
The diagnostic dilemma of co-infections requires novel solutions
Metagenomics offers advances for infectious disease diagnostics by providing the diagnostic technology to detect a potentially unlimited numbers of known and novel microbes from a […]

Metagenomics offers advances for infectious disease diagnostics by providing the diagnostic technology to detect a potentially unlimited numbers of known and novel microbes from a […]

In April 2020, I wrote a blog post entitled “Exploiting similarities between SARS-CoV-2 and other viruses to accelerate vaccine and therapy development,” where I described […]

Nina Garrett, R&D director at Abingdon Health discusses the key role antibody testing is playing in the pandemic and how an integrated approach with vaccines […]

In an interview with PharmaShots, Dr. Gill Diamond, Scientific Advisory Board Member and Joshua McClure, CEO and Co-founder of Maxwell Biosciences share their views on […]

Shots: BMS to get non-exclusive access to Xencor’s Xtend Fc technology to extend the half-life of a novel Ab combination therapy that is intended to […]

In the fight against emerging coronavirus variants and spike mutations, the need for long-lasting immunity is greater than ever. This is why the time is […]

Shots: Qiagen’s QIAstat-Dx and NeuMoDx tests remain accurate & effective in detecting SARS-CoV-2 infections The company started cross-checking variants in May’2020 & assessed the tests […]

Shots: The company presents the ongoing program of SARS-CoV-2 mutation surveillance for STI-2020 currently in pre/ clinical development for COVID-19 The data provides evidence of […]

Medivir, IGM Biosciences enters into an exclusive licensing agreement for Birinapant Medivir AB has entered into an exclusive licensing agreement with IGM Biosciences to receive […]

The two companies are working together to detect instances of the more contagious B.1.1.7 variant in the U.S. After Colorado identified the first case in […]

In a recent interview with PharmaShots Daniel-Adriano Silva, PhD, Co-founder and Vice President Head of Research at Neoleukin shared the significance and promise of the findings […]

Shots: Roche has launched Elecsys SARS-CoV-2 Ag test as an aid in the diagnosis of SARS-CoV-2 infections, in the markets accepting the CE Mark. Additionally, […]

Researchers at the Wellcome Sanger Institute in the UK are working on software to monitor the genome of SARS-CoV-2 coronavirus in the hope of spotting […]

Shots: Qiagen has initiated the commercialization of the portable digital QIAreach SARS-CoV-2 antigen test in the US that can be used by laboratories to detect […]

The company announced data on the combination of two antibodies against the SARS-CoV-2 virus, showing significant reductions in viral load compared with placebo. The company […]
The guidance calls for a median two months’ worth of follow-up safety data from well-designed Phase III studies as a precondition for a vaccine receiving […]

Shots: The Rolling Submission is based on the preliminary results from pre-clinical and early clinical studies in adults, which shows that BNT162b2 triggers the production […]

While the White House and doctors at Walter Reed have sought to reassure the public that Trump’s condition is improving, he has received numerous drugs […]

The company said it was meeting real-time supply demands for Veklury (remdesivir) in the U.S. and anticipated meeting global demands for the drug worldwide. AmerisourceBergen […]

Reuters reported Wednesday that U.S. regulators would look at data from previous clinical trials that came from the same scientists who created the vaccine, AZD1222, […]
The company published interim data from the Phase I study of mRNA-1273 in 40 older and elderly adults, showing responses comparable to those seen in […]
The company said that those who received the two-antibody cocktail REGN-COV2 showed a lower viral loads and faster alleviation of symptoms than those who received […]

The drug, AZD7442, is designed to potentially provide at least six months of protection from Covid-19. The trial, which is funded by federal defense and […]

The study, by researchers at Harvard, MIT, The Broad and other institutions, used genomic analysis of cases linked to the Biogen meeting, linking much of […]

Shots: Qiagen reports the launch of the Access Anti-SARS-CoV-2 Total test that runs on a portable device and provides accurate results on Total Ig Abs […]

The FDA commissioner took to Twitter Monday night to defend his performance at the agency, which came under sharp criticism amid accusations that the authorization […]

The biotech company reached a deal with the European Commission to supply an initial 80 million doses, while European Union member states will have the […]

While the emergency use authorization granted Sunday was not seen as scientifically unwarranted, it drew concerns from some experts that it could hinder enrollment in […]

Over the weekend, Trump called the agency, without evidence, part of a “deep state” that was slow-walking vaccines and drugs for Covid-19. The next day, […]
The companies also said their Phase II/III study, slated to enroll about 30,000 participants, had already dosed more than 11,000.
Doctors have called for rules to be tightened on coronavirus antibody tests in the UK amid fears that false readings could put the public at […]
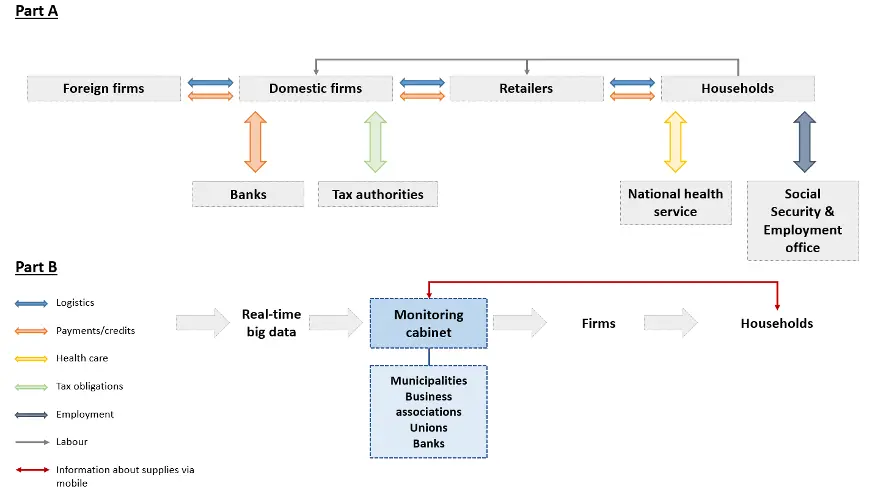
An overview of various aspects of the supply chain that would be crucial in the production and distribution of COVID-19 Vaccine. Write to us at […]
The New York Times reported that the FDA was prepared to issue an emergency use authorization for the treatment last week, but NIH Director Francis […]
The deal brings Roche’s considerable manufacturing heft to Regeneron’s efforts to develop the two-antibody cocktail, REGN-COV2. Regeneron will distribute the drug in the U.S., while […]
Could AI prevent future pandemics by developing an armoury of drugs that work against all coronaviruses? This is a question that a consortium of European […]

Earlier this month, Ohio Gov. Mike DeWine received a false-positive result from a rapid antigen test for Covid-19, raising concerns about the tests’ accuracy. The […]

The company plans to train 250 new racially and ethnically diverse clinical trial investigators and extend the reach of its trials to underserved rural and […]
In a commentary, physicians and pharmacists wrote that percentages of Black, Latino and Native American patients in two Phase III studies of the drug that […]
The agreement, which includes 100 million doses of mRNA-1273 and an option to acquire 400 million more, brings the total amount of money the biotech […]
Russian President Vladimir Putin said a vaccine developed at a Moscow research institute that appears to still be in Phase I development among fewer than […]
Gilead Sciences had said in its second-quarter earnings that it would partner with companies in North America, Europe and Asia to manufacture the antiviral drug, […]

Shots: The US FDA has granted EUA to the Vela’s PCR test that detects SARS-CoV-2 in nasopharyngeal and oropharyngeal swabs The manual version of the […]
The drug has consistently failed to show a benefit when tested in randomized, controlled clinical trials. Social science may explain why laypeople and doctors alike […]

Under the agreement with BARDA and the Department of Defense, the drugmaker would supply 100 million doses of its vaccine, for which it published preclinical […]

Three of the companies making drugs used in the Phase II I-SPY COVID-19 study – Amgen, AbbVie and Takeda – announced the patient enrollments Monday. […]

Financial terms of the deal were not disclosed, but it follows a $1.95 billion contract between the two companies and the U.S. government. Assuming clinical […]
The companies will provide 100 million doses of their jointly developed vaccine. But with a Phase I/II study planned in September, it is behind in […]

The company said that the COVACTA trial of Actemra in Covid-19 pneumonia had failed to meet its primary endpoint. Earlier this month, Regeneron and Sanofi […]
The companies said they had started a Phase II/III trial BNT162b2. The news comes the day after Moderna announced the launch of its Phase III […]
The company had received $483 million from BARDA in April to fund the development of the vaccine, mRNA-1273, through Phase III, but that amount was […]

A US government agency is to give a further $472 million to biotech Moderna, after it announced plans to expand a phase 3 trial for […]

The study adds to the growing body of evidence that the drug, promoted early in the pandemic by President Trump, is ineffective, despite its getting […]

MedCity spoke with Evidation and BrightInsight, two startups that have recently announced funding rounds, amid a Covid-19 pandemic worsening in the U.S.

The company said the CareStart Covid-19 MDx RT-PCR test is designed to detect RNA from the SARS-CoV-2 virus and produce results within 83 minutes.

The companies will supply 100 million doses of the vaccine by the end of the year, and the government will have the option to acquire […]

“Everybody’s an infectious disease company now,” ARCH Venture Partners Managing Director Bob Nelsen said. By contrast, a report last year showed a huge decline in […]

The company said that patients receiving the drug, SNG001, had a 79% lower risk of requiring ventilation or dying compared with those on placebo, sending […]
The company announced the publication of interim data from the Phase I/II study of a vaccine it is developing with the University of Oxford. The […]

Amid fears of a premature emergency use authorization for a Covid-19 vaccine granted for political reasons, the guidance that the FDA released at the beginning […]
The Beijing company said it selected the United Arab Emirates because of its ethnic diversity, which will enable it to determine the feasibility of taking […]
A joint report by cybersecurity agencies in the U.K. and Canada, endorsed by their U.S. counterparts, states that the hacker group Cozy Bear had been […]

Evaluation of durability is still under evaluation, but the responses to mRNA-1273 show neutralizing antibody titers multiple times higher than those of recovered patients. Additionally, […]
The companies anticipate releasing more clinical data from the BNT162 messenger RNA vaccine program and potentially starting a 30,000-participant Phase IIb/III study this month.

The study is enrolling 60 healthy volunteers aged 18-45, but the company hopes it will form the basis of trials of the drug as an […]

Research on the drug’s molecular target dates back to the 1950s. A spokesperson for Gilead noted that the company anticipates potentially investing more than $1 […]

The company said it is on track to start its 30,000-participant Phase III trial this month and has finished manufacturing enough supplies of the vaccine […]

The NIAID create the Covid-19 Prevention Trials Network by combining four existing ones, primarily focused on HIV and AIDS, which will continue that work in […]

The contract covers manufacture and supply of the two-antibody drug cocktail REGN-COV2. The company had announced Monday that it entered the drug into three late-stage […]

The company is running a Phase I/II study that started in May in Australia and plans to open a Phase III study of its vaccine […]

Researchers in Spain, one of Europe’s worst-hit countries, randomly selected more than 61,000 people, but found that even in places with high infection rates like […]

The company also said that the Phase III study of the autoimmune disease drug Kevzara that it was running with Sanofi failed to meet either […]
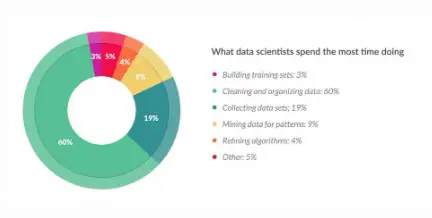
Since the spread of COVID-19 was first reported, researchers of all types have mobilized to meet the challenges its causative agent, SARS-CoV-2, presents to the […]

Drug development is a multidisciplinary field that requires a drug to successfully pass through different stages — from chemical design to biological experimentation on cells […]

Today I am going to share some important ways that doctors, hospitals and healthcare marketers can use social media to inform the public during the […]
Copyright © 2024 | WordPress Theme by MH Themes