
Amgen splashes out on ChemoCentryx and its blockbuster hopeful
Ten months after ChemoCentryx scored an unexpected FDA approval for autoimmune disease therapy Tavneos, Amgen has swooped in with a $3.7 billion bid to buy […]

Ten months after ChemoCentryx scored an unexpected FDA approval for autoimmune disease therapy Tavneos, Amgen has swooped in with a $3.7 billion bid to buy […]

Amgen’s drug portfolio boasts several biologic drugs that are blockbuster sellers. In acquiring ChemoCentryx for $3.7 billion, the pharmaceutical giant gains a company entirely focused […]

AstraZeneca has signed a deal to buy US biotech TeneoTwo in a deal worth up to $1.27 billion that will boost its position therapies for […]

The nonprofit Digital Medicine Society is partnering with the likes of Google and the FDA to develop a free resource designed to help digital health […]

Amgen is planning to push its olpasiran candidate for reducing lipoprotein(a) into phase 3 as quickly as possible as it tries to catch up with […]

The American Society of Clinical Oncology’s annual meeting begins this week, including the presentation of more than 2,800 abstracts. Amgen, Roche, and Sierra Oncology are […]

Mirati Therapeutics will hear from the FDA on its KRAS inhibitor adagrasib later this year, hoping to compete with first-to-market rival Lumakras from Amgen. However, […]

How Embark® App combines a medical and digital approach to support patients in managing their Enbrel® (etanercept) treatment.

Novartis has joined the select cadre of drugmakers developing KRAS inhibitors for cancer, opening its account with phase 1b data on JDQ443 showing efficacy in […]

NICE has reversed its stance on UCB and Amgen’s severe osteoporosis therapy Evenity, saying out can be prescribed on the NHS for women with the […]

Armed with new clinical trial results, Amgen is expanding studies of its KRAS inhibitor Lumakras in pancreatic cancer, as it tries to extend the label […]

Amgen now has regulatory approval on both sides of the Atlantic for Lumykras, the culmination of many years of effort to bring a drug to […]

Fresh from a sizeable funding round that raised $370 million, artificial intelligence-based drug discovery firm Generate Biomedicines has just signed its first major partnering deal […]

At its second attempt, Novartis has won FDA approval for its cholesterol lowering drug inclisiran, which can reduce levels with just two injections a year. […]

Amgen has won FDA approval for a stronger label for its oral plaque psoriasis therapy Otezla, as it prepares for competition from Bristol-Myers Squibb’s much-touted […]

The FDA has approved AstraZeneca and Amgen’s severe asthma hope tezepelumab, exonerating the drugmakers’ decision to press ahead with a regulatory filing despite a failed […]

The FDA has kicked off its review of Bristol-Myers Squibb’s psoriasis therapy deucravacitinib, setting an action date of 1 September next year, as the EU […]

Jazz’ Sunosi (solriamfetol) Receives NICE Recommendation for the Treatment of Excessive Daytime Sleepiness Caused by Narcolepsy Published: Nov 19, 2021 | Tags: Jazz, Sunosi, solriamfetol, […]

Injectable antibodies targeting PCSK9 have failed to take off as expected in the cholesterol-lowering market, but Merck & Co hopes to do much better with […]

Shots: The EMA’s CHMP has adopted a positive opinion recommending the conditional marketing authorization for Lumykras to treat advanced NSCLC with KRAS G12C mutation & […]

Eisai Presents Results of Lecanemab in P-IIb Study 201 Study for the Treatment of Alzheimer’s Disease at CTAD 2021 Published: Nov 12, 2021 | Tags: […]

Shots: The P-IV HER-MES study evaluates the tolerability & efficacy of Aimovig (70/140mg) vs topiramate in 777 patients with episodic or chronic migraine (≥4 migraine […]

Bristol-Myers Squibb’s hopes of extending the use of its highly-anticipated psoriasis pill deucravacitinib into inflammatory bowel disease (IBD) have been hit by a mid-stage failure […]

Neumora Therapeutics unveiled $500 million in capital and a research partnership with Amgen. The startup analyzes neurological data to inform its drug research and development; […]

Shots: Neumora has received a $100M as an equity investment and obtains an exclusive global right to develop and commercialize Amgen programs targeting casein kinase 1 delta […]

BMS Reports EMA’s Validation of MAA for Relatlimab and Nivolumab as 1L Treatment for Unresectable or Metastatic Melanoma Published: Oct 1, 2021 | Tags: BMS, […]

The third quarter of 2021 highlights the approvals, clinical data & acquisitions. The new alliance observed in third quarter includes AzurRx signed a reverse triangular […]

Shots: The approval is based on the P-IIIb HAUSER-RCT study evaluating the safety & efficacy of Repatha (420mg, SC) vs PBO in a ratio (2:1) […]

Everest’s SPR206 Receives the NMPA’s IND Approval for the Treatment of MDR Gram-Negative Bacterial Infections Published: Sept 24, 2021 | Tags: Everest, SPR206, NMPA, IND, […]

Shots: The companies collaborated to evaluate the safety, tolerability & efficacy of Verastem’s VS-6766 + Amgen’s Lumakras in the P-I/II trial for patients with KRAS […]

Amgen and AstraZeneca haven’t won approval or announced a price for their severe asthma therapy tezepelumab should it reach the market, but ICER in the […]

Amgen has made progress with its plans to expand the use of its first-in-class KRAS inhibitor Lumakras, reporting encouraging results when the drug is used […]

Shots: The companies collaborated to evaluate the synergistic effects of BI 1701963 (SOS1::pan-KRAS inhibitor) + Lumakras (KRAS G12C inhibitor) in a P-I trial for adult […]

Shots: The approval is based on P-II CodeBreaK 100 trial evaluating the efficacy and tolerability of Lumakras (PO, qd) in 126 patients with KRAS G12C […]

Amgen’s first-in-class KRAS inhibitor Lumykras has been approved in Great Britain and arrives already armed with an agreement to allow early access while its cost-effectiveness […]

Shots: The P-III NAVIGATOR study involves assessing Tezepelumab + SOC vs PBO +SOC in adults (18–80yrs.old) and adolescents (12–17yrs. old) with severe, uncontrolled asthma. The […]

Amgen and partner AstraZeneca have said their recently filed severe asthma drug tezepelumab has shown impressive results in patients who also have nasal polyps, a […]

Ascendis’ Skytrofa (lonapegsomatropin-tcgd) Receives the US FDA’s Approval for Pediatric Growth Hormone Deficiency Published: Aug 27, 2021 | Tags: Ascendis, Skytrofa, lonapegsomatropin-tcgd, US, FDA, Approval, […]

Shots: The P-III HUYGENS study evaluates Repatha + optimized statin therapy vs optimized statin therapy alone in patients with ACS for 52wks. The study met […]

Immunology deals with physiological functioning of the immune system in states of both health and disease as well as malfunctions of the immune system in […]

Roche Publishes the Results of Evrysdi (risdiplam) in FIREFISH Study to Treat of Type 1 SMA at NEJM Published: July 30, 2021 | Tags: Roche, […]

In an interview with PharmaShots, Justin Odegaard, Vice President of Clinical Development at Guardant Health shares his views on the Guardant360 CDx approval as the […]

Shots: Amgen to acquire all outstanding shares of Teneobio for $900M as up front and $1.6B in cash as contingent milestones to Teneobio equity holders. […]

Amgen is paying $900 million up front to acquire Teneobio, a biotech with technology for discovering and developing antibody drugs. Though Amgen already has its […]

Lysogene Receives the US FDA’s Fast Track Designation for LYS-GM101 Gene Therapy to Treat GM1 Gangliosidosis Published: July 9, 2021 | Tags: Lysogene, US, FDA, […]

Shots: The BLA is based on PATHFINDER clinical program, including P-III NAVIGATOR study evaluating Tezepelumab + SOC vs PBO + SOC in 1,061 adults (18–80yrs.) […]

The FDA has started a priority review of AstraZeneca and Amgen’s severe asthma antibody tezepelumab, setting a date for a decision in the first quarter […]

The second quarter of 2021 witnesses major acquisitions, approvals, and clinical data. There are major alliances in this quarter which include Merck signed ~$1.2B supply […]

The US FDA has approved 4 NDAs and 1 BLAs in 2021, leading to treatments for patients and advances in the health care industry. The […]

Dermavant is in line to make some big payments to GlaxoSmithKline if its topical psoriasis drug tapinarof reaches the market, and it has just cut […]

Tonix Pharmaceuticals Presents Results of TNX-102 SL in P-III RELIEF study for the Management of Fibromyalgia at ASCP 2021 Published: June 4, 2021 | Tags: […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency Biosimilars are expected to be a cost-effective […]

Amgen has made a play in a hot area of immunology R&D, paying $400 million upfront to claim rights to an OX40-targeting drug from Kyowa […]

Amgen is paying Kyowa Kirin $400 million to share in the development of a drug that could offer a new approach to treating atopic dermatitis, […]

Shots: Kyowa Kirin to receive ~$400M up front and an additional ~$850M as milestones along with royalties on future global sales. Both companies will share […]

After decades of mostly unsuccessful R&D, Amgen has claimed the first ever regulatory approval for a drug that inhibits KRAS – a cancer target that […]

Shots: The approval is based on the CodeBreaK 100 trial evaluating the efficacy & tolerability of Lumakras (960mg, qd) in 124 patients with KRAS G12C […]

FDA approval of Amgen drug sotorasib (Lumakras) makes the small molecule the first compound authorized by the regulator for treating cancers characterized by a KRAS […]

AzurRx BioPharma Reports Positive Interim P-II Data of MS1819 + PERT for Cystic Fibrosis Patients with Severe Exocrine Pancreatic Insufficiency Published: May 14, 2021 | […]

Shots: The P-III NAVIGATOR study involves assessing Tezepelumab + SOC vs PBO + SOC in adults (18–80 yrs.) & adolescents (12–17 yrs.) with severe uncontrolled […]

Shots: The submission is based on PATHFINDER clinical program including P-III NAVIGATOR that involves assessing Tezepelumab + SOC vs PBO + SOC in 1061 adults […]

AstraZeneca Reports Results of Imfinzi (durvalumab) and Imfinzi + Tremelimumab in P-III POSEIDON Study as 1L Treatment for Stage IV Non-Small Cell Lung Cancer Published: […]

Shots: The acceptance is based on a P-III ADVANCE trial assessing the efficacy of Otezla (30 mg, bid) vs PBO in 595 patients in a […]

A new drug discovery firm, Eikon Therapeutics, has launched with former Merck & Co research chief Roger Perlmutter at the helm. Eikon is backed with […]

A new drug discovery firm, Eikon Therapeutics, has launched with former Merck & Co research chief Roger Perlmutter at the helm. Eikon is backed with […]

Shots: The company reports the availability of Amgevita, referencing Humira to treat 11 chronic inflammatory conditions in Canada Amgevita is available as a prefilled syringe […]

Bristol-Myers Squibb has phase 3 data from its deucravacitinib psoriasis pill, showing it outperformed Amgen’s rival Otezla in patients with moderate to severe disease. The […]

The first quarter of 2021 highlights new approvals, M&A, and the clinical data of COVID-19 vaccines. The companies expand their working in COVID-19 by targeting […]

Boehringer Ingelheim and The University of Texas MD Anderson Cancer Center have extended and expanded a collaboration exploring medicines targeting lung cancer with KRAS mutations. […]

Despite having the global pandemic, the biopharma companies were involved in multiple M&A, licensing deals. The major focus of the year remains the production and […]

Pfizer and BioNTech Report Results of BNT162b2 to Prevent COVID-19 Published: Apr 2, 2021 | Tags: Pfizer and BioNTech, Report, Results, BNT162b2, Prevent, COVID-19 Acceleron […]

Breakthrough Therapy (BT) Designation is an expedited review program introduced in Section 902 of the Food and Drug Safety and Innovation Act of 2012 The […]

Eli Lilly and Incyte Report Results of Olumiant (baricitinib) in P-III BRAVE-AA2 Study for Severe Alopecia Areata Published: Mar 5, 2021 | Tags: Eli Lilly […]

Amgen is to buy US biotech Five Prime Therapeutics for around $1.9 billion, adding a potential gastric cancer drug to its pipeline. The big pharma […]

Shots: Amgen to acquire all of the outstanding shares of Five Prime for $38.00 /share in cash, representing an equity value of ~ $1.9B. The […]

The acquisition brings Amgen a novel Phase 3-ready drug for gastric cancers. For Five Prime Therapeutics, the deal is a lifeline following a series of […]

Shots: The P-III 20120215 study involves assessing of Blincyto vs SoC consolidation CT before alloHSCT in pediatric patients with high-risk first relapsed B-cell ALL. The […]

Shots: The P-III NAVIGATOR study involves assessing Tezepelumab + SOC vs PBO + SOC in 1061 adults (18–80yrs.) & adolescents (12–17yrs.) with severe, uncontrolled asthma, […]

AstraZeneca says full data from the NAVIGATOR trial of tezepelumab in severe asthma keep it on course to file for approval in the first half […]

Regeneron Reports IDMC Clearance of Efficacy for REGEN-COV (Casirivimab + Imdevimab) in P-III COVID-19 Outpatient Outcomes Trial Published: Feb 26, 2021 | Tags: Regeneron, Reports, […]

Shots: The sNDA filing is based on P-III ADVANCE trial data, assessing Otezla (30 mg, bid) vs PBO in 595 patients in a ratio (1:1) […]

How is a Rare disease defined? Well, it depends on the geography in question, since there is no universal definition, even so, the definition revolves […]

The biopharma industry saw numerous deal terminations in 2020. Clinical and regulatory results, change in control limitations, and strategic reprioritizations were among the most common […]

Reistone Report Results for SHR0302 in P-II Study to Treat Ulcerative Colitis Published: Feb 5, 2020 | Tags: Reiston, Report, Result, SHR0302, P-II, Study, Treat, […]

Shots: The BT designation is based on P-II CodeBreaK 100 study assessing Sotorasib in 126 patients with KRAS G12C-mutated locally advanced or metastatic NSCLC who […]

Roche Collaborate with Cambridge Quantum Computing to Develop Quantum Algorithm for Drug Discovery and Development Published: Jan 29, 2020 | Tags: Roche, Collaborate, Cambridge Quantum Computing, […]

Shots: The P-II cohort of the CodeBreaK 100 clinical study involves assessing Sotorasib (AMG 510, 960mg, qd) in 126 patients with KRAS G12C-mutated advanced NSCLC. […]

Biosimilars are developed to be highly similar versions of approved biologics in terms of safety, purity, and potency. Biosimilars are expected to be a cost-effective […]

In its report released last Tuesday, a nonprofit drug pricing research group claims that seven drugs have prices that do not align with any newly […]

Novartis’ Ligelizumab (QGE031) Receives the US FDA’s Breakthrough Designation for Patients with Chronic Spontaneous Urticaria Published: Jan 15, 2020 | Tags: Novartis’, Ligelizumab (QGE031), Receives […]

US cost effectiveness watchdog ICER found 10 examples of substantial price rises for top-selling medicines in 2019, and concluded that seven of those were not […]

Shots: Evoq to receive ~$240M up front & milestones along with royalties on the sales of therapies emerges from the collaboration The companies collaborated on […]

“Lockdown’ declared Collins Dictionary word of the year. The year 2020, well known as COVID-19 year has been a busy year for global pharma and […]

Amgen has decided that a drug for leprosy and tuberculosis it inherited as part of its acquisition of Celgene’s psoriasis blockbuster Otezla last year would […]

AstraZeneca and Amgen looked on course to cruise to approval of their severe asthma drug tezepelumab with a pair of positive clinical trials, but the […]

Sandoz to Launch Hyrimoz (biosimilar, adalimumab) in Canada Published: Dec 17, 2020 | Tags: Authorization, Biosimilar, canada, health, Humira, Hyrimoz, Launch, receives, Sandoz Amgen’s Riabni […]

Shots: The approval is based on trial assessing Riabni (375 mg/m2, IV) vs Rituxan once weekly for 4wks. followed by dosing @12wks. & 20wks. in […]

Amgen has filed its groundbreaking KRAS inhibiting drug sotorasib with the FDA for a group of lung cancer patients with an aggressive form of the […]

Shots: The NDA submission is based P-II CodeBreaK 100 study assessing Sotorasib in patients with KRAS G12C-mutated NSCLC prior treated with CT and immunotherapy The […]

Roche Launches Elecsys SARS-CoV-2 Antigen Test to Support High-Volume COVID-19 Testing Published: Dec 11, 2020 | Tags: Antigen, COVID-19, High, Laboratory, Launch, patients, Roche, SARS-CoV-2, […]

Shots: The BT designation is based on P-II CodeBreaK 100 study assessing Sotorasib in patients with advanced NSCLC with KRAS G12C mutation whose cancer had […]

Shots: Amgen, UCB, and Takeda reported that the first patient has been enrolled in the COMMUNITY trial. The study will test whether Amgen’s Otezla, Takeda’s […]

Qiagen Launches Portable Digital SARS-CoV-2 Antigen Test in the US Published: Nov 13,2020 | Tags: Antigen Test, Digital, Launches, Portable, Qiagen, SARS-CoV-2, US GSK and […]

Shots: The P-III NAVIGATOR study involves assessing Tezepelumab + SOC vs pbo + SOC in adults (18–80yrs.) & adolescents (12–17yrs.) with severe, uncontrolled asthma, who […]

AstraZeneca’s Brilinta (ticagrelor) Receives the US FDA’s Approval to Reduce the Risk of Stroke in Patients with an Acute Ischemic Stroke or High-Risk Transient Ischemic […]

Novartis’ injectable migraine prevention antibody Aimovig has been shown to be more effective than topiramate – a go-to oral therapy for people with chronic migraine […]

Bristol-Myers Squibb could be on the verge of a major coup in psoriasis after its deucravacitinib pill outperformed Amgen’s rival Otezla. BMS had to sell […]

Immunology is an important branch of science which deals with the study of the immune system. The immune system is a highly regulated and balanced […]

Shots: The P-lll GALACTIC-HF study involves the assessing of Omecamtiv mecarbil (25mg, bid with the maintenance dose of 50 mg, 37.5mg, or 25mg, bid) vs […]

Shares in Amgen were down nearly 7% after close of trading yesterday after the company’s heart failure drug omecamtiv mecarbil disappointed in a large phase […]
The company did not provide data, but said results for patients who received sotorasib at the 960mg dose were consistent with those seen in the […]

Shots: The P-ll CodeBreaK 100 study involves assessing of Sotorasib (proposed INN for AMG 510, 960mg) in 126 patients with KRAS G12C-mutant advanced NSCLC, whose […]
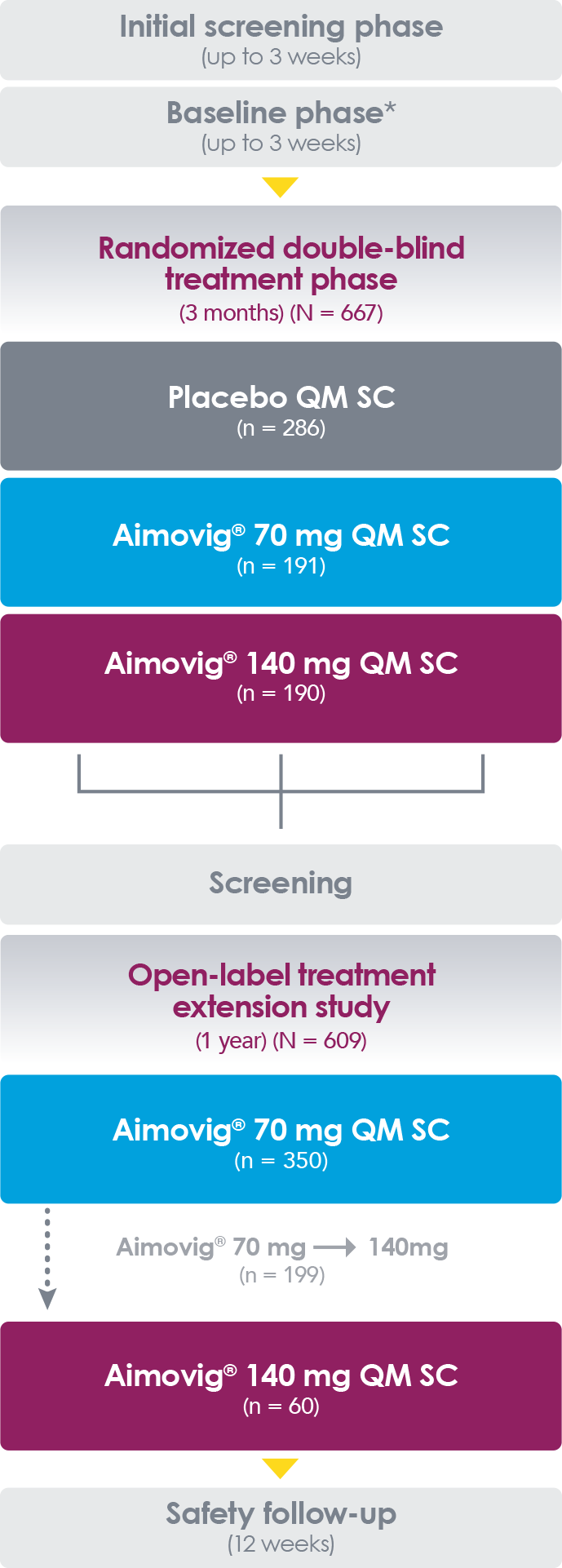
Shots: The OLE P-II study involves assessing of Aimovig monthly (70mg) vs PBO in 383 eligible adult patients with an episodic migraine for 12wks. 250 […]

Three of the companies making drugs used in the Phase II I-SPY COVID-19 study – Amgen, AbbVie and Takeda – announced the patient enrollments Monday. […]
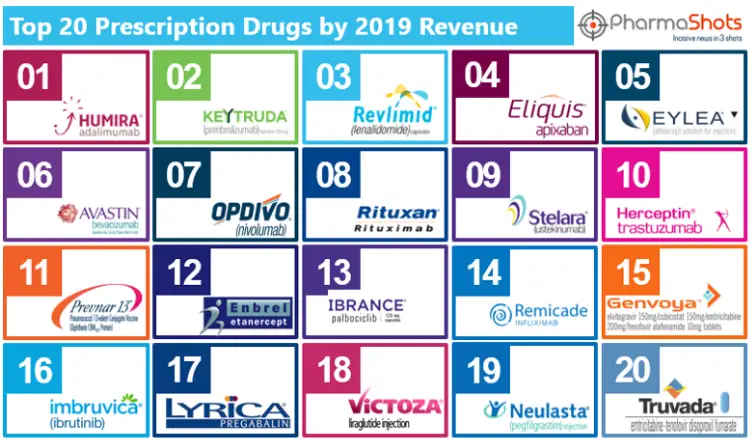
The average life expectancy span of Human Beings are increased due to better medical facilities and drugs developed by Biopharma companies. Pharmaceutical products or drugs […]
Copyright © 2024 | WordPress Theme by MH Themes