
UK life science competitiveness indicators 2022: measuring what matters most?
The latest life science competitiveness indicators (LSCIs) were published by the Office of Life Sciences (OLS) in July 2022. The indicators did not bring universal […]

The latest life science competitiveness indicators (LSCIs) were published by the Office of Life Sciences (OLS) in July 2022. The indicators did not bring universal […]

Walgreens helped fuel the opioid epidemic in San Francisco by dispensing hundreds of thousands of prescriptions without performing adequate due diligence, a federal judge ruled […]

Over the last few years, our industry has become familiar with CDISC standards. This has largely been driven by regulation, with national regulators such as FDA […]

A jury in Minnesota ruled on Friday that a pharmacist did not commit discrimination when he did not fill an emergency contraception prescription for a […]

Home care providers have always taken measures to ensure that they’re able to hold on to staff – especially amid labor shortages – through clauses […]

The public health emergency (PHE) was once considered an oasis of sorts, both for home health agencies and health systems looking to deliver hospital-level care […]

The Centers for Disease Control and Prevention (CDC) announced this week that it would be ending its monitoring program for COVID-19 cases on cruise ships. […]

Texas is suing the Biden administration over its recent guidance reminding hospitals and physicians that federal law requires them to provide abortions if there is […]

A federal judge has ruled that abortion services can resume in the state of Louisiana, blocking the state from enforcing its so-called trigger ban. Judge […]

The Department of Justice (DOJ) on Tuesday announced the creation of a new Reproductive Rights Task Force aimed at protecting abortion access and enforcing federal […]

FDA wants drugs to be more targeted to factors that impact patient’s lives in ways that they care about. To achieve this goal, last month […]

Abortion providers in Oklahoma filed a lawsuit challenging the state’s two abortion bans on Friday, just a week after the Supreme Court overturned the constitutional […]

SAE Media Group’s Inaugural Conference… Medical Devices and IVD 2022 14 – 15 November 2022 London, UK http://www.medicaldevices-ivd.com Navigating The MDR and IVDR and Best […]

A group of abortion clinics and abortion rights organizations filed a lawsuit in the Ohio Supreme Court on Wednesday seeking to block the state’s six-week […]

The U.S. Department of Health and Human Services (HHS) and the Centers for Medicare & Medicaid Services (CMS) are taking steps to improve oncology care […]

E-cigarette giant Juul argued in court documents filed on Tuesday that the Food and Drug Administration (FDA) had overlooked a mountain of data that it […]

Abortion rights activists have quickly filed a slew of lawsuits in states with trigger bans on abortions that went into effect following the Supreme Court’s […]

The Supreme Court on Monday sided with two doctors challenging their convictions on drug distribution charges for over-prescribing opioid medications in a decision that could […]

The Supreme Court’s extraordinary decision to eliminate the universal right to abortion is a watershed moment in the American experiment, ending half a century when […]

Abortion pills are likely to become the next major front of the fight over reproductive healthcare in the wake of the Supreme Court’s decision overturning […]

The Food and Drug Administration (FDA) is set to propose limits on the levels of nicotine tobacco companies can place in traditional cigarettes in an […]

The U.S. Centers for Medicare & Medicaid Services (CMS) released its FY 2023 home health proposed payment rule late Friday. It comes with a decrease […]

Despite being a significant regulatory change to the home health care industry, some providers aren’t so sure whether the Home Health Value-Based Purchasing (HHVBP) Model […]

June 7, 2022, Washington DC – Fishawack Health (FH) announces the acquisition of leading healthcare consulting and advisory firm Avalere Health (Avalere) from Inovalon, which […]

Face-to-face encounter documentation remains a pain point for home health providers. It makes sense why, too. The rules and regulations around it are often inconsistent, […]

The traditional brick-and-mortar hospital system is broken, often leaving patients in a condition that’s worse than when they first arrived. That’s according to a May […]

Home health care providers and Medicare-for-all advocates certainly agree on their general disliking of Medicare Advantage (MA). The one problem is that these advocates are […]

With the nationwide rollout of the Home Health Value-Based Purchasing (HHVBP) Model inching closer, home health agencies are feeling confident about their ability to adapt. […]

The Food and Drug Administration (FDA) on Monday announced a series of measures aimed at increasing the domestic U.S. supply of infant formula to counteract […]

Vice President Harris on Tuesday warned the rights of all Americans are at risk if the Supreme Court weakens the right to privacy by overturning […]

President Biden on Tuesday said that other rights, including same sex marriage and access to birth control, are in question if a leaked Supreme Court […]

Rep. Alexandria Ocasio-Cortez (D-N.Y.) on Monday warned progressives that the Supreme Court “isn’t just coming for abortion” after a leak of a Supreme Court draft […]

Planned Parenthood reacted with horror and dismay to the Monday publication of a draft Supreme Court ruling that would strike down Roe v. Wade, the […]

Aging in place is difficult for many Americans, but it’s even more difficult for those living in rural areas. Things are additionally hard on in-home […]

It would be silly to have an argument over whether value-based care is a good principle. That’s unless, of course, you had to parse out […]

When new drugs are approved, FDA limits their on-label use to the treatment of specific diseases. A key question is whether the definition of a […]

A federal judge on Thursday temporarily blocked a restrictive new abortion law in Kentucky, handing a win to abortion providers challenging the measure. In a […]

The U.S. Department of Health and Human Services (HHS) on Wednesday opted to renew the COVID-19 public health emergency (PHE), keeping in place several regulatory […]

A federal appeals court on Thursday ruled to uphold the Biden administration’s vaccine mandate for federal workers, ordering that a preliminary injunction issued against the […]

The National Academies of Sciences, Engineering and Medicine (NASEM) on Wednesday released its long-anticipated report on how to improve nursing home care in the U.S. […]

Home health agencies have had to navigate several changes since 2020, from the payment mechanics of the Patient-Driven Groupings Model (PDGM) to the new policies […]

Pushed by the White House, federal watchdogs have a new directive – cracking down on private equity in health care. While they’ll largely be targeting […]

Over the past few years, the U.S. health care system has learned that home-based care companies are well-positioned to respond to unique challenges, with at-home […]

This article is an exclusive feature available as a part of your subscription to HHCN + With the light at the end of the COVID-19 […]

US House Energy & Commerce Committee Chair Frank Pallone (D-NJ) has introduced a new bill to enhance the Food and Drug Administration’s (FDA) authority to […]

What disease components are most burdensome to patients? How do researchers find this information out? To answer this question, the Food and Drug Administration (FDA) […]

Policy concerns in home-based care are considered timely and critical for those in the industry. The issue is, unfortunately, they aren’t always viewed in that […]

The next year will be a big one for home care. There will be increased opportunities for agencies with a larger national spotlight shone on […]

The life sciences industry is more quickly adapting to new methods, standards and requirements. The focus is now on how to drive greater data governance […]

Former U.S. Centers for Medicare & Medicaid Services (CMS) Administrator Seema Verma is bullish on a lot. Speaking at the Home Care 100 conference Tuesday, […]

Four home care operators in Portland, Maine, were indicted on counts of wage fixing and labor market allocation charges, the U.S. Department of Justice (DOJ) […]

This article is an exclusive feature available as a part of your subscription to HHCN + Certificate of Need (CON) laws for home health agencies […]

Japan’s health ministry has authorised the use of Pfizer and BioNTech COVID-19 vaccine in children aged between five and 11. The vaccine, Comirnaty, is claimed […]

Scotland has approved AstraZeneca’s Tagrisso (osimertinib) as a monotherapy for the for the first-line treatment of adult patients with locally advanced or metastatic non-small cell […]

A World Health Organization (WHO) panel has given the green light to two new drugs to treat COVID-19. US-based Eli Lilly’s baricitinib (Olumiant), a Janus […]

Aveanna Healthcare Holdings (Nasdaq: AVAH) has been bullish on its plans to expand into a leading provider of senior home health services since it went […]

In 2021, the Food and Drug Administration’s Center for Drug Evaluation and Research (CDER) approved 50 new drugs, either as new molecular entities (NMEs) under […]

The U.S. Supreme Court on Friday listened to arguments regarding the issue that has been top of mind for most home-based care providers for the […]

The year 2020 ended on a note of hope, with the 8th December vaccination of 91-year-old Margaret Keenan, the world’s first person to receive a […]

The World Health Organization (WHO) recently held a virtual panel discussion during Digital Health Week with speakers from around the world to talk about global […]

The U.S. Occupational Safety and Health Administration (OSHA) regulation requiring workers at companies with 100 or more employees to be vaccinated against COVID-19 or undergo […]

On the last day of November, a nationwide injunction was issued by a federal judge in Louisiana to freeze the COVID-19 vaccine mandate from the […]

On Monday, the Supreme Court refused to block New York state’s requirement that health care workers must be vaccinated against COVID-19. For home-based care providers, […]

This article is an exclusive feature available as a part of your subscription to HHCN + In 2013, the Obama administration included live-in caregivers in […]

The health care worker vaccine mandate is now on ice. A federal judge on Tuesday issued a preliminary injunction to stop the start of the […]

First it was the COVID-19 vaccination mandate from the U.S. Occupational Safety and Health Administration (OSHA). Now, it’s the one from the Centers for Medicare […]

One of the largest home health groups in New York has agreed to pay millions of dollars to resolve wage-and-hour violations. The home health group […]

Due to the nature of the job, home-based care workers face the risk of experiencing workplace violence. It’s paramount for providers to enact measures that […]

The proliferation of hospital-at-home models has been one of the major home-based care trends during the COVID-19 crisis, and players on all sides are bullish […]

Formedix has been strong advocates for the use of CDISC data standards in clinical and non-clinical research for decades, ever since we realized how it […]

With more eyes than ever on the home care industry, experts are trying to read the tea leaves to determine if greater standardization will eventually […]

The U.S. Department of Health and Human Services (HHS) announced Friday that it is releasing another $25.5 billion under the Provider Relief Fund (PRF). Since […]

An interesting paper from Kevin Chiu in the Journal of Health Economics looks at the relationship between certificate of need laws and health outcomes: Certificate […]

Recently, the Food and Drug Administration (FDA) approved Prograf (tacrolimus) in combination with other immunosuppressant drugs for the new indication of preventing organ rejection for […]

An action plan shared by the FDA earlier this year showed how the regulator is thinking about future regulations for AI and machine learning in […]

The FDA has extended a collaboration with the UK firm CN Bio to investigate a “lung-on-a-chip” model to investigate inhaled drugs. In a statement the […]

Glasgow, UK – (March 29, 2021) – Formedix, a leading provider of clinical trial software solutions, announces the launch of ryze, an innovative all-in-one cloud-based […]

Europe’s regulator has begun a rolling review of Sinovac’s COVID-19 vaccine, known as Vero Cell. The vaccine is based on an inactivated SARS-CoV-2 virus, which […]

Roche’s Tecentriq can stay on the US market as a therapy for triple negative breast cancer, advisers to the FDA have said as part of […]

In the final days of the Trump Administration, the Department of Health and Human Services sought to deregulate 83 different types of medical devices, citing […]

The benefits of Johnson & Johnson’s COVID-19 vaccine outweigh a tiny risk of unusual blood clots forming, Europe’s drugs regulator has said. In a decision […]

After a long and troubled development the fate of Pfizer/Lilly’s tanezumab painkiller injection is in the balance ahead of a key meeting of advisers later […]

The U.S. Senate on Thursday narrowly confirmed California Attorney General Xavier Becerra as the next secretary of the Department of Health and Human Services (HHS). […]

The answer to this question likely will be shaped in part by a new multilateral working group that will include not only representatives from the […]

The federal price transparency rule for hospitals went into effect Jan. 1, but a new analysis shows that a majority of providers studied were “unambiguously […]

Despite the once-in-a-generation COVID-19 pandemic, the home health industry’s transition to the Patient-Driven Groupings Model (PDGM) was pretty seamless. Looking into the not-too-distant future, the […]

European safety regulators have launched an investigation into the safety of a batch of the Oxford University/AstraZeneca COVID-19 vaccine, as countries including Denmark suspended its […]

UK chancellor Rishi Sunak has announced a budget loaded with initiatives designed to kick-start the UK’s economy as it recovers from the coronavirus pandemic, with […]

Diversity in clinical trials Recently, the FDA’s Center for Drug Evaluation and Research’s (CDER’s) released its 2020 Drug Trials Snapshots Summary Report. Part of the […]

How do you evaluate treatment efficacy and safety outside of the clinical trial setting? This is not just a question of academic interest. In last […]

China’s national regulator has approved Sinovac Biotech’s COVID-19 vaccine for use by the general public. This is the second vaccine approved by China’s National Medical […]

The change of administration in the US has also seen a new leader at the FDA, with Dr Janet Woodcock appointed as interim commissioner by […]

A white paper has outlined a pathway that could allow Italy to realise the potential of digital therapeutics (DTx). The official journal of the Smith […]

Novavax’s COVID-19 vaccine could be added to the UK’s growing arsenal of shots against the disease after it showed 89.3% efficacy in a late-stage trial. […]

The Covid-19 pandemic has put further pressure on clinical trial suppliers to be fleet of foot in getting products dispatched when a new patient is […]

AstraZeneca has said it has had “constructive talks” with European leaders in a bid to cool the row over vaccine supplies to the EU. The […]

President Joe Biden looks likely to ditch the previous administration’s move to create a quicker route to market for digital health products and other devices […]

President Joe Biden focused on the ongoing COVID-19 pandemic during his first full day in office Thursday. In doing so, he once again drew attention […]

The agency published its first action plan last week for how it plans to regulate machine learning-based software as a medical device. To start, the […]

Agency veteran Dr Janet Woodcock is the new interim FDA commissioner appointed by president Joe Biden to replace outgoing Trump appointee Stephen Hahn. Woodcock has […]

The U.S. Department of Labor (DOL) recently issued an opinion letter allowing home care agencies to implement consistent shift rates that include pre-payment of overtime […]

Wage-and-hour litigation, confusing state-level regulations and an increase in federal audits were among the biggest legal trends of 2020. While many of these issues will […]

AI tools increasingly occupy a regulatory gray area in healthcare. For clinicians to assess whether they are trustworthy, they need transparency on how they work, […]

The European Union has approved the coronavirus vaccine from Moderna, leaving the UK trailing because of changes to post-Brexit drug approval rules. With the UK […]

The FDA approval process is slow. Even for COVID-19 vaccines, the process can be time consuming. However, regulators around the world do have some expedited […]

BioNTech CEO Ugur Sahin has said that it is “highly likely” the company’s COVID-19 vaccine will work against the more infectious strain circulating in the […]

In late November, the aging-focused advocacy organization LeadingAge wrote a note to Congress, calling for relief on behalf of the 5,500 adult day service providers […]

Reviewers from the FDA have given their blessing to the Pfizer/BioNTech COVID-19 vaccine ahead of a key meeting tomorrow – but the regulator noted that […]

President-elect Joe Biden has picked Xavier Becerra, the current attorney general of California, to lead the U.S. Department of Health and Human Services (HHS), according […]

Bureaucracy, political upheaval, and lack of regulations continue to make Latin America a difficult market for European and North American pharma to enter – and […]

This is the question that a recent paper by Chorniy et al. (2020) attempt to answer. This issue is clearly very relevant as the UK […]

Pfizer and BioNTech are preparing to deliver their COVID-19 vaccine to the UK after the country’s drugs regulator became the first authority in the world […]

Over the past several years, it seems as though the home health industry has been inching closer and closer toward the elimination of Medicare’s strict […]

In an effort to boost value-based care, the U.S. Centers for Medicare & Medicaid Services (CMS) recently announced that it has finalized changes to the […]

In an effort to increase hospital capacity amid the current COVID-19 surge, the U.S. Centers for Medicare & Medicaid Services (CMS) on Wednesday announced “unprecedented” […]

The FDA has approved Alnylam’s gene silencing drug Oxlumo, the first treatment for primary hyperoxaluria type 1 (PH1), an ultra-rare and life-threatening genetic disorder. Oxlumo […]

Given the incredible speed in which vaccine development for COVID-19 is occurring, it merits thinking back on the typical development and approval time of previous […]

COVID-19 vaccines could be approved and available early next year in the UK ahead of European countries, after its national regulator began its own rolling […]

The U.S. Centers for Medicare & Medicaid Services (CMS) released its final home health payment rule for CY 2021 on Thursday, with essentially no changes […]

In response to ongoing opposition from both lawmakers and providers, the Department of Health and Human Services (HHS) recently announced it has made changes to […]

The 21st Century Cures Act required the Food and Drug Administration (FDA) establish a program for evaluating the use of real-world data (RWD) to support […]

Pfizer said it will wait until after the US election to file its COVID-19 vaccine with the US regulator, as it waits for important safety […]

The boom in digital tech over COVID means that pharma can move faster than ever – but the industry’s traditional processes for asset approval are […]

There are a host of new, previously unimaginable tools and techniques – from analytics to robotic process automation (RPA) and artificial intelligence (AI) – available […]

European regulators have started a first ‘rolling review’ of a COVID-19 vaccine, which is being developed by AstraZeneca in collaboration with the University of Oxford. […]

SMi reports: Alan Franklin, CEO, ForwardVue Pharma will be presenting at the Ophthalmic Drugs conference The 4th Annual Ophthalmic Drugs Conference taking place on the […]

US biotech BioMarin has hit back with a filing for a rare disease drug, after suffering what was described by analysts as a “major setback” […]

The FDA has approved a new therapy for the rare muscle wasting disease Duchenne muscular dystrophy (DMD) as Japan’s NS Pharma takes on Sarepta and […]
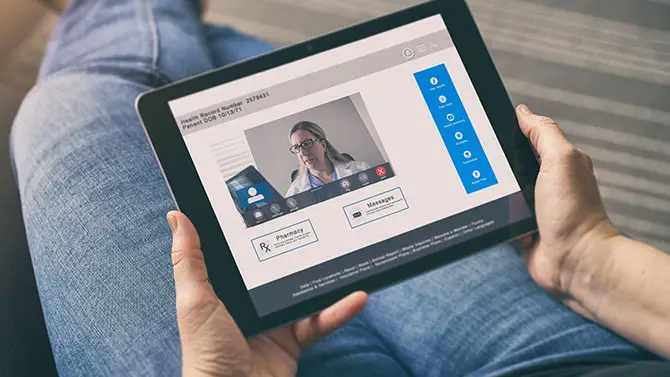
The Centers for Medicare & Medicaid Services (CMS) has proposed a new rule that would make certain COVID-19-related telehealth flexibilities introduced over the past few […]

Home-based care providers are at the forefront of the nation’s fight against the coronavirus; yet they still lack the full federal help they need to […]

For months now, home health providers have had to worry about an onslaught of new issues, from volume disruptions and personal protective equipment (PPE) shortages, […]

The Paycheck Protection Program (PPP) has helped home-based care providers who received less than $150,000 in loans save more than 160,000 jobs nationwide, according to […]

Home-based care providers who received loans under the Paycheck Protection Program (PPP) could face unexpected expenses when it comes time to file their 2020 taxes. […]

The UK’s regulator has granted early access to Alnylam’s ultra-rare disease drug lumasiran, allowing patients with primary hyperoxaluria type 1 to gain access ahead of […]

As we have discussed here previously, real-world data (RWD) and real-world evidence (RWE) offer many potential benefits in every stage of the drug discovery and […]
Copyright © 2024 | WordPress Theme by MH Themes