
More spending has lead to better health outcomes
That is according to a paper by Weaver et al. (2022). The authors use expenditure data from the Disease Expenditure Project and DALYs from the […]

That is according to a paper by Weaver et al. (2022). The authors use expenditure data from the Disease Expenditure Project and DALYs from the […]

Last month, the Office of Health Economics published a report titled “Health Technology Assessment of Gene Therapies: Are Our Methods Fit for Purpose?” I summarize […]

How do you measure changes in quality of life for patients with cancer? Today we will review 3 different approaches: health state based, health state […]

How does ICER plan to determine whether health plans offer fair access to pharmaceuticals? I summarized their approach last year as outlined in their white […]

Last month, ICER updated their protocol for identifying drugs that have unsupported price increases (UPI). Not only is ICER examining rising drug prices, but California […]

What considerations beyond quality adjusted life years and cost should be considered when evaluating gene therapies? A panel from ISPOR provided just such a discussion. […]

The value of medical devices caries with it less certainty than pharmaceuticals for a variety of reasons. As described in Kovács et al. (2022) medical […]

If you were a drug manufacturer, you naturally would want to focus your research on more common, highly prevalent diseases. If there are more patients […]

That is the name of a recent paper in Value in Health from three of pharmacoeconomics field’s leading scholars: Peter Neumann, Lou Garrison and Richard […]

In the United Kingdom, the National Institute for Health and Care Excellence (NICE) made an update to their health technology evaluations manual in January 2022. […]

Standard quality approach compares the difference in QALYs which is a function of survival and quality of life during each survival period. Consider the comparison […]

That is a title of a new white paper from FTI Consulting and Alexion. The key findings from the article are: HTA practices are designed […]

Check out an event this Friday, December 10, at 10am EST/7am PST on at The Hill on Rare Diseases: A Conversation on Value Assessments. A […]

Genomics England, the Department of Health and Social Care’s genome-sequencing hub, has this year announced that support for whole genome sequencing (WGS) has reached a […]

After the “stress test” of COVID, how can the sector create resilient, sustainable healthcare systems for the long term? COVID has taught us that future […]

A paper by Rand and Kesselheim (2021) in Health Affairs this month conducts a systematic literature review to answer this question. Based on 113 articles […]

The tectonic plates of healthcare technology assessment (HTA) have shifted – but where does the patient voice fit within this new evaluation paradigm? Recent advances […]

That is the question that a recent study by Tisdale et al. (2021) aim to answer. They first look at the top 250 Medicare Part […]

Last fall, ICER published a white paper titled “Cornerstones of ‘Fair’ Drug Coverage: Appropriate Cost-Sharing and Utilization Management Policies for Pharmaceuticals.” To paraphrase, ICER’s goal […]

In an article in Health Affairs, Rick van der Vegte and I argue that in four specific cases, the answer is yes. …the International Society […]

On June 10, I will be presenting at ISPOR’s Summit on “Value Assessment: New Insights, Approaches, and Applications,” which will highlight evolving practices, new methods, […]

Market access issues don’t stop once a product has passed HTA – and this is especially true for disruptive therapies like ATMPs. Experts from Executive […]

Episode 35 of the pharmaphorum podcast hears from Michael Smyth about Galapagos UK and joining the company as medical director just as COVID-19 hit. With […]

It’s fair to say this is the most interesting time ever for market access. Not only has COVID forced companies, governments and healthcare systems to […]

That is the topic of a recent paper by Augustovski et al. (2021). The authors conduct a targeted literature review of previous diagnostics value frameworks […]

A new drug comes on the market. Is the health benefit worth the cost? What is it’s value? Value assessment frameworks are one way to […]

Standard cost-effectiveness measures the value of a treatment based on the cost for an additional unit of health where units of health are measured in […]

What is a rare disease? The definition varies across countries, but according to the European Medicines Agency (EMA) rare diseases are those with a prevalence […]

Standard cost-effectiveness analysis assumes that any gain in quality-adjusted life years (QALYs) should be valued equally. This does not sound unreasonable, but is it true […]

Cost effectiveness analysis aims to capture identify (and monetize) all of a treatments benefits, risk and costs and then determine if the treatment is worth […]

In the latest of a suite of features looking at the biggest markets in Europe, Leela Barham takes stock of what 2021 could bring for […]

Should the US have a formal, centralized health technology assessment (HTA) organization? In the United Kingdom, the National Institute for Health and Care Excellence (NICE) […]

The FDA approval process is slow. Even for COVID-19 vaccines, the process can be time consuming. However, regulators around the world do have some expedited […]

UK artificial intelligence startup Okra Technologies has launched a new software platform that it claims can take the guesswork out of the price that can […]

While many health technology assessment (HTA) organizations acknowledge that many new treatments reduce the burden placed on caregivers, the value that these new treatments provide […]
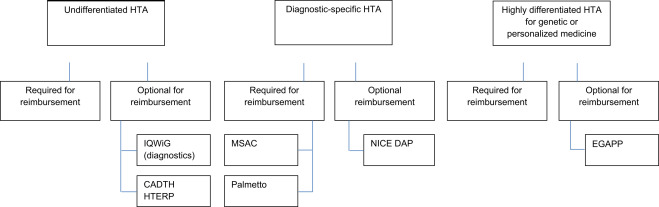
When evaluating a new diagnostic, HTA agencies must assess two separate issues: analytical and clinical validity. Analytical validity basically indicates whether the test works; is […]
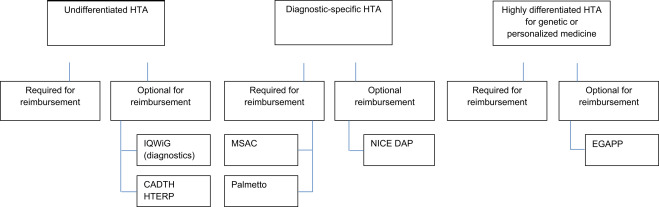
When evaluating a new diagnostic, HTA agencies must assess two separate issues: analytical and clinical validity. Analytical validity basically indicates whether the test works; is […]

Created by the Affordable Care Act (ACA) in 2010, the Patient-Centered Outcomes Research Institute (PCORI) has funded a wide variety of research comparing different treatments […]

Check out my interview on the Redeeming Value podcast with Brian Reid. PRECISIONheor Vice President Jason Shafrin pops in to talk about the Institute of […]
New research looks at the factors that speed up and slow down HTA appraisals for rare disease medicines across Europe. Rare diseases drugs have always […]
Copyright © 2024 | WordPress Theme by MH Themes