
Abecma hits the mark in earlier-stage multiple myeloma
Bristol-Myers Squibb is on course to move its BCMA-targeting CAR-T therapy further up the treatment pathway in multiple myeloma, after reporting that it met its […]

Bristol-Myers Squibb is on course to move its BCMA-targeting CAR-T therapy further up the treatment pathway in multiple myeloma, after reporting that it met its […]

Bristol Myers Squibb has data showing that its multiple myeloma treatment Abecma beat the standard of care, preliminary Phase 3 results that could support moving […]

Bristol-Myers Squibb has formed an R&D partnership with GentiBio, aimed at developing regulatory T cel (Treg) therapies for inflammatory bowel disease (IBD). The deal includes […]

AI could address the most significant issue in drug discovery, finds Ben Hargreaves, and also offers the potential to use tissue samples to create precision […]

Bristol-Myers Squibb has revealed that its checkpoint inhibitor combination of Opdivo and Yervoy failed a phase 3 trial as adjuvant (post-surgery) therapy for renal cell […]

Bristol-Myers Squibb’s new immune checkpoint inhibitor combination Opdualag has only been on the US market for a few weeks, but is already off to a […]

Roche has been trumpeting the rapid uptake of its new eye disease drug Vabysmo as it takes on Regeneron and Bayer’s mighty Eylea in the […]

NICE has recommended routine use of Bristol-Myers Squibb’s Opdivo for the adjuvant treatment of some patients with urothelial carcinoma (UC), the most common form of […]

Novartis is still waiting for FDA approval of its PD-1/PD-L1 inhibitor latecomer tislelizumab in relapsed oesophageal cancer, but is already planning a move the drug […]

Just weeks after Bristol-Myers Squibb claimed an FDA green light for Opdivo as a neoadjuvant therapy for non-small cell lung cancer, AstraZeneca is looking to […]

The new FDA approval for Breyanzi moves the Bristol Myers Squibb cancer cell therapy into an earlier line of treatment for an aggressive type of […]

Owkin has enticed another big pharma partner with its artificial intelligence-powered clinical development platform, Bristol-Myers Squibb, which is paying $80 million upfront to tap into […]

Eisai’s first-ever attempt at developing an antibody-drug conjugate for cancer was rewarded with a $3.1 billion licensing deal from Bristol-Myers Squibb, and new data reported […]

Bristol-Myers Squibb’s all-cash offer for Turning Point Therapeutics – which values the biotech at $4.1 billion – bolsters its position in precision oncology and gives […]

Bristol Myers Squibb is splashing out $4.1 billion to acquire Turning Point Therapeutics, a clinical-stage biotech whose lead drug is currently in pivotal testing as […]

The FDA has cleared two combination drug regimens based on Bristol-Myers Squibb’s PD-1 inhibitor Opdivo for previously-untreated advanced oesophageal cancer, encroaching on territory held by […]

Pfizer placed a $6.7 billion bet on the outcome of two phase 3 trials of Arena Pharmaceuticals’ etrasimod in ulcerative colitis (UC) when it acquired […]

At the upcoming INVEST PharmaTech virtual conference scheduled for July 26, the conversations will span myriad topics at the intersection of digital health and drug […]

Bristol-Myers Squibb’s flurry of dealmaking has continued with en expansion of its partnership with BridgeBio on drugs targeting SHP2, in the hope of combining them […]

Bristol Myer Squibb is getting rights to BBP-398, a contender in the chase for drugs that address difficult-to-target KRAS mutations. Meanwhile, BridgeBio Pharma receives some […]

Bristol-Myers Squibb’s Celgene unit has clearly been impressed by progress in its four-year-old alliance with Evotec on protein degradation. It’s just paid $200 million to […]

As an oncologist, Kristen Hege first encountered cancer cell therapy research in the mid-1990s. Now as a Bristol Myers Squibb executive, she oversees efforts to […]

CAR T cell therapies for cancer still pose challenges in manufacturing, safety, and the ability to address solid tumors. A panel at the World Medical […]

Bristol Myers Squibb drug Camzyos has FDA approval for treating obstructive hypertrophic cardiomyopathy, a rare and potentially fatal heart disorder. The drug is projected to […]

Bristol-Myers Squibb’s has become the first company to bring a cardiac myosin inhibitor through to FDA approval, claiming a green light from the US regulator […]

Four years after Bristol-Myers Squibb paid $1.85 billion upfront to make Nektar’s bempegaldesleukin a companion drug to its cancer immunotherapy Opdivo, the alliance is being […]

Bristol Myers Squibb and Nektar Therapeutics are ending a cancer drug research alliance after data showed the combination of their respective drugs failed clinical trials […]

Cytokinetics’ hopes of resurrecting cardiac myosin inhibitor omecamtiv mecarbil have suffered another setback, after the drug failed to show a benefit in a phase 3 […]

Gilead Sciences’ Kite Pharma has become the first drugmaker to get FDA approval to use a CAR-T therapy after just one earlier systemic therapy, moving […]

Johnson & Johnson is in course for approval of its multiple myeloma CAR-T therapy Carvykti in the EU the next few weeks, after getting a […]

It’s only been two weeks since Pfizer closed its $6.7 billion takeover of Arena Pharmaceuticals, and it already has the positive data it was hoping […]

Bristol-Myers Squibb is the first big pharma to strike a deal with Volastra Therapeutics, a start-up that has developed a drug discovery platform based on […]

Bristol Myers Squibb’s Opdualag has been approved by the FDA, a first for a drug that goes after a new cancer target called LAG-3. The […]

Bristol-Myers Squibb has won FDA approval for the first LAG-3 inhibitor drug ahead of its rivals in cancer immunotherapy including Merck & Co. The US […]

Four years ago, Bristol-Myers Squibb paid $1.85 billion upfront to claim rights to Nektar’s bempegaldesleukin, setting it up to be companion drug to its cancer […]

Bristol Myers Squibb bet more than $1.8 billion that Nektar Therapeutics’ drug could be combined with the pharma giant’s Opdivo to expand the reach of […]

Bristol-Myers Squibb’s blood cancer blockbuster Revlimid is finally facing generic competition in the US, which will eat into revenues from a drug that made a […]

Bristol-Myers Squibb’s Opdivo has been playing second fiddle to Merck & Co’s Keytruda in the non-small cell lung cancer (NSCLC) market for years, but now […]

Neuroscience drug developer Biohaven Pharmaceuticals is expanding its pipeline to include epilepsy and spinal muscular atrophy. The drug developer is acquiring Channel Bioscience and licensing […]

Bristol-Myers Squibb has new data backing up the value of mavacamten for obstructive hypertrophic cardiomyopathy (HCM), as it waits for a delayed verdict on its […]

The FDA has granted fast-track status to Bayer’s oral Factor Xia inhibitor asundexian, which is in phase 2 testing as an anticoagulant that could offer […]

Bristol-Myers Squibb chief executive Giovanni Caforio has promised to invest in the company’s pipeline at the virtual JP Morgan Healthcare Conference, and demonstrated that commitment […]

Bristol Myers Squibb is partnering with Century Therapeutics, placing a $150 million bet that the biotech’s technology for engineering stem cells can produce new off-the-shelf […]

Amgen has won FDA approval for a stronger label for its oral plaque psoriasis therapy Otezla, as it prepares for competition from Bristol-Myers Squibb’s much-touted […]

The insurers referred to deals Gilead struck with Janssen, Bristol Myers Squibb to prevent the generic version of its retroviral medications from being used as […]

Bristol-Myers Squibb’s rheumatoid arthritis drug Orencia has been approved by the FDA to prevent graft versus host disease (GvHD), a serious complication of haematopoietic stem […]

Bristol Myers Squibb has notched another FDA approval for its blockbuster immunology drug Orencia, this time for preventing acute graft versus host disease in bone […]

Bristol Myers Squibb is paying $150 million up front for global rights to an Immatics biologic drug designed to recruit a patient’s T cells to […]

Pfizer has found another use for the windfall profits it is making from it BioNTech-partnered COVID-19 vaccine, agreeing to buy Arena Pharma and its late-stage […]

A pair of trials have suggested that CAR-T therapies could offer an improved option for patients with large B cell lymphoma (LBCL) who have relapsed […]

The FDA has kicked off its review of Bristol-Myers Squibb’s psoriasis therapy deucravacitinib, setting an action date of 1 September next year, as the EU […]

Merck & Co’s Keytruda become the first cancer immunotherapy to be approved as a first-line oesophageal cancer treatment in the EU earlier this year, but […]

The company’s senior vice president for major markets (Europe, Canada and Japan) talks about the continued evolution of pharma’s approach to communications and commercialisation. The […]

Bristol Myers Squibb’s Zeposia (ozanimod) Receives CHMP’s Positive Opinion for Adult Patients with Moderately to Severely Active Ulcerative Colitis Published: 15 Oct, 2021 | Tags: […]

Shots: The CHMP’s recommendation is based on P-III True North study evaluating Zeposia as an induction and maintenance therapy vs PBO in adult patients with […]

Patients in Scotland with newly-diagnosed advanced kidney cancer can now access a therapy combining Bristol-Myers Squibb’s immunotherapy Opdivo with Ipsen’s targeted drug Cabometyx, ahead of […]

Bristol Myers Squibb drug deucravacitinib missed the main and secondary goals of a Phase 2 study in ulcerative colitis. Despite the clinical trial failure, the […]

Bristol-Myers Squibb’s hopes of extending the use of its highly-anticipated psoriasis pill deucravacitinib into inflammatory bowel disease (IBD) have been hit by a mid-stage failure […]

US biopharma company Acceleron is the next company heading for a takeover by a big pharma group – according to a Bloomberg report citing people […]

An artificial intelligence algorithm already shown to be able to detect atrial fibrillation (AF) in clinical testing has also shown its worth in a real-world […]

Gilead Sciences’ Kite Pharma has won a key victory on appeal in its long-running dispute with Bristol-Myers Squibb over patents relating to CAR-T therapies, overturning […]

Bristol-Myers Squibb has scored a win in its drive to get Opdivo into earlier lines of cancer therapy, getting FDA approval for the drug for […]

Bristol Myers Squibb has exercised an option on an immune-modulating drug candidate developed by UK artificial intelligence specialist Exscientia, paying $20 million for the buy-in […]

The FDA has approved GlaxoSmithKline’s latecomer PD-1 inhibitor Jemperli for a second use that will significantly extend the patient population eligible for treatment with the […]

Bristol-Myers Squibb’s HDAC inhibitor Istodax has been on the US market for a decade as a treatment for peripheral T-cell lymphoma (PTCL), but will now […]

Bristol-Myers Squibb is in a battle with Merck & Co for position in the upper gastrointestinal cancer immunotherapy market, and has just reinforced its position […]

Bristol Myers Squibb blockbuster drug Opdivo is being pulled from the market after failing to meet the main goal of a confirmatory study. The voluntary […]

Bristol-Myers Squibb has withdrawn its US marketing approval for PD-1 inhibitor Opdivo as a second-line, single-agent treatment for hepatocellular carcinoma, a common form of liver […]

Shares in Cytokinetics surged after it reported positive mid-stage results for CK-274, a drug for hypertrophic cardiomyopathy (HCM) that could tread on the toes of […]

During the last two decades, the landscape for psoriasis treatment has exploded. The advent of biologic therapies at the turn of the millennium and the […]

During the last two decades, the landscape for psoriasis treatment has exploded. The advent of biologic therapies at the turn of the millennium and the […]

Japan’s Eisai has hit the jackpot with its first ever antibody-drug conjugate (ADC) drug candidate, attracting a licensing deal from Bristol-Myers Squibb that could top […]

Bristol-Myers Squibb has taken another step towards achieving its blockbuster hopes for beta thalassaemia-associated anaemia therapy Reblozyl, after hitting the target in a phase 2 […]

Bristol-Myer Squibb has only just won FDA approval for its CAR-T Breyanzi in large B-cell lymphoma (LBCL), but is already eyeing an expansion into earlier-line […]

Johnson & Johnson has secured a six-month FDA review for its multiple myeloma CAR-T therapy ciltacabtagene autoleucel (cilta-cel), narrowing the lead for Bristol-Myers Squibb and […]

Bristol Myers Squibb reported Phase 3 data that show its experimental cancer immunotherapy relatlimab, combined with the company’s blockbuster drug Opdivo, was better at stopping […]

LAG-3 drugs from arch-rivals Bristol-Myers Squibb and Merck & Co look set to become talking points at this year’s ASCO conference after trial results showed […]

Exscientia has signed up another big pharma partner for its artificial intelligence (AI) based drug discovery platform, this time attracting a $50 million upfront buy-in […]

Bristol-Myers Squibb has paid $200 million, and promised up to $1.36 billion in “biobucks” for exclusive rights to a cancer antibody still in the labs […]

Bristol Myers Squibb already has a cancer program targeting the TIGIT protein, but in acquiring rights to Agenus’s drug, the pharma giant adds a bispecific […]

The FDA’s Oncologic Drugs Advisory Committee (ODAC) has voted to strip Merck & Co’s Keytruda of its accelerated approval in gastric and gastroesophageal junction (GEJ) […]

FDA advisors will scrutinise three cancer immunotherapies granted conditional approvals at a three-day meeting this week, to see if they should stay on the market. […]

Bristol-Myers Squibb has phase 3 data from its deucravacitinib psoriasis pill, showing it outperformed Amgen’s rival Otezla in patients with moderate to severe disease. The […]

Despite having the global pandemic, the biopharma companies were involved in multiple M&A, licensing deals. The major focus of the year remains the production and […]

The FDA has approved a Bristol Myers Squibb cell therapy made by engineering a patient’s immune cells to target a cancer protein called BCMA. The […]

The FDA has approved Bristol-Myers Squibb’s Abecma (idecabtagene vicleucel) for multiple myeloma, the first cell therapy to treat the disease. Abecma’s review was delayed after […]

Cho discusses Bristol Myers Squibb’s research efforts in the field of protein degradation to advance potential treatments for a broad range of diseases. In a […]

Ho Sung Cho, senior vice president of Biodiscovery Therapeutics at Bristol Myers Squibb, talks about lessons learned from drug discovery, recent success tied to Covid-19, […]

In an interview, BrightInsight Co-founder and CEO Kal Patel, MD, offered a preview of the report, The Role of Digital Health in Immuno-oncology Therapy Development […]

New phase 3 data have shored up the position of Merck & Co’s cancer immunotherapy Keytruda in the increasingly competitive first-line kidney cancer market. The […]

The pairing of Heat Biologics’ lead drug HS-110 with Bristol-Myers Squibb’s blockbuster checkpoint inhibitor Opdivo has boosted survival in patients with non-small cell lung cancer […]

Bristol-Myers Squibb finally has FDA approval for its CAR-T therapy liso-cel, which has been cleared by the US regulator as Breyanzi for certain forms of […]

After some setbacks and delays, the FDA approved Bristol Myers Squibb’s Breyanzi for certain lymphomas. It’s the third CAR T treatment to win the regulatory […]

Bristol-Myers Squibb’s commercial hopes for new multiple sclerosis drug Zeposia have yet to bear fruit thanks to the pandemic, but it’s already cued up a […]

NICE has said that the NHS should not fund Bristol-Myers Squibb’s multiple sclerosis pill Zeposia (ozanimod) for relapsing multiple sclerosis in first draft guidance. The […]

US cost effectiveness watchdog ICER found 10 examples of substantial price rises for top-selling medicines in 2019, and concluded that seven of those were not […]

Almirall and Tyris Collaborate to Develop Next Generation Gene Therapies Published: Jan 7, 2020 | Tags: Almirall, Tyris, Collaborate, Develop, Next Generation, Gene Therapies Sarepta […]

Shots: Evotec has received $6M as milestones under its iPSC-based neuroscience alliance with BMS. The milestones follow BMS’ decision to add another drug discovery project […]

Bristol Myers-Squibb has put out another downbeat “update” announcement about its immunotherapy Opdivo in an aggressive form of brain cancer, saying that it looks unlikely […]

Shots: The approval is based on P-lll ATTRACTION-3 study involve the assessment of Opdivo vs CT (docetaxel or paclitaxel) for patients with esophageal cancer refractory/ […]

The San Diego-based biopharma firm has several oncology therapies in development and has partnered in the past with the likes of Astellas, BeiGene and Bristol-Myers […]

Holders of a risky “bet” on three Bristol-Myers Squibb drugs are looking increasingly unlikely to get their pay-out after the company said COVID-19 travel restrictions […]

Bristol-Myers Squibb posted a solid set of financial results for the third quarter, but shares slid on investor fears that a payout tied new product […]

Bristol-Myers Squibb could be on the verge of a major coup in psoriasis after its deucravacitinib pill outperformed Amgen’s rival Otezla. BMS had to sell […]

insitro has landed another big biopharma partnership, signing a five-year collaboration with Bristol Myers Sqibb to develop therapies for amyotrophic lateral sclerosis (ALS) and frontotemporal […]

Bristol-Myers Squibb could be mere months away from claiming a second US approval for an Opdivo-based combination immunotherapy that will help defend its first-line kidney […]

The companies expect the deal to close in the fourth quarter. MyoKardia’s lead drug candidate is mavacamten, which it is developing for hypertrophic cardiomyopathy and […]

Bristol-Myers Squibb has said it will buy biotech MyoKardia in a $13.1 billion takeover that marks a step up in its development of cardiovascular drugs. […]

Bristol-Myers Squibb’s immunotherapy combination of Opdivo and Yervoy has been approved in the US in a new indication for untreated mesothelioma, a rare but aggressive […]

With the ink barely dry on a deal to develop Dragonfly’s lead cancer immunotherapy, Bristol-Myers Squibb’s business development team has agreed to buy Canadian biotech […]

Bristol-Myers Squibb has signed an agreement worth $475 million to develop a new form of cancer immunotherapy drug with US biotech Dragonfly Therapeutics. BMS is […]

The company plans to train 250 new racially and ethnically diverse clinical trial investigators and extend the reach of its trials to underserved rural and […]

The FDA has accepted TG Therapeutics’ filing for its lymphoma drug umbralisib, which if approved could compete against a combination therapy from Bristol-Myers Squibb and […]
Bristol-Myers Squibb’s Opdivo has improved survival in a trial involving patients with mesothelioma, a form of lung cancer, getting one up over rival checkpoint inhibitor […]
Roche’s checkpoint inhibitor Tecentriq has been cleared for another new use in the US – in melanoma – but could struggle to displace rival drugs […]
The companies’ resubmission of their application Wednesday seeking approval for idecabtagene vicleucel was in line with the timeline they provided in May, when the FDA […]
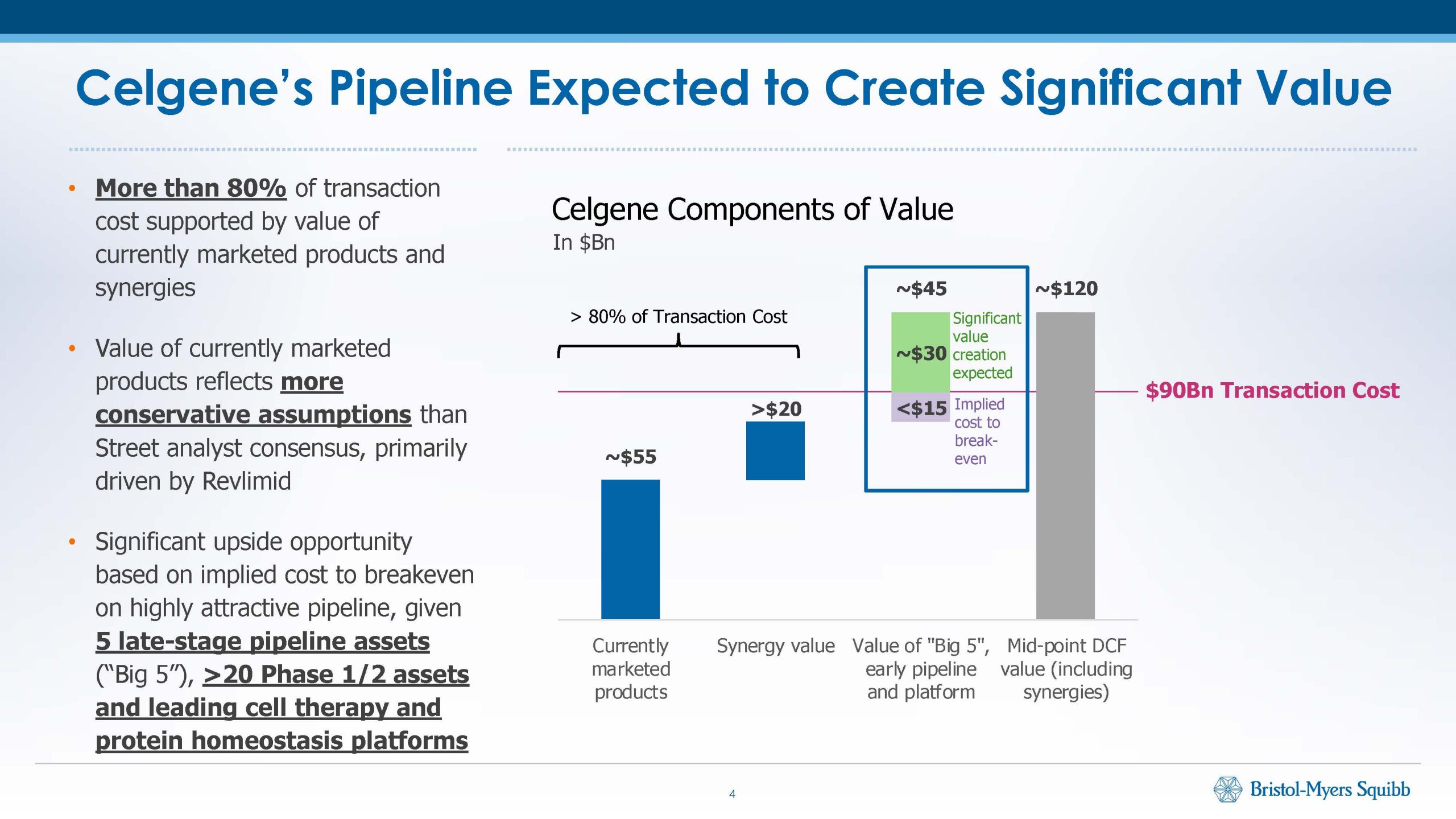
Bristol-Myers Squibb and bluebird bio have refiled their CAR-T therapy for multiple myeloma, leaving the outcome of a three drug “bet” with former Celgene shareholders […]
Copyright © 2024 | WordPress Theme by MH Themes