
Pfizer’s Arena buyout looks canny, as etrasimod aces phase 3 trials
Pfizer placed a $6.7 billion bet on the outcome of two phase 3 trials of Arena Pharmaceuticals’ etrasimod in ulcerative colitis (UC) when it acquired […]

Pfizer placed a $6.7 billion bet on the outcome of two phase 3 trials of Arena Pharmaceuticals’ etrasimod in ulcerative colitis (UC) when it acquired […]

Eli Lilly has the results it was looking for in a second, confirmatory trial of its anti-IL-23p19 antibody mirikizumab in ulcerative colitis as it waits […]

A pharmaceutical giant reported a Phase 3 success for a potential blockbuster drug, while four other companies reported disappointing data from their respective clinical trials. […]

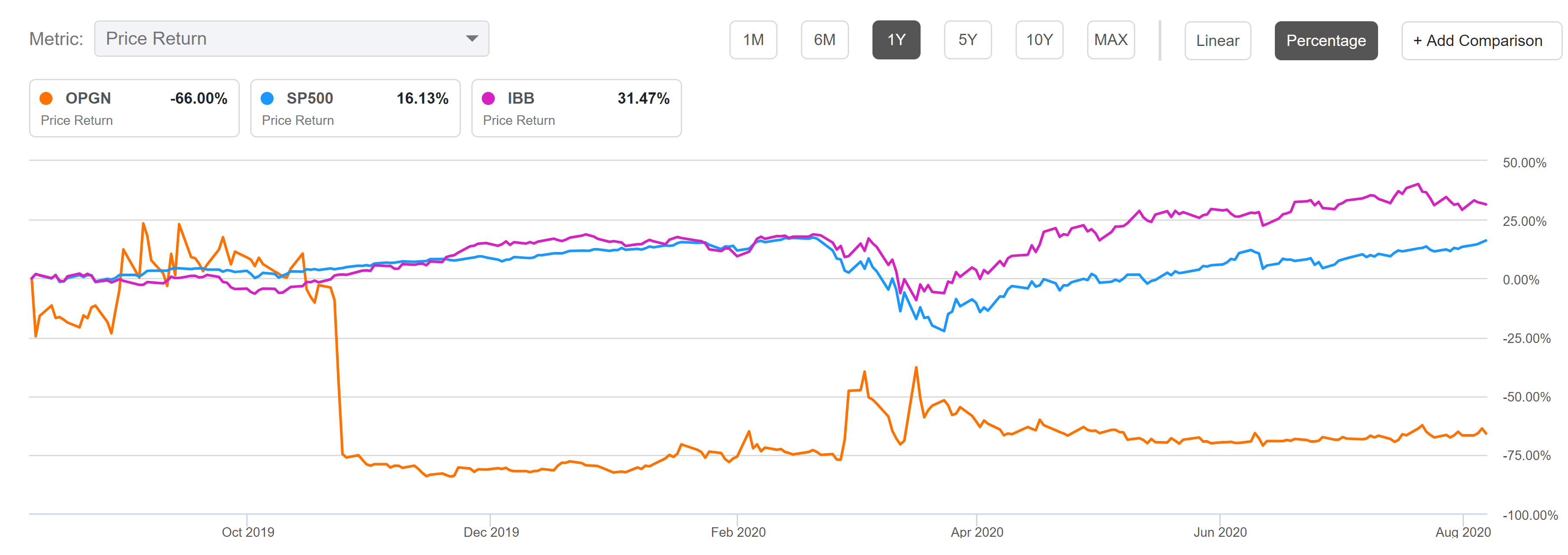
It’s only been two weeks since Pfizer closed its $6.7 billion takeover of Arena Pharmaceuticals, and it already has the positive data it was hoping […]

Eli Lilly has reported phase 3 results with its ulcerative colitis challenger mirikizumab, as it waits for the readout of a second trial in the […]

Pfizer has found another use for the windfall profits it is making from it BioNTech-partnered COVID-19 vaccine, agreeing to buy Arena Pharma and its late-stage […]

Sales of blockbuster Pfizer drug Xeljanz are under pressure from a stronger safety warning placed across that drug’s entire class. In acquiring Arena Pharmaceuticals for […]

Shots: The company has completed the enrolment in the P-II CALDOSE-1 trial to evaluate the efficacy & safety of IMU-838 (10/30/45mg, PO) vs PBO in […]

Bristol Myers Squibb drug deucravacitinib missed the main and secondary goals of a Phase 2 study in ulcerative colitis. Despite the clinical trial failure, the […]

Bristol-Myers Squibb’s hopes of extending the use of its highly-anticipated psoriasis pill deucravacitinib into inflammatory bowel disease (IBD) have been hit by a mid-stage failure […]

Shots: The P-II LATTICE-UC study evaluates the safety and efficacy of deucravacitinib (PO) vs PBO in patients with mod. to sev. UC The trial did […]

Shots: The non-IND/CTA study evaluates KB295 in patients with UC. The study evaluates other exploratory assessments including changes in microbiome composition, SCFA levels & biomarkers […]

The Theravance Biopharma drug is the lead program in an alliance with Johnson & Johnson subsidiary Janssen Biotech, which paid $100 million up front to […]

Shots: The P-IIb dose-finding induction study evaluates izencitinib (PO, qd) vs PBO in 239 patients with a mod. to sev. active UC for 8wks. The […]

Shots: Takeda will lead the development of FIN-524/TAK-524 to treat UC prior to the start of its clinical program & will leverage its expertise in […]

Galapagos has had to send its SIK inhibitor programme back to the drawing board after its lead compound GLPG3970 has failed two of three clinical […]

Shots: The P-III SELECTION trial comprises 2 induction trials & a maintenance trial evaluating filgotinib (200/100 mg) vs PBO in adult patients with active UC […]

Eli Lilly has walked away from a major indication for its blockbuster hopeful mirikizumab, shelving plans to file it for psoriasis as the market is […]

Shots: The P-II study involves assessing Olamkicept (300/600mg, q2w, IV) vs PBO in 91 patients in a ratio (1:1:1) who had active UC with a […]

Shots: The submission is based on P-IIb/III SELECTION study evaluating the efficacy and safety of filgotinib (200mg, qd) vs PBO in patients with mod. to […]

Shots: The approval is based on P-III ENVISION I study that involves assessing the efficacy, safety, and PK of Humira (SC) in pediatric patients aged […]

Merck & Co/MSD has agreed to buy Pandion for $1.85 billion, bolting on a pipeline of drugs for autoimmune and other immunological disorders headed by […]

Shots: The approval is based on P-III ENVISION I study that involves assessing Humira (SC) vs PBO in pediatric patients aged 4-17yrs. with mod. to […]

Shots: The P-III U-ACCOMPLISH induction study involves assessing upadacitinib (45 mg, qd) vs PBO in patients with mod. to sev. UC The study met its […]

Is the exaggerated reaction of many Crohn’s disease patients to baker’s, brewer’s, and nutritional yeast just a consequence of their inflamed leaky gut, or might […]

Shots: The P-II AMBER2 study assessing the efficacy and safety of SHR0302 (8mg qd/4mg bid/4mg qd) vs PBO in 164 adults with moderate to severe […]

Bristol-Myers Squibb’s commercial hopes for new multiple sclerosis drug Zeposia have yet to bear fruit thanks to the pandemic, but it’s already cued up a […]

GlaxoSmithKline has suffered another research setback after it axed a phase 2 trial of an anti-LAG3 drug in patients with ulcerative colitis, following a major […]

What You Should Know: – Trellus Health secures $5M in seed funding and collaborates with Mount Sinai to deliver resilience-driven, connected care, integrating expert clinical […]

The FDA has rejected Gilead and Galapagos’ rheumatoid arthritis (RA) pill filgotinib over concerns that it could damage male fertility, prompting speculation that the big […]
Roche Set to Compete With Biogen and Novartis in the SMA Market With its First Oral Therapy The US FDA has recommended the use of […]
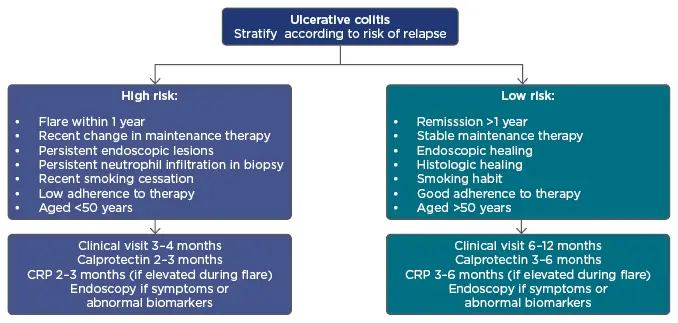
Roche’s late-stage trial programme for ulcerative colitis candidate etrolizumab has generated some positive and negative results, but leans firmly towards the latter. The Swiss drugmaker […]

Shots: In the HIBISCUS I induction study, in patients without prior anti-TNF treatment, etrolizumab met its 1EPs while in HIBISCUS II study in the same […]
Copyright © 2024 | WordPress Theme by MH Themes