ireland that will be used to produce active ingredients […]

The FDA has limited the use of COVID-19 antibody therapies developed by Eli Lilly and Regeneron, on the back of data suggesting they have are […]

Use of the newly ineffective treatments produced by Regeneron Pharmaceuticals and Eli Lilly is highest in a dozen states. They include several Southern states with […]

A World Health Organization (WHO) panel has given the green light to two new drugs to treat COVID-19. US-based Eli Lilly’s baricitinib (Olumiant), a Janus […]

Sanofi and Regeneron have another challenger to their big-selling drug Dupixent for atopic dermatitis, now that the FDA has approved Leo Pharma’s rival antibody tralokinumab. […]

Three life science companies unveiled Series B rounds of funding Thursday, early Christmas gifts that top $219 million combined. Along with Verge Genomics, the other […]

Eli Lilly is building its cancer drug pipeline via a research alliance with Foghorn Therapeutic, a biotech whose technology discovers drugs addressing targets in a […]

Eli Lilly’s authorized antibody drug for treating and preventing Covid-19 infection in adolescents and adults is now authorized for pediatric patients, including newborns. While tests […]

Eli Lilly’s RET inhibitor Retsevmo can now be prescribed on the NHS for people with RET fusion-positive advanced non-small cell lung cancer (NSCLC) in England […]

Roche and Blueprint Medicines have opened a second front in their rivalry with Eli Lilly in RET-mutated cancers, after getting approval for Gavreto from the […]

Eisai Presents Results of Lecanemab in P-IIb Study 201 Study for the Treatment of Alzheimer’s Disease at CTAD 2021 Published: Nov 12, 2021 | Tags: […]

Shots: The sNDA is based on P-III EMPEROR-Preserved trial evaluates the safety and efficacy of Jardiance (10mg, qd) vs PBO in 5,988 patients with chronic […]

Shots: The P-III EMPEROR-Preserved trial evaluates the safety & efficacy of Jardiance (10mg, qd) vs PBO in 5,988 patients with chronic HFpEF with/out diabetes The […]

Eli Lilly has followed through on its plan to file for approval of its Alzheimer’s disease candidate donanemab, and is planning to take on Biogen […]

Eli Lilly made a late entry into the checkpoint inhibitor market when it licensed ex-China rights to Innovent’s sintilimab last year, and a new trial […]

Bristol Myers Squibb’s Zeposia (ozanimod) Receives CHMP’s Positive Opinion for Adult Patients with Moderately to Severely Active Ulcerative Colitis Published: 15 Oct, 2021 | Tags: […]

BMS Reports EMA’s Validation of MAA for Relatlimab and Nivolumab as 1L Treatment for Unresectable or Metastatic Melanoma Published: Oct 1, 2021 | Tags: BMS, […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

Shots: The P-III SURPASS-3 MRI sub-study evaluates the effects of tirzepatide (5/10/15mg, qw) vs titrated insulin degludec on LFC, VAT and ASAT in 296 patients […]

Any lingering hope that Pfizer’s may finally get a regulatory approval for its nerve growth factor (NGF) inhibitor tanezumab looks to have been dashed after […]

Shots: Twist to receive an up front for each program and $710M in clinical, regulatory, and commercial milestones for the multiple target discovery programs Boehringer […]

Shots: The designation is based on the P-III EMPEROR-Preserved trial evaluates the safety and efficacy of empagliflozin (10mg, qd) vs PBO in 5,988 patients with […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

Forte’s FB-401 Fails to Meet Primary Endpoints in P-II Clinical Trial For the Treatment of Atopic Dermatitis Published: Sept 3, 2021 | Tags: Forte, FB-401, […]

The FDA has concluded its safety review of Pfizer’s JAK inhibitor Xeljanz and Xeljanz XR, requiring revised warnings for the drugs as well as others […]

Shots: The P-III EMPEROR-Preserved trial evaluates the safety & efficacy of empagliflozin (10mg, qd) vs PBO in 5988 patients with chronic HFpEF The results demonstrated […]

Eli Lilly is entering the field of protein-degrading drugs through a partnership with Lycia Therapeutics, a startup whose technology goes further than the first wave […]

Eli Lilly has bolstered its position the fast-emerging area of protein degrader drugs, agreeing a $1.6 billion-plus deal with Lycia Therapeutics spanning up to five […]

Janssen Presents Results of Rybrevant (amivantamab-vmjw) in P-I CHRYSALIS Study for Advanced NSCLC with METex14 Mutations at WCLC Published: Aug 20, 2021 | Tags: Astellas, […]

Shots: The approval is based on the P-III EMPEROR-Reduced trial evaluating the effect of Jardiance (10 mg, qd) vs PBO in 3730 adults with HFrEF […]

Eli Lilly and Boehringer Ingelheim have claimed FDA approval for their diabetes therapy Jardiance in heart failure with reduced ejection fraction (HFreF), setting up a […]

Eli Lilly is one of the front runners in the bid to follow Biogen and Eisai onto the market with an anti-amyloid drug for Alzheimer’s […]

Shots: The approval is based on P-III PRONTO-PUMP-2, a treat-to-target study that evaluates the efficacy & safety of Lyumjev (insulin lispro-aabc injection, 100 units/mL) vs […]

Eli Lilly had a lot riding on its two phase 3 trials of IL-13 inhibitor lebrikizumab in atopic dermatitis (AD) – and it now looks […]

Junshi and Coherus’s Toripalimab Receive the US FDA’s Breakthrough Therapy Designation as 1L Treatment of Nasopharyngeal Carcinoma Published: Aug 13, 2021 | Tags: Junshi, Coherus, […]

Immunology deals with physiological functioning of the immune system in states of both health and disease as well as malfunctions of the immune system in […]

After rejecting Eli Lilly’s Verzenio for breast cancer therapy earlier this year, NICE has relented, recommending the drug for routine NHS use alongside hormonal therapy […]

AstraZeneca’s Forxiga has become the first SGLT2 inhibitor to be approved in Europe for use in people with chronic kidney disease, extending its lead over […]

Shots: The P-III ORIENT-11 study evaluates the efficacy & safety of sintilimab (200mg) + pemetrexed and platinum CT vs PBO + CT in 397 patients […]

Eli Lilly and Incyte have another rival in the rear-view mirror for Olumiant in alopecia areata after Pfizer reported that its ritlecitinib improved scalp hair […]

The FDA’s approval of Biogen’s Aduhelm for Alzheimer’s disease emboldened Eli Lilly to move forward plans to file its own candidate donanemab later this year, […]

In an interview with PharmaShots, Dr. Lotus Mallbris, M.D., Ph.D., Vice President of Immunology Development at Lilly shared her views on the abstracts demonstrating data on […]

Eli Lilly is beginning a research alliance discovering and developing small molecules that stimulate immune responses against cancer. If drugs from the partnership reach the […]

Eli Lilly Signs an Agreement with Banner Alzheimer’s Institute to Evaluate Donanemab in P-III TRAILBLAZER-ALZ 3 Trial for Alzheimer’s Disease Published: July 16, 2021 | […]

Shots: Eli Lilly to acquire Protomer Technologies for ~$1B, with the achievement of future development and commercial milestones Earlier, Lilly led an equity investment in […]

Eli Lilly has Protomer Technologies, a preclinical startup developing next-generation insulin that senses blood glucose levels and adjusts its activity accordingly. The technology could eliminate […]

Eli Lilly bought a stake in Protomer Technologies and its glucose-sensing insulin platform last year, and it must like what it has seen in the […]

Eli Lilly is partnering with startup Verge Genomics in a move intended to add ALS drugs to its neuroscience pipeline. To date, Verge’s artificial intelligence […]

Boehringer Ingelheim and Eli Lilly have the results they were hoping for in a large-scale study of Jardiance therapy in heart failure with preserved ejection […]

Two months after Eli Lilly began an alliance with MiNA Therapeutics, a startup developing a new type of RNA therapy, the pharmaceutical giant is making […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

A north-south divide has emerged in UK medicines access after the Scottish Medicines Consortium (SMC) okayed Kyowa Kirin’s Poteligeo for two rare blood cancers, a […]

Novo Nordisk is celebrating the FDA approval of its GLP-1 agonist semaglutide as a treatment for obesity, which it believes has the potential to redefine […]

Novo Nordisk is celebrating the FDA approval of its GLP-1 agonist semaglutide as a treatment for obesity, which it believes has the potential to redefine […]

Seelos’ SLS-005 (Trehalose) Receives EC’s Orphan Drug Designation to Treat Amyotrophic Lateral Sclerosis Published: May 28, 2021 | Tags: Seelos, SLS-005, Trehalose, EC, Orphan Drug […]

Concert Pharmaceuticals has started the second of two planned phase 3 trials of its drug candidate for alopecia areata (AA), a common cause of hair […]

Shots: Eli Lilly and Dicerna have reported the US FDA’s IND acceptance of LY3819469 to treat cardiometabolic diseases. The IND acceptance triggers $10M as a […]

BeiGene Reports Results of Tislelizumab + CT in P-III RATIONALE 309 Study as 1L Treatment for Recurrent or Metastatic Nasopharyngeal Cancer Published: May 21, 2021 […]

Eli Lilly has scored five out of five with its phase 3 trials of type 2 diabetes candidate tirzepatide, setting up regulatory filings for a […]

Eli Lilly diabetes drug tirzepatide beat long-acting insulin in the last of a series of global studies designed to support regulatory submissions for the type […]

The FDA has accepted Eli Lilly and China’s Innovent filing for their latecomer immunotherapy sintilimab, which is expected to compete on price with rivals such […]

Shots: The submission is based on a P-III ORIENT-11 study assessing sintilimab (200mg) + pemetrexed and platinum CT vs PBO + CT as a 1L […]

UK biotech Arecor Therapeutics has announced plans to float on the London stock market, to fund plans to develop novel formulations of insulins and other […]

UK biotech MiNA Therapeutics has signed up another big pharma partner for its small activating RNA (saRNA) platform, which upregulates the activity of proteins, with […]

Shots: Eli Lilly collaborates with DexCom, Glooko, myDiabby Healthcare and Roche to integrate the data and technology into connected insulin pen solutions and provide streamlined […]

What We Should Know: – Glooko Inc., a provider of remote patient monitoring and data management solutions for diabetes and other chronic conditions announced a […]

AstraZeneca is already seeing sales rocket for its SGLT2 inhibitor Farxiga in heart failure, and is expected to gather even more momentum after claiming FDA […]

Shots: The new analyses of the P-III BREEZE-AD5 trial and an extended safety analysis across multiple trials for Olumiant (2mg, qd) vs PBO in patients […]

Eli Lilly has walked away from a major indication for its blockbuster hopeful mirikizumab, shelving plans to file it for psoriasis as the market is […]

ThermoFisher to Acquire PPD for ~$17.4B Published: Apr 16, 2021 | Tags: ThermoFisher, Acquire, PPD, ~$17.4B Roche’s Evrysdi (risdiplam) Receives Health Canada’s Approval for Spinal […]

Shots: Lilly to receive up front, milestones, as well as royalties on the products developed and commercialize by SciNeuro. Lilly retains all rights outside Greater […]

Eli Lilly’s JAK inhibitor Olumiant has missed its main objective in a phase 3 trial in hospitalised COVID-19 patients, but had a significant effect on […]

In an interview with PharmaShots, Marie Schiller Vice President of Product Development for Connected Care and Insulins at Lilly shared her views on its collaboration with Welldoc […]

Pfizer and BioNTech Report Results of BNT162b2 to Prevent COVID-19 Published: Apr 2, 2021 | Tags: Pfizer and BioNTech, Report, Results, BNT162b2, Prevent, COVID-19 Acceleron […]

Shots: The P-II BLAZE-4 study involves assessing bamlanivimab as monothx. and bamlanivimab (700mg) + VIR-7831 (500mg) vs PBO in 1000 participants with symptomatic low-risk COVID-19 […]

Two antibodies targeting different parts of the COVID-19 virus spike protein, from Eli Lilly and GlaxoSmithKline/Vir Biotech, can cut viral load dramatically within a week. […]

Regeneron has posted findings from a large trial its COVID-19 antibody cocktail showing it reduced risk of hospitalisation or death by 70%. The US pharma […]

After a long and troubled development the fate of Pfizer/Lilly’s tanezumab painkiller injection is in the balance ahead of a key meeting of advisers later […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Takeda Collaborates with Anima to Discover and Develop mRNA Translation Modulators for Neurological Diseases Published: Mar 19, 2021 | Tags: Takeda, Collaborates, Anima, Discover, Develop, […]

Shots: The P-II TRAILBLAZER-ALZ study involves assessing donanemab vs PBO in 272 patients with early symptomatic AD who were selected based on cognitive assessments in […]

The UK regulator has authorised Eli Lilly’s targeted cancer drug Retsevmo for tumours with RET fusion positive lung cancers and thyroid cancers, which the company […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Eli Lilly reported data from a second group in a Phase 3 study testing its combination antibody drug for Covid-19 showing that the treatment reduced […]

With so many novel drug candidates for Alzheimer’s disease failing in clinical development, researchers in the US have started using artificial intelligence (AI) to screen […]

Last year, Belgium’s ExeVir Bio raised 23 million euros ($27 million) to finance development of llama antibodies – and now it is ready to begin […]

The EMA’s human medicines committee has said two Eli Lilly antibodies are effective in people with COVID-19 who are at risk of developing severe disease, […]

Eli Lilly and Incyte Report Results of Olumiant (baricitinib) in P-III BRAVE-AA2 Study for Severe Alopecia Areata Published: Mar 5, 2021 | Tags: Eli Lilly […]

Eli Lilly’s JAK inhibitor Olumiant has become the first drug in the JAK inhibitor class to be recommended for NHS use in England and Wales […]

Eli Lilly has become the latest drugmaker to have a go at developing RIP kinase inhibitors, a new class of drugs with potential in autoimmune […]

Eli Lilly is paying $125 million up front to team up with Rigel Pharmaceuticals on drugs that block a signaling protein involved in inflammation. With […]

“Patent losses could sink Allergan”.1 “Drug prices to plummet in wave of expiring patents”.2 “Price decay after loss of brand exclusivity”.3 These are just three […]

Visby’s POC COVID-19 Test Receives the US FDA’s EUA for Use in CLIA Waived Settings Published: Feb 12, 2020 | Tags: Visby, POC, COVID-19, Test, Receives, […]

The biopharma industry saw numerous deal terminations in 2020. Clinical and regulatory results, change in control limitations, and strategic reprioritizations were among the most common […]

The EUA for Eli Lilly brings to patients a second antibody cocktail, following the FDA’s fall authorization of an antibody combination from Regeneron Pharmaceuticals. Lilly […]

The FDA has granted an emergency licence for Eli Lilly’s COVID-19 antibody combination therapy, to reduce chances of high-risk patients progressing from mild to moderate […]

Shots: The FDA authorized EUA for bamlanivimab (700 mg) and etesevimab (1400 mg) for the treatment of mild to mod. COVID-19 patients aged ≥12yrs. who […]

Roche Collaborate with Cambridge Quantum Computing to Develop Quantum Algorithm for Drug Discovery and Development Published: Jan 29, 2020 | Tags: Roche, Collaborate, Cambridge Quantum Computing, […]

US drugmaker Eli Lilly – still waiting for an FDA decision on one non-opioid pain drug – has just added another to its pipeline via […]

US drugmaker Eli Lilly – still waiting for an FDA decision on one non-opioid pain drug – has just added another to its pipeline via […]

Regeneron is considering an emergency filing for its antibody cocktail REGEN-COV to protect people against COVID-19 after early trial results showed it conferred 100% protection […]

Shots: The P-II/III BLAZE-1 trial involves assessing of Bamlanivimab + Etesevimab (2800 mg) vs PBO in 1035 patients recently diagnosed with COVID-19 in the ambulatory […]

Takeda’s Alunbrig (brigatinib) Receives MHLW’s Approval as a 1L and 2L Treatment for ALK+ Advanced or Recurrent NSCLC Published: Jan 22, 2020 | Tags: Takeda, […]

Life sciences companies are gearing up to enter new markets as they look to secure their positions after a spate of M&A, licensing, and research […]

Shots: The P-III BLAZE-2 COVID-19 prevention trial involves assessing bamlanivimab (4,200mg) vs PBO in residents and staff at skilled nursing and assisted living facilities. The […]

Always a big player in oncology, Eli Lilly has fallen behind some of its rivals when it comes to cancer immunotherapy, but a new R&D […]

South Korea’s Celltrion has announced encouraging top-line results for its potential antibody therapy for COVID-19, with the first part of a phase 2/3 trial showing […]

When Eli Lilly reported results for donanemab in Alzheimer’s disease earlier this week it was hailed as a rare win for the amyloid hypothesis, although […]

Boehringer and Eli Lilly have moved closer to a heart failure indication for their SGLT2 inhibitor Jardiance, as the FDA starts a fast-track review of […]

Biopharma merger and acquisition activity in 2020 was mainly filled with late-stage, bolt-on acquisitions, which were orders of magnitude smaller than the mega M&A deals […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

The lawsuit follows action by at least a half-dozen drug companies to rein in what they see as waste and abuse in the program, overseen […]

Eli Lilly has acquired Prevail, a biotech focusing on gene therapies for neurodegenerative diseases including Parkinson’s, in a deal potentially worth more than $1 billion. […]

Eli Lilly’s decision to throw large amounts of research dollars at its next-generation diabetes drug tirzepatide could be vindicated, according to its first phase 3 […]

Eli Lilly’s buyout of Loxo Oncology last year has already yielded one approved drug, and it now has a path to market for a second […]

Merck Collaborates with Siemens for the Digitalization of Production Published: Nov 26, 2020 | Tags: Agreement, Collaborative, Digitalization, Merck, Siemens, Sign CoSara Receives CDSCO’s Approval […]

Regeneron’s antibody cocktail is the latest COVID-19 drug to receive Emergency Use Authorization in the US, becoming the first therapy of this kind to become […]

Doctors in the US are to get another option to treat COVID-19 after the FDA granted an Emergency Use Authorization (EUA) for Eli Lilly’s baricitinib. […]

Shots: The EUA is based on ACCT-2 study assessing baricitinib (4mg, qd for 14 days or until hospital discharge) in combination with remdesivir vs PBO […]

UK cost-effectiveness agency NICE has said that Eli Lilly’s Emgality can be made available through the NHS for migraine prevention, the second drug in the […]

Shots: Seed Therapeutics to receive $10M upfront, $10M equity investment, ~$780M as pre/clinical development, regulatory & commercial milestones along with royalties on net sales of […]

Eli Lilly has said it will start shipping supplies of its COVID-19 drug bamlanivimab immediately, after claiming emergency-use authorisation (EUA) for the antibody. The AbCellera-partnered […]

Eli Lilly’s chief information and digital officer (CIDO) Aarti Shah is leaving the company next year after 27 years at the company. Shah is retiring […]

Regeneron’s coronavirus antibody cocktail therapy against COVID-19, famously used to treat president Donald Trump, has hit a safety issue after independent safety experts recommended it […]

Our planet is facing a major pandemic outbreak due to COVID-19 and health agencies are taking every measure to stop it. The COVID-19 virus has […]

The US government said it will buy 300,000 doses of Eli Lilly’s COVID-19 therapy bamlanivimab for $375 million if the drug gets Emergency Use Authorisation […]

Eli Lilly says no more patients will be treated with its COVID-19 therapy bamlanivimab in a trial run by the US National Institute of Allergy […]

Shots: The companies present the findings from a new exploratory sub-analysis of the P-III EMPEROR-Reduced study demonstrating that Jardiance (empagliflozin) reduced the risk of adverse […]

Immunology is an important branch of science which deals with the study of the immune system. The immune system is a highly regulated and balanced […]
Eli Lilly is to buy the private biotech Disarm Therapeutics, which is working on a new class of disease-modifying drugs for neurological diseases, in a […]

Shots: The independent DSMB recommended pausing the enrollment of the ACTIV-3 clinical trial. The trial is evaluating Lilly’s bamlanivimab (LY-CoV555, 7000mg) + remdesivir as a […]
Eli Lilly’s Olumiant (baricitinib) has a significant effect on recovery from COVID-19 when combined with Gilead Veklury (remdesivir), according to a large trial backed by […]

Eli Lilly has asked the FDA for an Emergency Use Authorization for its COVID-19 antibody therapy, after announcing new efficacy data from an ongoing trial. […]

Shots: Eli Lilly reports additional data on its SARS-CoV-2 neutralizing Ab programs including interim data on combination therapy in diagnosed patients with mild-to-mod. COVID-19 and […]

Eli Lilly has doubled down on its partnership with Chia’s Innovent with a licensing deal for ex-China rights to Tyvyt, their PD-1 inhibitor, which some […]

1. Roche’s Evrysdi (risdiplam) Receives the US FDA’s Approval for SMA in Adults and Children Published: Aug 10, 2020 | Tags: Roche, Evrysdi, risdiplam, Receives, US, […]

Shots: The NMPA has accepted the sNDA for Tyvyt (sintilimab) in combination with Gemzar (gemcitabine) and platinum as 1L therapy in sq. NSCLC The sNDA […]

US biotech Pieris Pharmaceuticals has begun a collaboration and supply agreement with Eli Lilly, trialling its cancer drug PRS-343 in combination with the big pharma’s […]

Denmark’s Novo Nordisk has beaten profit forecasts in the second quarter after overcoming tough conditions in the diabetes market by improving its productivity. Novo said […]

Shots: Eli Lilly initiates P-III BLAZE-2 study assessing LY-CoV555 to prevent SARS-CoV-2 infection and COVID-19 in residents and staff at long-term care facilities in the […]

While the world waits an effective coronavirus vaccine, Eli Lilly has started late-stage human testing an antibody drug as an alternative way to prevent viral […]
The FDA’s approval of AstraZeneca’s SGLT2 inhibitor Farxiga in heart failure was a first for the class, but Boehringer Ingelheim and Eli Lilly are closing […]

Shots: The P-III EMPEROR Trial involves assessing of Jardiance (10 mg) vs PBO in two studies EMPEROR-Reduced [NCT03057977, N=3,730] & EMPEROR-Preserved [NCT03057951, N=5,990] in patients […]

Shots: The study involved 1402 patients across 3 cohorts: a) 81 patients in Arizona (34 with AD and 47 without), b) 699 in Sweden (301 […]

Shots: Chi-Med to receive 70%-80% of Elunate sales in the form of royalties, manufacturing costs and service payments with no upfront regarding this amendment. Lilly […]
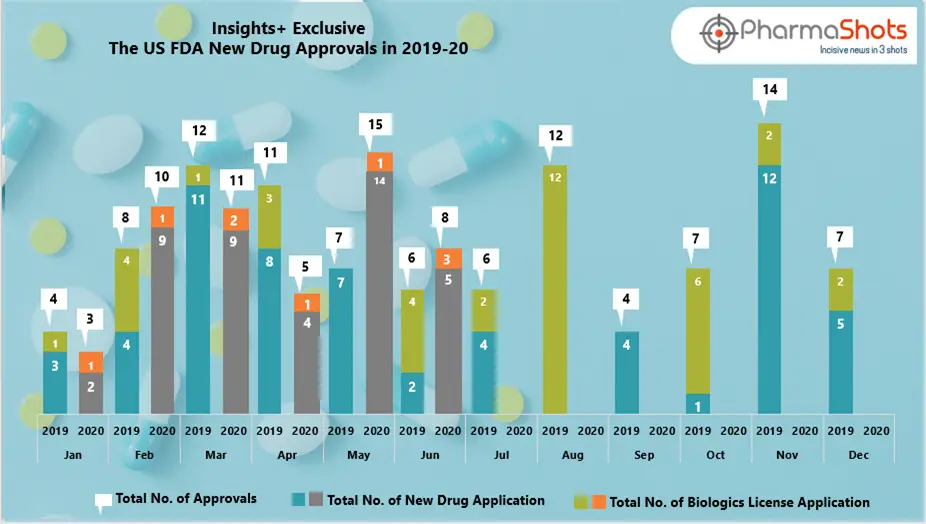
The US FDA has approved multiple NDAs and BLAs in Jun 2020, leading to treatments for patients and advances in the health care industry. The […]

Eli Lilly is a latecomer in the new generation of biologic therapies for psoriasis with its mirikizumab candidate, but a win in a head-to-head trial […]

Shots: The P-III OASIS-2 study involves assessing mirikizumab vs PBO & Cosentyx (secukinumab) in 1,465 patients with mod. to sev. PsO. The patients were randomized […]

Shots: The second P-III LIBRETTO-531 involves assessing of selpercatinib vs physician’s choice of cabozantinib or vandetanib in 400 patients in ratio (2:1) with advanced or […]
Copyright © 2024 | WordPress Theme by MH Themes