
BMS’ plan to move Opdivo into adjuvant RCC gets knocked back
Bristol-Myers Squibb has revealed that its checkpoint inhibitor combination of Opdivo and Yervoy failed a phase 3 trial as adjuvant (post-surgery) therapy for renal cell […]

Bristol-Myers Squibb has revealed that its checkpoint inhibitor combination of Opdivo and Yervoy failed a phase 3 trial as adjuvant (post-surgery) therapy for renal cell […]

NICE has recommended routine use of Bristol-Myers Squibb’s Opdivo for the adjuvant treatment of some patients with urothelial carcinoma (UC), the most common form of […]

Novartis is still waiting for FDA approval of its PD-1/PD-L1 inhibitor latecomer tislelizumab in relapsed oesophageal cancer, but is already planning a move the drug […]

Just weeks after Bristol-Myers Squibb claimed an FDA green light for Opdivo as a neoadjuvant therapy for non-small cell lung cancer, AstraZeneca is looking to […]

The FDA has cleared two combination drug regimens based on Bristol-Myers Squibb’s PD-1 inhibitor Opdivo for previously-untreated advanced oesophageal cancer, encroaching on territory held by […]

Bristol-Myers Squibb’s flurry of dealmaking has continued with en expansion of its partnership with BridgeBio on drugs targeting SHP2, in the hope of combining them […]

Bristol-Myers Squibb has won FDA approval for the first LAG-3 inhibitor drug ahead of its rivals in cancer immunotherapy including Merck & Co. The US […]

Bristol Myers Squibb bet more than $1.8 billion that Nektar Therapeutics’ drug could be combined with the pharma giant’s Opdivo to expand the reach of […]

Bristol-Myers Squibb’s Opdivo has been playing second fiddle to Merck & Co’s Keytruda in the non-small cell lung cancer (NSCLC) market for years, but now […]

Lung cancer drug Opdivo administered with chemotherapy has been shown to boost survival rates in patients with resectable non-small cell lung cancer (NSCLC). The phase […]

Merck & Co’s Keytruda become the first cancer immunotherapy to be approved as a first-line oesophageal cancer treatment in the EU earlier this year, but […]

Patients in Scotland with newly-diagnosed advanced kidney cancer can now access a therapy combining Bristol-Myers Squibb’s immunotherapy Opdivo with Ipsen’s targeted drug Cabometyx, ahead of […]

In an interview with PharmaShots, Dr. Ian Waxman, MD, Development Lead, Gastrointestinal Cancer at BMS shared his views on the EC’s approval of Opdivo (nivolumab) […]

Shots: The P-III CheckMate -214 trial evaluates Opdivo (3mg/kg) + Yervoy (1mg/kg, q3w for 4 doses) vs sunitinib in 1096 patients with previously untreated advanced/mRCC […]

Shots: The application is based on P-III CheckMate -648 study evaluating Opdivo (3mg/kg, q2w, IV) + Yervoy (1mg/kg, q6w up to 24mos.) or Opdivo + […]

Shots: The P-III CheckMate -743 trial evaluates Opdivo (3mg/kg, q2w) + Yervoy (1mg/kg, q6w) vs CT in 605 patients with previously untreated unresectable MPM. The […]

Bristol-Myers Squibb has scored a win in its drive to get Opdivo into earlier lines of cancer therapy, getting FDA approval for the drug for […]

The FDA has approved GlaxoSmithKline’s latecomer PD-1 inhibitor Jemperli for a second use that will significantly extend the patient population eligible for treatment with the […]

Shots: The applications are based on P-III CheckMate -648 trial evaluates Opdivo (3 mg/kg, q2w) + Yervoy (1 mg/kg, q6w up to 24mos.) or Opdivo […]

Bristol-Myers Squibb is in a battle with Merck & Co for position in the upper gastrointestinal cancer immunotherapy market, and has just reinforced its position […]

Shots: The companies will initiate P-I/II study evaluating the safety and preliminary efficacy of BBP-398 in combination with both Opdivo as dual therapy & Opdivo […]

Bristol Myers Squibb blockbuster drug Opdivo is being pulled from the market after failing to meet the main goal of a confirmatory study. The voluntary […]

Bristol-Myers Squibb has withdrawn its US marketing approval for PD-1 inhibitor Opdivo as a second-line, single-agent treatment for hepatocellular carcinoma, a common form of liver […]

Shots: The P-III CheckMate -648 trial evaluating Opdivo + CT or Opdivo (3 mg/kg, q2w) + Yervoy (1 mg/kg, q6w up to 24mos.) in patients […]

Shots: The approval is based on P-III CheckMate -743 study assessing Opdivo + Yervoy vs SOC Pt. based CT in patients with previously untreated unresectable […]

Shots: The opinion is based on P-II CheckMate -142 study assessing Opdivo + Yervoy in patients with mismatch repair deficient or microsatellite instability–high metastatic colorectal […]

Shots: The filing is based on results from the P-III CheckMate -274 trial evaluating Opdivo (240mg, q2w for up to 1 yrs.) vs PBO in […]

The FDA’s Oncologic Drugs Advisory Committee (ODAC) has voted to strip Merck & Co’s Keytruda of its accelerated approval in gastric and gastroesophageal junction (GEJ) […]

Shots: The CHMP has recommended the approval of Opdivo (nivolumab) + Yervoy (ipilimumab) as 1L treatment for unresectable MPM and is based P-III CheckMate -743 […]

Shots: The CHMP recommendation is based on P-III CheckMate -9ER trial involves assessing Opdivo + Cabometyx vs sunitinib in 651 patients with previously untreated advanced […]

New phase 3 data have shored up the position of Merck & Co’s cancer immunotherapy Keytruda in the increasingly competitive first-line kidney cancer market. The […]

The pairing of Heat Biologics’ lead drug HS-110 with Bristol-Myers Squibb’s blockbuster checkpoint inhibitor Opdivo has boosted survival in patients with non-small cell lung cancer […]

NICE has rejected Bristol Myers Squibb’s immunotherapy Opdivo as a second-line treatment for head and neck cancer in first draft guidance, unconvinced by evidence unearthed […]

Bristol Myers-Squibb has put out another downbeat “update” announcement about its immunotherapy Opdivo in an aggressive form of brain cancer, saying that it looks unlikely […]

Shots: The approval is based on P-lll ATTRACTION-3 study involve the assessment of Opdivo vs CT (docetaxel or paclitaxel) for patients with esophageal cancer refractory/ […]

Bristol-Myers Squibb could be mere months away from claiming a second US approval for an Opdivo-based combination immunotherapy that will help defend its first-line kidney […]
Shots: The P-III CheckMate-816 study involves assessing of Opdivo (360mg) + CT (q3w for 3 doses) vs CT (q3w for 3 doses), followed by surgery […]

Bristol-Myers Squibb’s immunotherapy combination of Opdivo and Yervoy has been approved in the US in a new indication for untreated mesothelioma, a rare but aggressive […]

Shots: The approval is based on P-III CheckMate -9LA study assessing Opdivo + Yervoy combined with two cycles of platinum-doublet CT vs CT (four cycles […]
Bristol-Myers Squibb’s Opdivo has improved survival in a trial involving patients with mesothelioma, a form of lung cancer, getting one up over rival checkpoint inhibitor […]
Roche’s checkpoint inhibitor Tecentriq has been cleared for another new use in the US – in melanoma – but could struggle to displace rival drugs […]
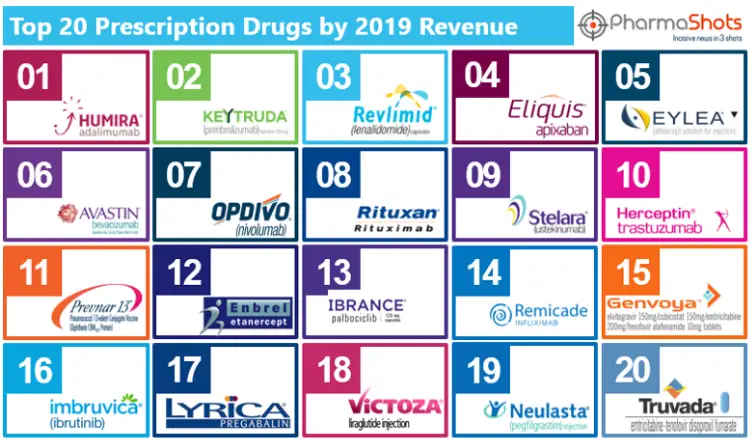
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
Copyright © 2024 | WordPress Theme by MH Themes