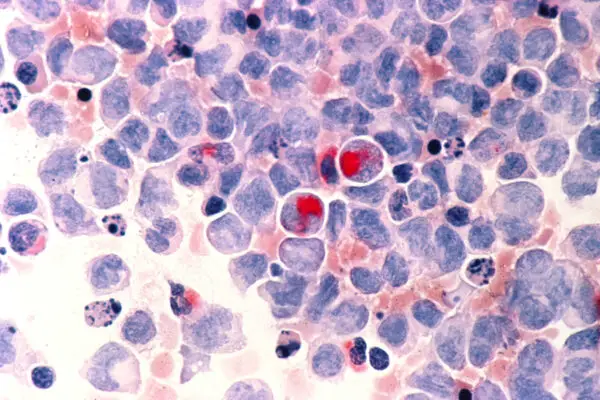
The FDA placed a partial clinical hold on a Kura Oncology leukemia drug following the death of one patient in the early-stage study. Kura executives said the death may be related to differentiation syndrome, a known complication of leukemia therapies.