
Keytruda fluffs its lines in prostate, liver cancer trials
Two phase 3 trials of Merck & Co’s Keytruda in metastatic castration-resistant prostate cancer (CRPC) and advanced liver cancer have ended in failure, proving once […]

Two phase 3 trials of Merck & Co’s Keytruda in metastatic castration-resistant prostate cancer (CRPC) and advanced liver cancer have ended in failure, proving once […]

While immunotherapies have transformed treatment of many types of cancer, they do sometimes fail to have an impact – as Merck & Co has just […]

Novartis is still waiting for FDA approval of its PD-1/PD-L1 inhibitor latecomer tislelizumab in relapsed oesophageal cancer, but is already planning a move the drug […]

Just weeks after Bristol-Myers Squibb claimed an FDA green light for Opdivo as a neoadjuvant therapy for non-small cell lung cancer, AstraZeneca is looking to […]

While Merck & Co’s Keytruda is the undisputed leader of the non-small cell lung cancer (NSCLC) category, there’s one category where it lags behind Roche’s […]

The FDA has cleared two combination drug regimens based on Bristol-Myers Squibb’s PD-1 inhibitor Opdivo for previously-untreated advanced oesophageal cancer, encroaching on territory held by […]

Around 100 patients in England and Wales with triple negative breast cancer (TNBC) that has spread to other parts of the body could soon access […]

Merck & Co has quietly added another drug to its immuno-oncology pipeline via an agreement with China’s Sichuan Kelun Pharmaceutical, and is keeping the details […]

Orders for Merck & Co’s oral antiviral for COVID-19 helped to swell the drugmaker’s first quarter revenues by a massive 50%, although underlying growth was […]

AstraZeneca’s hopes of developing its PD-L1 inhibitor Imfinzi for cervical cancer have been hit by a failed phase 3 trial of the drug alongside chemotherapy […]

Merck & Co’s KEYNOTE-158 trial of Keytruda has underpinned another FDA approval, this time for the PD-1 inhibitor as a single-agent therapy as a therapy […]

Merck & Co’s Keytruda is poised to move even earlier in the treatment of patients with non-small cell lung cancer (NSCLC) after showing efficacy when […]

Merck & Co’s two powerhouse oncology drugs – Keytruda and Lynparza – have failed to show efficacy when used in combination for patients with previously […]

There isn’t enough evidence to support the use of Merck & Co/MSD’s Keytruda in combination with chemotherapy as a first-line treatment for advanced triple negative […]

Bristol-Myers Squibb’s Opdivo has been playing second fiddle to Merck & Co’s Keytruda in the non-small cell lung cancer (NSCLC) market for years, but now […]

Merck & Co has passed another milestone in its drive to move PD-1 inhibitor Keytruda earlier in the treatment pathway for cancer, with the European […]

Merck & Co’s Keytruda is the undisputed market leader in immunotherapy for non-small cell lung cancer (NSCLC), but was leapfrogged by Roche’s Tecentriq in the […]

Merck & Co’s strategy of pushing Keytruda earlier in the treatment pathway for cancers has secured anther victory, after the FDA approved the immunotherapy for […]

Shots: The approval is based on the P-III KEYNOTE-564 trial evaluating Keytruda (200mg, IV, q3w for 1yrs.) vs PBO in a ratio (1:1) in 994 […]

Merck & Co has chalked up another win in drive to move PD-1 inhibitor Keytruda earlier in the treatment pathway for cancer, after the FDA […]

Shots: The new data from KEYNOTE-006/587/716 studies evaluate Keytruda in patients with melanoma. Patients were eligible for transition to KEYNOTE-587 for extended follow-up after KEYNOTE-006 […]

Take two recently-approved immunotherapies for triple negative breast cancer (TNBC), put them together, and what do you get? Hopefully a new standard of care for […]

The dominance of Merck & Co’s Keytruda in the treatment of previously-untreated non-small cell lung cancer (NSCLC) means it will be hard to dislodge – […]

Shots: The companies collaborated to evaluate the Evaxion’s EVX-01 + MSD’s Keytruda in P-IIb study in patients with metastatic melanoma stage III & IV. The […]

Shots: The companies collaborated to evaluate datopotamab deruxtecan + Keytruda vs Keytruda alone in P-III TROPION-Lung08 trial in ~740 patients with PD-L1 high advanced or […]

Merck & Co has claimed EU approval for Keytruda in triple-negative breast cancer (TNBC), a particularly hard-to-treat form of the disease. The European Commission has […]

Merck & Co’s Keytruda become the first cancer immunotherapy to be approved as a first-line oesophageal cancer treatment in the EU earlier this year, but […]

Roche has claimed an earlier-than-expected FDA approval for its checkpoint inhibitor Tecentriq in non-small cell lung cancer (NSCLC), giving the drug a lead in early-stage […]

Shots: The US FDA has approved Keytryda + CT, with/out bevacizumab, for the treatment of patients with persistent, recurrent or metastatic cervical cancer whose tumors […]

Merck & Co’s FDA approval for Keytruda in advanced liver cancer looks secure, after an Asian clinical trial found that the cancer immunotherapy improved survival […]

Shots: The P-III KEYNOTE-826 trial evaluates Merck’s Keytruda (200mg, IV, q3w for up to 35 cycles) + Pt-based CT vs PBO + CT with/out bevacizumab […]

There’s no shortage of cancer immunotherapies trying to knock Merck & Co’s Keytruda off the top of the tree in previously-untreated non-small cell lung cancer […]

MSD’s Keytruda has become the first immunotherapy cleared by NICE for previously-untreated patients with advanced oesophageal cancer, making it an option for routine NHS treatment. […]

Shots: The companies collaborated to evaluate Hookipa’s HB-200 (arenaviral immunotherapeutic) + Merck’s Keytruda (anti-PD-1 therapy) in a P-II trial for patients with advanced HNSCC, based […]

Shots: The companies collaborated to evaluate IO’s IO102-IO103 + MSD’s Keytruda vs Keytruda alone in the P-III trial for patients with previously untreated unresectable or […]

Bristol-Myers Squibb has scored a win in its drive to get Opdivo into earlier lines of cancer therapy, getting FDA approval for the drug for […]

Shots: The companies collaborated to evaluate ADG106 + Keytruda in dose-escalation and expansion clinical study to treat advanced or metastatic solid tumors & hematological malignancies […]

The FDA has approved GlaxoSmithKline’s latecomer PD-1 inhibitor Jemperli for a second use that will significantly extend the patient population eligible for treatment with the […]

FDA approval of a therapy that pairs drugs from Eisai and Merck allows the combination to become a first-line treatment for renal cell carcinoma. It’s […]

Shots: The approval is based on P-III CLEAR /KEYNOTE-581 trial that evaluates Keytruda (200 mg, IV, q3w) + Lenvima (20 mg, PO, qd) or Lenvima […]

Shots: The application is based on ORR data from cohorts D & K of KEYNOTE-158 trial evaluating Keytruda as monothx. (200 mg, q3w) in 90 […]

Merck & Co is looking at a decision from the FDA in December on its application to market Keytruda as a post-surgery treatment for people […]

Shots: The sBLA is based on P-III KEYNOTE-564 trial evaluating Keytruda monothx. (200mg, IV on day 1 of each 3wks. cycle for ~17 cycles) vs […]

Sanofi and Regeneron’s latecomer checkpoint inhibitor Libtayo has largely been protected from heavyweight competition in its first niche markets, but now looks set to for […]

Shots: The priority review is based on P-III KEYNOTE-716 trial assessing Keytruda (anti-PD-1 therapy) vs PBO in 954 patients aged ≥12yrs. with stage II resected […]

Roche is preparing for a December verdict form the FDA on adjuvant use of its PD-L1 inhibitor Tecentriq as an adjuvant treatment for some patients […]

Merck & Co’s Keytruda has just picked up its 30th approval from the FDA, and it is a good one – early-stage triple-negative breast cancer […]

Bristol-Myers Squibb has withdrawn its US marketing approval for PD-1 inhibitor Opdivo as a second-line, single-agent treatment for hepatocellular carcinoma, a common form of liver […]

Shots: The companies collaborated to evaluate OncoSec’s TAVO + Merck’s Keytruda vs SoC in a P-III KEYNOTE-C87 trial in ~400 patients with metastatic melanoma who […]

Shots: The approval is based on data from the second interim analysis of the P-II KEYNOTE-629 trial assessing Keytruda (200mg, IV, q3w) in 159 patients […]

Merck & Co will voluntarily withdraw a US marketing approval for cancer immunotherapy blockbuster Keytruda in gastric cancer, after studies intended to confirm its efficacy […]

Shots: Linnaeus will conduct 6 additional P-II cohorts evaluating LNS8801 + Keytruda in several cancer indications while the companies are currently evaluating the combination in […]

Shots: The P-III KEYNOTE-564 evaluating Keytruda monothx (200 mg, IV, on day1 of each 3wks. cycle for up to 17 cycles) vs PBO in 950 […]

Shots: The companies reported the positive results from two studies (EV-201 and EV-103) evaluating Padcev as monothx. and in combination with Merck’s Keytruda in patients […]

Shots: The 1st sNDA and sBLA is based on results from the P-III CLEAR Study evaluating Lenvima + Keytruda or in combination with everolimus vs […]

The FDA’s Oncologic Drugs Advisory Committee (ODAC) has voted to strip Merck & Co’s Keytruda of its accelerated approval in gastric and gastroesophageal junction (GEJ) […]

Ken Frazier’s final appearance on Merck & Co’s financial results call as chief executive was a muted affair, as the company reported lower-than-expected revenues dragged […]

Merck & Co has announced interim data from its pivotal Phase 3 KEYNOTE-564 trial evaluating immunotherapy Keytruda for the potential adjuvant treatment of patients with […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Merck & Co has voluntarily withdrawn its Keytruda immunotherapy from a lung cancer niche in the US, amid a crackdown by the FDA on drugs […]

New phase 3 data have shored up the position of Merck & Co’s cancer immunotherapy Keytruda in the increasingly competitive first-line kidney cancer market. The […]

NICE has backed regular NHS funding England and Wales for Merck, Sharp and Dohme’s Keytruda in combination with pemetrexed and platinum chemotherapy in certain lung […]

A former Merck & Co scientist could face up to 10 years in prison after he was accused of stealing trade secrets relating to drugs […]

The deal will broaden Merck’s oncology portfolio as it picks up an antibody-drug conjugate, a class of biological drugs that target tumor cells even as […]

An experimental gene therapy developed by Texas biotech Genprex will be paired with AstraZeneca’s Tagrisso and Merck & Co’s Keytruda – both leading their respective […]

Shots: Sanofi will sponsor the clinical trials while MSD will provide KEYTRUDA. Additionally, Sanofi is separately evaluating the activity of THOR-707 in combination with other […]

The FDA has started a priority review of Regeneron and Sanofi’s checkpoint inhibitor Libtayo in first-line non-small cell lung cancer (NSCLC), based on data they […]

As recently as June, NICE was minded not to back routine NHS of MSD’s Keytruda as a first-line treatment for advanced head and neck cancer, […]

Shots: The approval is based on P-III KEYNOTE-204 study assessing Keytruda (200mg, IV, q3w vs BV (1.8 mg/kg, IV, q3w) in 304 patients in a […]

Shots: The first approval is for certain ESCC patients, which is based on P-III KEYNOTE-181 trial assessing Keytruda vs CT in patients with recurrent or […]
Bristol-Myers Squibb’s Opdivo has improved survival in a trial involving patients with mesothelioma, a form of lung cancer, getting one up over rival checkpoint inhibitor […]
Roche’s hopes of extending the use of its PD-L1 inhibitor Tecentriq in triple-negative breast cancer (TNBC) have been dashed by a late-stage trial failure looking […]
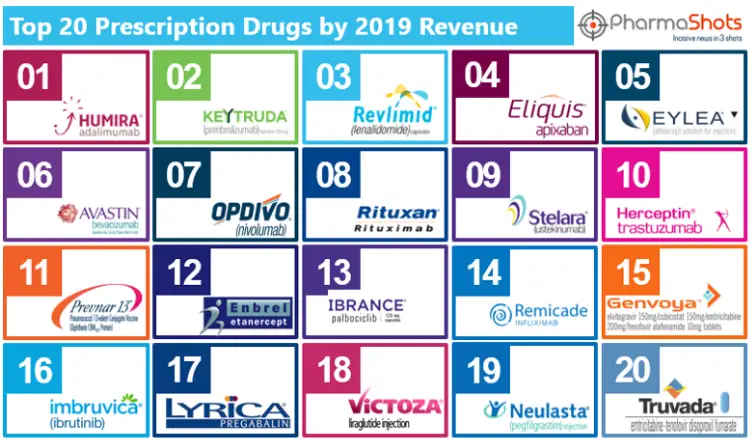
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
Shots: The companies plan to conduct a P-II pilot study of tumor treating fields concomitant with Keytryda for 1L treatment of intrathoracic advanced/ metastatic PD-L1+ […]
Copyright © 2024 | WordPress Theme by MH Themes