
Amylyx Submits NDA to the US FDA for AMX0035 to Treat Amyotrophic Lateral Sclerosis
Shots: The NDA is based on CENTAUR trial evaluates AMX0035 vs PBO in 137 patients with ALS The results showed that patients receiving AMX0035 had […]

Shots: The NDA is based on CENTAUR trial evaluates AMX0035 vs PBO in 137 patients with ALS The results showed that patients receiving AMX0035 had […]

Shots: The submission is based on P-II studies i.e., CITADEL-203/204/205 that evaluate parsaclisib in patients with r/r FL, MZL & MCL. The anticipated PDUFA date […]

Shots: The NDA & MAA submissions are based on ongoing 2 studies i.e., ACTIVATE & ACTIVATE-T evaluates safety, tolerability & efficacy of mitapivat vs PBO […]

Shots: The withdrawal follows the recent discussion with the US FDA regarding the palovarotene’s NDA review that initiated in May’21 The additional analysis will be […]

Shots: The NDA is based on P-III PSOARING 1 & 2 along with interim results from PSOARING 3 OLE study evaluating the safety & efficacy […]

Shots: The NDA is based on two P-III studies of surufatinib in patients with pancreatic and extra-pancreatic NET in China. The anticipated PDUFA date is […]

Shots: The NDA is based on data from the BREEZE study evaluating Tyvaso DPI in 51 patients with PAH transitioning from Tyvaso inhalation solution which […]

Shots: The submission is based on data from the P-III PSOARING 1 & PSOARING as well as interim results from PSOARING 3 long-term safety study […]

Shots: The NDA submission is based on data from the ongoing P-III MOVE trial evaluating the efficacy and safety of palovarotene (chronic/flare-up dosing regimen) in […]

Shots: The NDA submission is based on P-III studies (SERENITY I & II) assessing BXCL501 (120 & 180mcg doses) vs PBO for the acute treatment […]

Shots: The submission is based on P-IIb/III SELECTION study evaluating the efficacy and safety of filgotinib (200mg, qd) vs PBO in patients with mod. to […]

Shots: The US FDA has accepted IND to assess the safety, tolerability, PK, and preliminary antitumor activity of XB002 in patients with advanced solid tumors. […]

Shots: The NDA submission is based on data from P-III PROTECTIVE-2 study evaluating Plinabulin + Neulasta vs Neulasta alone for the prevention of CIN The […]

Shots: The NDA is based on P-II study-004 trial involves assessing Belzutifan (120 mg, PO, qd) in 61 patients with VHL disease who had at […]

Shots: The submission is based on P-III HELP study evaluating lanadelumab (300mg, q2w & q4w) vs PBO in 125 patients with HAE attack for 26wks. […]

Shots: The submission is based on P-III HELP study evaluating lanadelumab (300mg, q2w & q4w) vs PBO in 125 patients with HAE attack for 26wks. […]

Shots: The submission is based on EV-301 and EV-201 trials in Japan. The P-III EV-301 trial assessing Enfortumab Vedotin vs CT in ~ 600 patients […]

Shots: The NDA submission is based on two P-III studies (COUGH-1 & -2) involves assessing gefapixant (45/15mg, bid) vs PBO in 2,044 patients with RCC […]

Shots: The NDA is based on two P-III TRuE-AD 1 & 2 assessing ruxolitinib cream (0.75%/1.5%, bid) vs vehicle (non-medicated cream) in 600 patients aged […]

The US FDA has approved 4 NDAs in Jan 2021, leading to treatments for patients and advances in the health care industry. The Center for […]

Shots: The NDA is based on STORM and SADAL studies. The P-IIb STORM study involves assessing of ATG-010 + low-dose dexamethasone in patients with rrMM […]

Shots: Chi-Med initiated the filing of NDA to the US FDA for surufatinib to treat pancreatic & non-pancreatic NET & plans to complete the NDA […]

Shots: The NDA submission is based P-II CodeBreaK 100 study assessing Sotorasib in patients with KRAS G12C-mutated NSCLC prior treated with CT and immunotherapy The […]

Shots: The US FDA has accepted the NDA and granted PR for TAK-721 for the treatment of EoE. If approved, TAK-721 will be the first […]

Shots: The acceptance marks the first marketing application accepted for achondroplasia in the US with an anticipated PDUFA date as of Aug 20, 2021. The […]

Shots: The NDA submission is based on FIREFISH study assessing Risdiplam in infants with symptomatic SMA Type 1 & SUNFISH study in children and young […]

Shots: The NDA submission is based on P-III UPTRAVI IV study he safety and tolerability of 20 patients with PAH temporarily switching from oral UPTRAVI […]

Shots: The NDA submission was submitted to NMPA’s CDE and is based on P-III VICTORIA study, seeking regulatory approval of vericiguat in China In Oct’2014, […]

Shots: BioMarin has submitted an NDA to the US FDA for vosoritide to treat children with achondroplasia The company also reported that EMA has begun […]

Shots: The NDA submission is based on three clinical studies in which Trilaciclib was administered prior to chemotherapy treatment in patients with SCLC and has […]
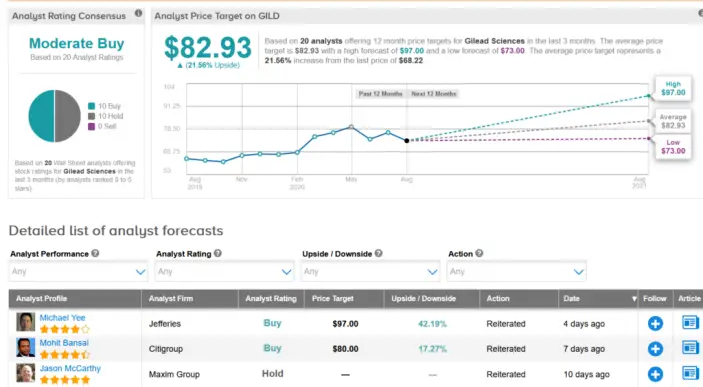
Shots: The NDA filing is based on P-III studies evaluating Veklury vs PBO, conducted by NIAID. The studies demonstrated that Veklury led to faster time […]
Copyright © 2024 | WordPress Theme by MH Themes